Covalent bonding and the octet rule
Covalent bond = electrostatic attraction between a shared pair of electrons and the positive nuclei of the bonded atoms.
Octet rule: atoms tend to achieve 8 valence electrons in the outer shell.
Lewis formulas / Lewis structures show all valence electrons, including bonding pairs and lone pairs.
Be able to draw molecules and ions with up to 4 electron pairs around each atom.
Include single bonds, double bonds, triple bonds, and lone pairs correctly.
Not all species obey the octet rule: some have incomplete octets (for example BF₃, BeCl₂).
In Lewis structures, electron pairs may be shown as dots, crosses, or lines.
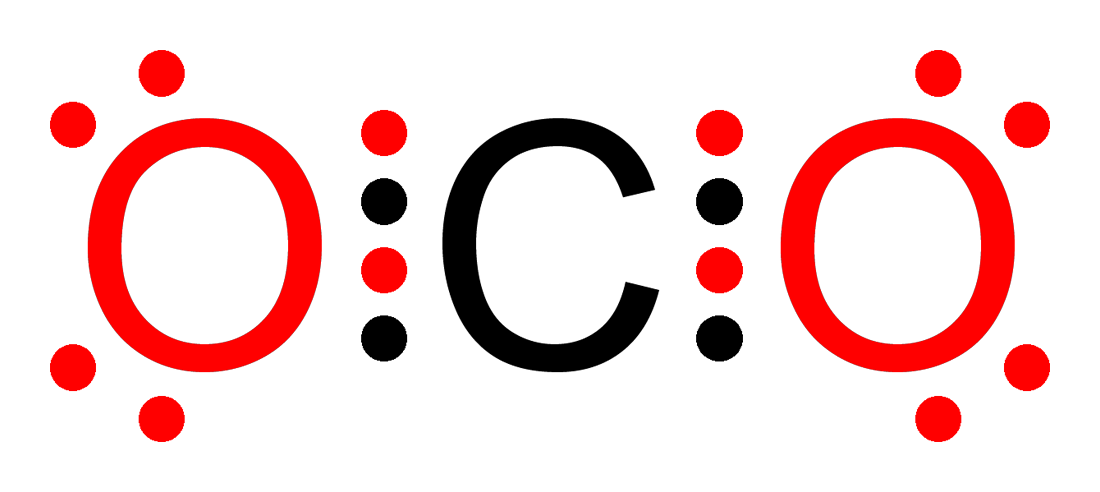
This image shows a Lewis structure with double bonds and lone pairs, useful for practicing octet-rule counting. It helps students see how shared pairs and non-bonding pairs are represented in a standard exam-style diagram. Source
Bond order, bond length and bond strength
Single bond = 1 shared pair of electrons.
Double bond = 2 shared pairs of electrons.
Triple bond = 3 shared pairs of electrons.
As bond order increases, bond length decreases and bond strength increases.
Therefore: triple bonds are generally shorter and stronger than double bonds, which are shorter and stronger than single bonds.
Stronger bonds usually require more energy to break.
Coordinate (dative covalent) bonds
A coordinate bond is a covalent bond in which both electrons in the shared pair come from the same atom.
Identify the electron-pair donor atom in diagrams.
Once formed, a coordinate bond behaves like any other covalent bond.
Common exam skill: identify coordinate bonding in species such as NH₄⁺ or adducts like NH₃ → BF₃.

This image shows a coordinate bond formed when nitrogen donates a lone pair to electron-deficient boron. It is a strong visual example of how a dative bond is represented in Lewis-style structures. Source
VSEPR: electron domain geometry and molecular shape
VSEPR model predicts shape from repulsion between electron domains around a central atom.
Count electron domains: each bonding region counts as 1 domain; each lone pair counts as 1 domain.
For this topic, know species with up to 4 electron domains.
2 electron domains → linear electron geometry, bond angle 180°.
3 electron domains → trigonal planar electron geometry, bond angle 120°.
4 electron domains → tetrahedral electron geometry, bond angle 109.5°.
Distinguish electron domain geometry from molecular geometry.
Lone pairs repel more strongly than bonding pairs, so they reduce bond angles.
Multiple bonds count as one electron domain, but can slightly increase repulsion compared with single bonds.
