Layout of the periodic table
Periods = horizontal rows; groups = vertical columns; blocks = s, p, d, f.
Period number tells you the highest occupied main energy level.
Group number helps identify the number of valence electrons for main group elements.
Be able to locate metals, metalloids and non-metals on the periodic table.
Know these named groups: Group 1 alkali metals, Group 17 halogens, Group 18 noble gases, transition elements.
s-block = groups 1–2, p-block = groups 13–18, d-block = transition elements, f-block = lanthanides + actinides.
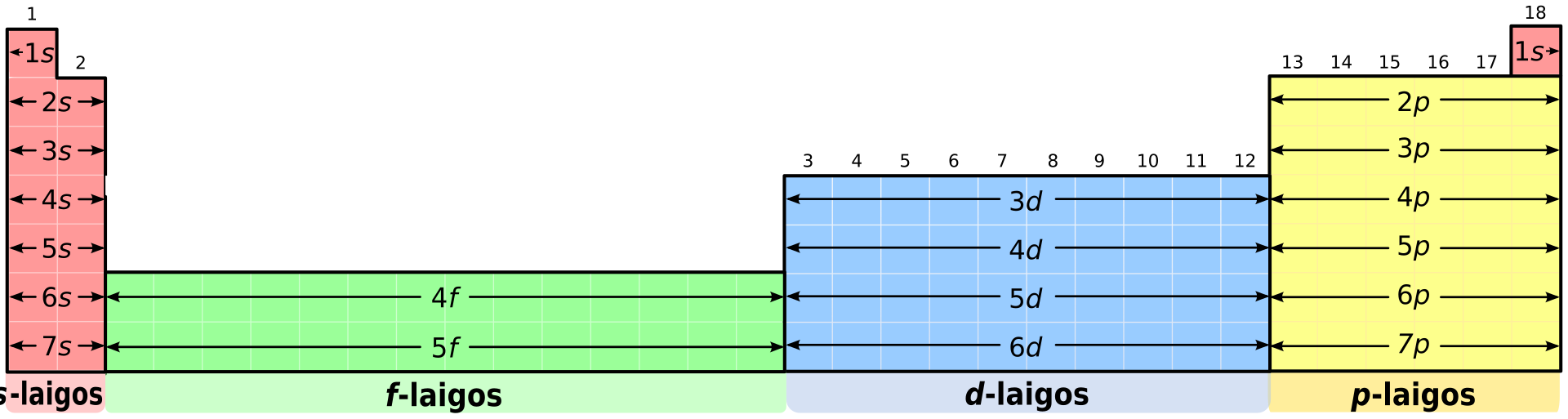
This diagram shows the s-, p-, d- and f-blocks on the periodic table, so it is ideal for linking electron sublevels to table layout. Use it to revise where transition elements and the inner transition elements sit. Source
Position ↔ electron configuration
You must be able to deduce electron configuration from position in the periodic table, and position from electron configuration, for atoms up to .
Period number tells you the outer occupied shell.
For main-group elements, group number gives the number of valence electrons.
Example pattern: Group 1 ends in ; Group 2 ends in .
p-block elements fill orbitals after the s-sublevel.
d-block elements involve filling sublevels.
Exam link: connect sublevel filling to the block structure of the periodic table.
Periodic trends you must explain
Periodicity = repeating trends in properties across periods and down groups.
You must explain trends in: atomic radius, ionic radius, first ionization energy, electron affinity and electronegativity.
Across a period: nuclear charge increases while shielding changes less, so attraction between nucleus and outer electrons usually becomes stronger.
Down a group: distance from nucleus increases and shielding increases, so outer electrons are generally less strongly attracted.
This explains many trends in metallic and non-metallic character.
