Dynamic equilibrium
Dynamic equilibrium is reached in a closed system when the rate of the forward reaction = rate of the backward reaction.
At equilibrium, concentrations/macroscopic properties stay constant, but particles are still reacting.
Applies to both physical equilibrium and chemical equilibrium.
In exam wording, constant concentration does not mean the reaction has stopped.

This figure shows dynamic equilibrium: particles continue changing in both directions, but the overall amounts remain constant because the forward and reverse rates are equal. It is a useful visual reminder that equilibrium is dynamic, not static. Source
Equilibrium constant,
The equilibrium law gives an expression for from the stoichiometric coefficients in the balanced equation.
For a general homogeneous reaction, :
Products go on top, reactants on the bottom.
Each concentration is raised to the power of its coefficient.
In this topic, you must be able to deduce the equilibrium expression from the equation.
For IB exam technique: write the balanced equation first, then build the expression carefully from coefficients.
What the size of means
The magnitude of shows the extent of reaction at equilibrium.
→ equilibrium lies far to the left; mostly reactants.
→ reactants favored.
→ appreciable amounts of both reactants and products.
→ products favored.
→ equilibrium lies far to the right; mostly products.
is temperature dependent.
For the reverse reaction at the same temperature: .
Le Châtelier’s principle
Le Châtelier’s principle predicts how an equilibrium system responds to a stress.
The system shifts to oppose the change and re-establish equilibrium.
You must apply this to changes in concentration, pressure, and temperature.
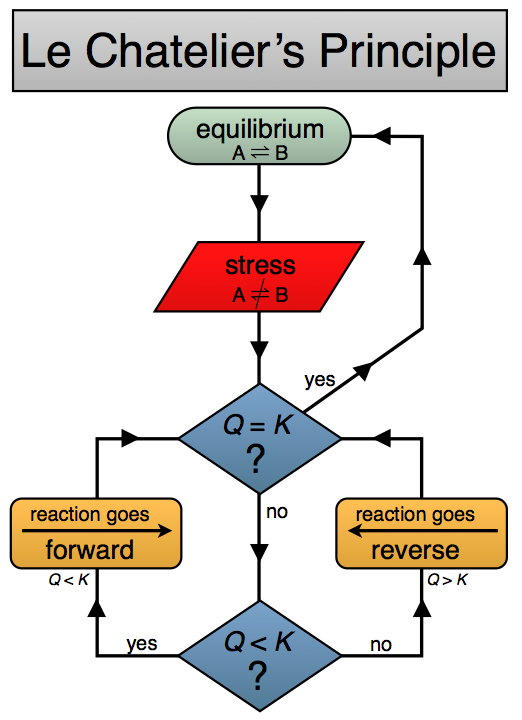
These diagrams summarize how equilibrium shifts when concentration, pressure, or temperature changes. They are helpful for building a fast decision process in exam questions. Source
Changing concentration
Increasing a reactant shifts equilibrium to the right.
Increasing a product shifts equilibrium to the left.
Removing a reactant shifts equilibrium to the left.
Removing a product shifts equilibrium to the right.
does not change when only concentration changes.
The equilibrium composition changes, but the value of stays the same.
Changing pressure
Pressure changes matter only when gases are involved.
Increasing pressure favors the side with fewer moles of gas.
Decreasing pressure favors the side with more moles of gas.
If both sides have the same total moles of gas, changing pressure causes no shift.
does not change when only pressure changes.
Always compare gaseous moles only; ignore solids and liquids.
Changing temperature
Treat heat as a reactant or product.
For an endothermic forward reaction, increasing temperature shifts equilibrium to the right.
For an exothermic forward reaction, increasing temperature shifts equilibrium to the left.
Temperature change is the one common stress that changes the value of .
If the forward reaction is endothermic, increasing temperature increases .
If the forward reaction is exothermic, increasing temperature decreases .
Catalysts and equilibrium
A catalyst speeds up both the forward and backward reactions.
A catalyst helps the system reach equilibrium faster.
A catalyst does not change the value of .
A catalyst does not change the equilibrium composition.
Heterogeneous equilibria
Le Châtelier’s principle can also be applied to heterogeneous equilibria, for example:
Focus on how changing conditions affects the position of equilibrium.
In equilibrium expressions, remember that in many cases pure solids and pure liquids are omitted.
Checklist: can you do this?
State the conditions required for dynamic equilibrium.
Write a correct equilibrium expression from a balanced homogeneous equation.
Interpret the size of to describe whether reactants or products are favored.
Predict the shift caused by a change in concentration, pressure, or temperature.
Distinguish between changes that affect equilibrium composition and changes that affect the value of .
HL only – Reaction quotient,
The reaction quotient, , uses the same form as the equilibrium expression but with non-equilibrium concentrations.
Compare with to predict the direction of change:
→ system is at equilibrium.
→ reaction proceeds forward to make more products.
→ reaction proceeds backward to make more reactants.
This is a high-value exam skill for determining the direction the mixture will shift before equilibrium is reached.
HL only – Solving equilibrium problems
You must solve problems using together with initial and equilibrium concentrations.
A common method is an ICE table:
I = initial concentrations
C = change
E = equilibrium concentrations
Use the stoichiometric ratios to write the change terms.
Substitute equilibrium values into the expression.
In this syllabus, quadratic equations are not expected.
Only homogeneous equilibria are assessed quantitatively here.
HL only – Gibbs energy and equilibrium
and both describe the position of equilibrium.
Use:
Meanings to know:
→ , so products are favored.
→ , so reactants are favored.
→ .
In calculations, use Kelvin for temperature.
Watch units carefully when using and .
Exam traps to avoid
Do not say equilibrium means equal concentrations; it means equal rates.
Do not include substances incorrectly in a or expression.
Do not say a catalyst shifts equilibrium; it only makes equilibrium faster to reach.
Do not confuse changing concentration/pressure with changing ; only temperature changes .
Do not forget that for the reverse reaction is the reciprocal of the forward reaction.
Fast exam summary
Equilibrium = closed system, forward rate = backward rate, constant macroscopic quantities.
Large means products favored; small means reactants favored.
Concentration and pressure can change the position of equilibrium, but not .
Temperature changes both the position of equilibrium and .
HL: use , ICE tables, and to quantify equilibrium.