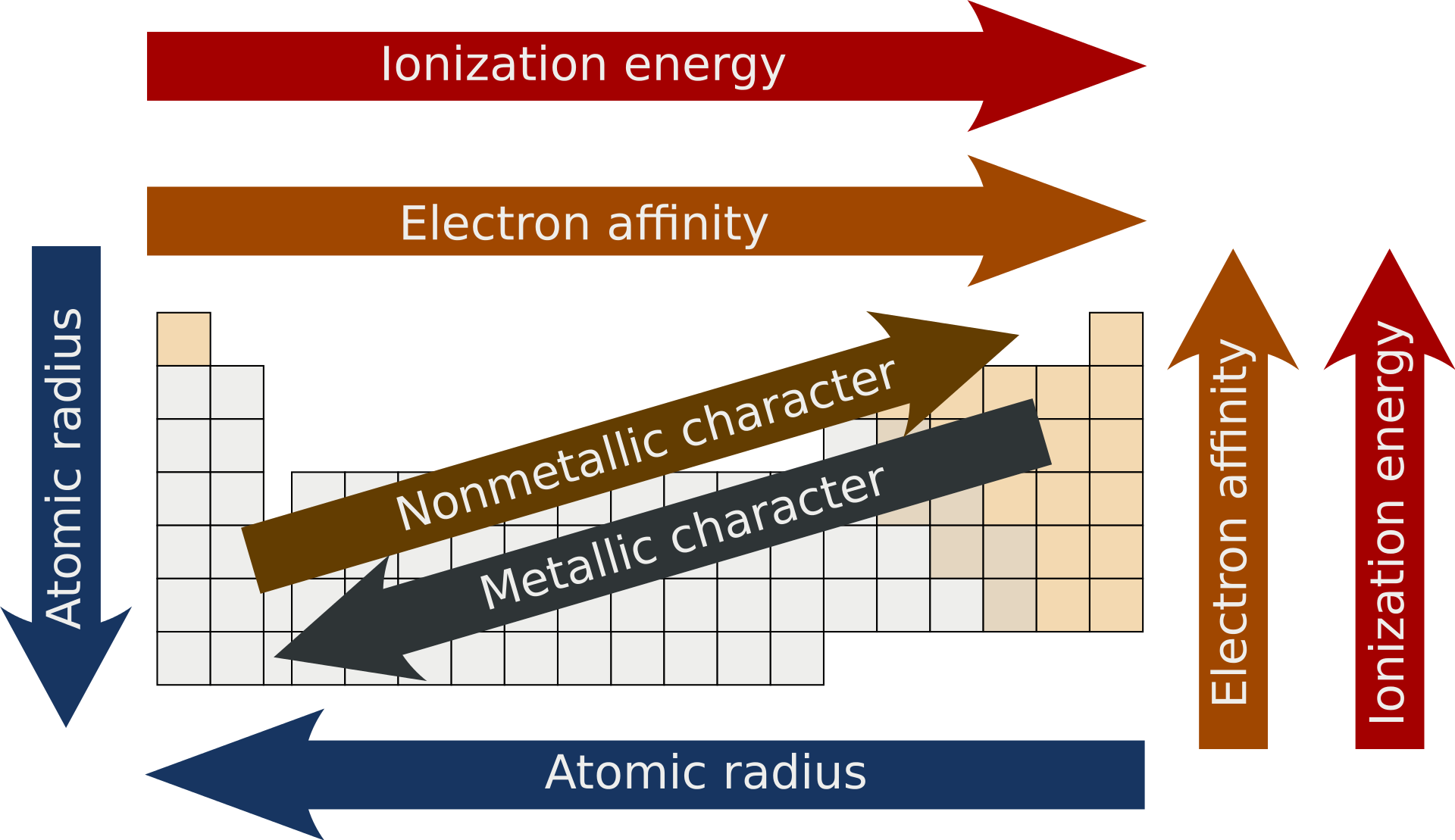
This image summarizes the major periodic trends in one place, including direction changes across periods and down groups. It is useful for comparing atomic radius, ionization energy, electronegativity and related trends at a glance. Source
Atomic radius and ionic radius
Atomic radius generally decreases across a period and increases down a group.
Across a period, greater nuclear charge pulls electrons closer.
Down a group, extra electron shells make atoms larger.
Cations are smaller than their parent atoms because they lose an outer shell or experience greater effective attraction per electron.
Anions are larger than their parent atoms because extra electrons increase electron–electron repulsion.
Within an isoelectronic series, radius decreases as nuclear charge increases.
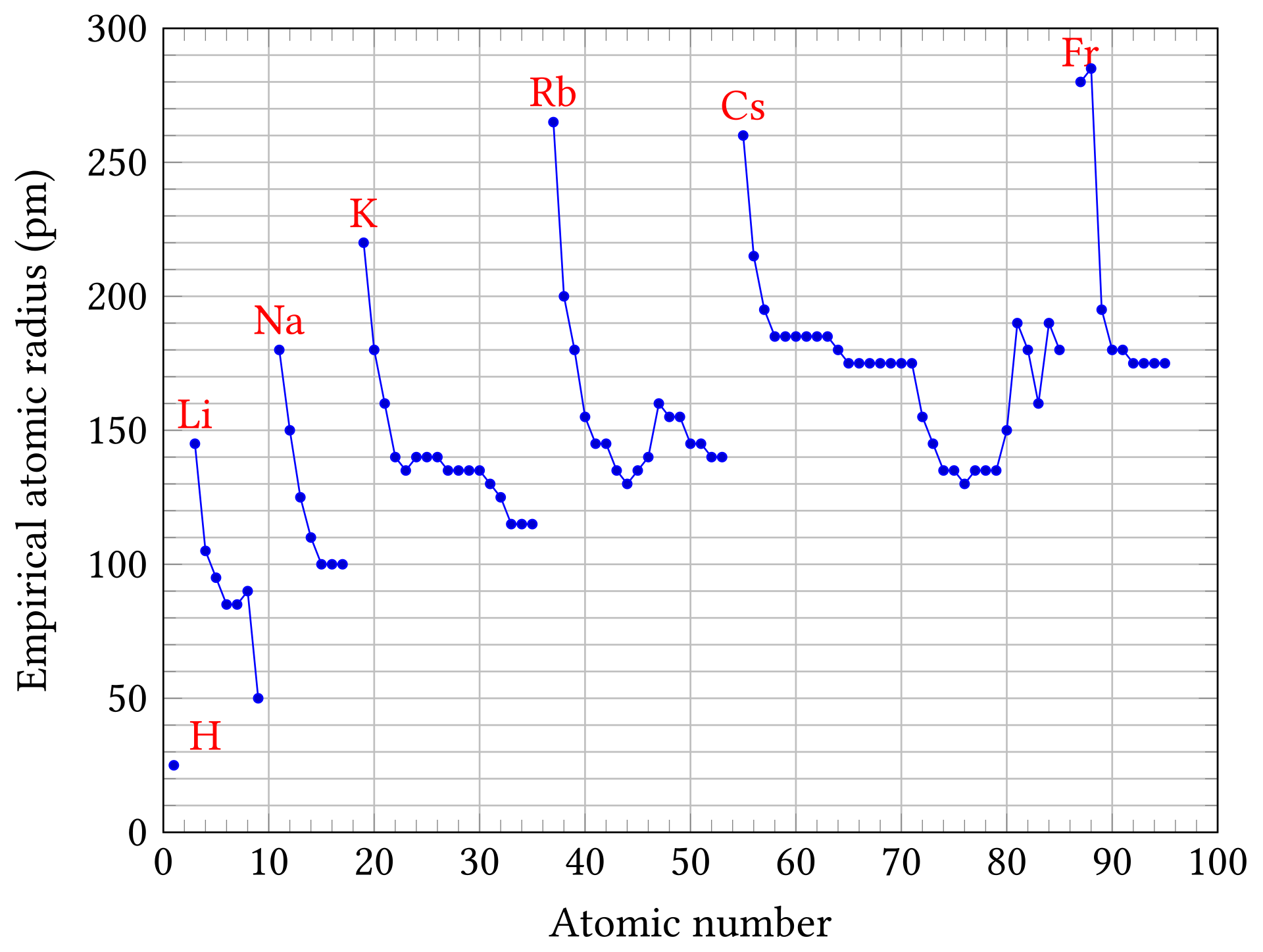
This graph plots atomic radius against atomic number, making the repeating pattern across periods very clear. It helps you see both the overall decrease across a period and the jump up at the start of a new period. Source
Ionization energy, electron affinity and electronegativity
First ionization energy = energy required to remove one electron from each atom in one mole of gaseous atoms.
Ionization energy generally increases across a period and decreases down a group.
Higher nuclear charge and smaller radius increase attraction, so more energy is needed to remove an electron.
More shielding and greater distance from nucleus lower ionization energy down a group.
Electron affinity generally becomes more negative / more exothermic across a period and less so down a group, although there are exceptions.
Electronegativity generally increases across a period and decreases down a group.
The most electronegative elements are toward the top right of the periodic table (excluding noble gases in simple trend discussions).
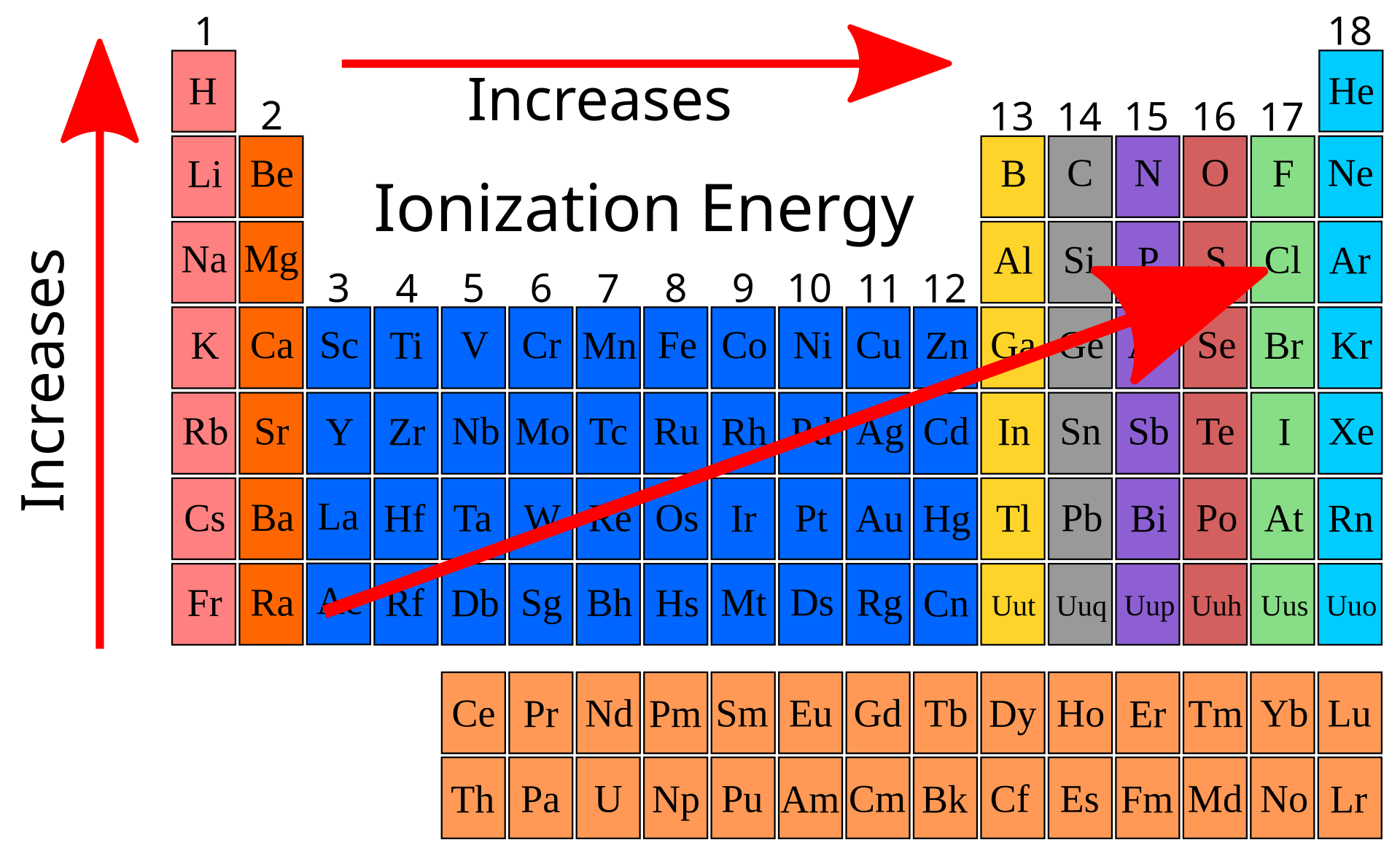
This image shows the direction of increasing ionization energy on the periodic table. It is a quick visual reminder that ionization energy increases up a group and across to the right. Source
Group trends in reactivity
Group 1 metals become more reactive down the group.
Reason: the outer electron is further from the nucleus and more shielded, so it is lost more easily.
Reaction with water: metal + water → metal hydroxide + hydrogen.
General equation: for Group 1 metals.
Group 17 halogens become less reactive down the group.
Reason: larger atoms with more shielding attract an incoming electron less strongly.
A more reactive halogen displaces a less reactive halide ion from solution.
Reactivity order to know: .
Metallic character, non-metallic character and oxides
Metallic and non-metallic properties form a continuum across the periodic table.
Down Group 1, metallic character increases.
Down Group 17, non-metallic character decreases.
Oxides trend from basic metal oxides → amphoteric oxides → acidic non-metal oxides.
Know reactions with water of oxides of Group 1, Group 2, carbon and sulfur.
Basic oxides form alkaline solutions with water.
Acidic oxides form acids in water and are linked to acid rain and ocean acidification.
dissolving in water contributes to ocean acidification.
Sulfur oxides and nitrogen oxides in the atmosphere contribute to acid rain.
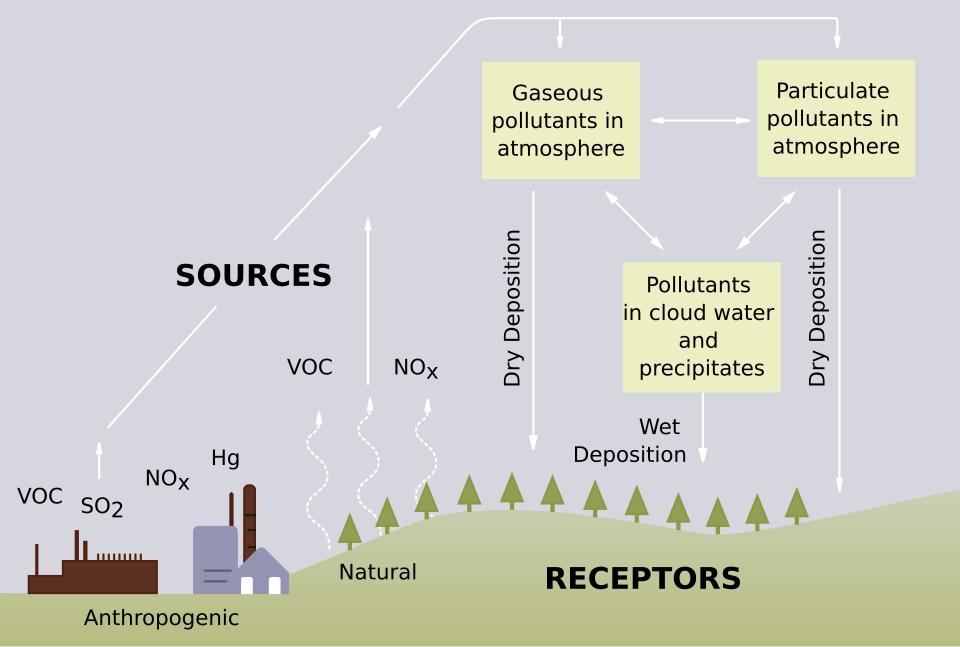
This diagram shows how sulfur dioxide released by burning fuels leads to acid rain after interacting with water in the atmosphere. It helps connect the chemistry of acidic non-metal oxides to environmental consequences tested in IB questions. Source
Oxidation states
Oxidation state = the charge an atom would have if bonding were completely ionic.
The oxidation state of an element in its uncombined form is 0.
You must be able to deduce oxidation states in ions and compounds.
Sum of oxidation states in a neutral compound = 0.
Sum of oxidation states in a polyatomic ion = ion charge.
Important exceptions to know: hydrogen is in metal hydrides; oxygen is in peroxides.
Oxidation states are written as , , etc.
Be ready to use oxidation states to support later work on redox.
Checklist: can you do this?
Identify whether an element is a metal, metalloid, non-metal, and place it in the correct group, period and block.
Deduce an atom’s electron configuration from its position in the periodic table, and the reverse.
Explain trends in atomic radius, ionic radius, ionization energy, electron affinity and electronegativity.
Predict and write equations for Group 1 reactions with water, halogen displacement reactions, and oxide reactions with water.
Calculate / assign oxidation states correctly, including common exceptions.
HL only — discontinuities and transition elements
Across a period, the increase in first ionization energy is not perfectly smooth.
Discontinuities provide evidence for the existence of energy sublevels.
Explanations should focus on the energy of the electron removed, not vague ideas of “special stability”.
Transition elements have incomplete d-sublevels in at least one of their atoms or ions.
Characteristic transition-element properties: variable oxidation states, high melting points, magnetic properties, catalytic properties, coloured compounds, and complex ion formation.
Variable oxidation state occurs because successive ionization energies are relatively close in value.
You must be able to deduce electron configurations of ions of first-row transition elements.
Transition-metal complexes are coloured because electrons are promoted between split d-sublevels.
The colour observed is complementary to the colour absorbed.
Use and the colour wheel to connect absorbed wavelength/frequency to the observed colour.
HL exam traps
Do not say ionization-energy discontinuities are only due to filled or half-filled sublevels being “more stable”; explain using the sublevel/orbital energy of the electron removed.
When writing transition-metal ions, remove electrons from before for first-row transition metals.
A species is not a transition element just because it is in the d-block; focus on incomplete d-sublevels.
For coloured complexes, remember: absorbed colour and observed colour are complementary, not the same.
In oxidation-state questions, check exceptions such as peroxides and metal hydrides before assigning values mechanically.