Typical shapes to recognize:
Linear: CO₂
Trigonal planar: BF₃
Bent: H₂O, SO₂
Tetrahedral: CH₄
Trigonal pyramidal: NH₃
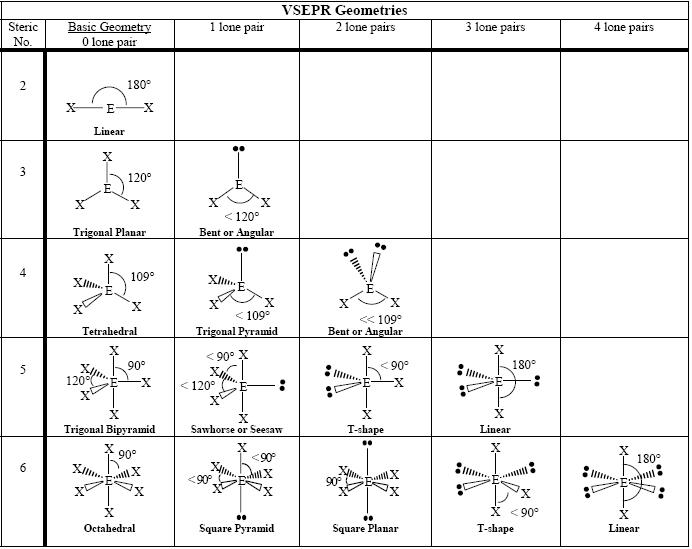
This diagram summarizes electron-domain geometries and the resulting molecular shapes, including the effect of lone pairs on bond angle. It is ideal for matching Lewis structures to 3D geometry in exam questions. Source
Bond polarity
Bond polarity arises from a difference in electronegativity between bonded atoms.
The more electronegative atom attracts the bonding pair more strongly and becomes δ−; the other becomes δ+.
A polar bond can be shown using partial charges or a dipole arrow.
Use electronegativity values from the data booklet to decide whether a bond is non-polar covalent or polar covalent.
Molecular polarity
Molecular polarity depends on both:
bond polarity
molecular geometry
A molecule is polar if bond dipoles produce a net dipole moment.
A molecule is non-polar if bond dipoles cancel out because of a symmetrical shape.
Always do both steps in exams:
decide whether the bonds are polar
decide whether the shape cancels the dipoles
Common contrasts:
CO₂ = polar bonds but non-polar molecule because it is linear
H₂O = polar bonds and polar molecule because it is bent
CCl₄ = polar bonds but non-polar molecule because it is symmetrical tetrahedral
NH₃ = polar molecule because it is trigonal pyramidal
Covalent network structures and allotropes
Carbon and silicon can form giant covalent / covalent network structures.
These have strong covalent bonds throughout the structure, so they usually have very high melting points and are hard.
Diamond:
each carbon forms 4 covalent bonds
tetrahedral 3D network
very hard
does not conduct electricity because there are no delocalized electrons
Graphite:
each carbon forms 3 covalent bonds
arranged in layers of hexagons
one electron per carbon is delocalized
conducts electricity
layers are held together by weak intermolecular forces, so they can slide
Graphene:
single layer of graphite
strong, light, and conductive
Fullerenes:
molecular forms of carbon with hollow cage / tube-like structures
properties differ from diamond and graphite because structure differs
Silicon and silicon dioxide:
giant covalent structures
high melting points
generally do not conduct electricity as solids
Allotropes = different structural forms of the same element with different bonding/arrangements, so they have different physical properties.

This image compares two allotropes of carbon and links their structure to their properties. It is especially helpful for explaining why diamond is hard while graphite conducts and has slippery layers. Source
Intermolecular forces (IMFs)
Intermolecular forces are attractions between molecules, not the covalent bonds within molecules.
Types required:
London (dispersion) forces
dipole-induced dipole forces
dipole–dipole forces
hydrogen bonding
van der Waals forces is an inclusive term for London, dipole-induced dipole, and dipole–dipole forces.
London (dispersion) forces:
present in all molecules
only IMF in non-polar molecules
increase with molecular size / number of electrons / polarizability
Dipole–dipole forces:
occur between polar molecules
stronger than London forces for molecules of similar size
Dipole-induced dipole forces:
occur when a polar molecule induces a temporary dipole in a non-polar molecule
Hydrogen bonding:
strongest IMF in this topic
occurs when H is covalently bonded to N, O or F and is attracted to a lone pair on N, O or F of a neighbouring molecule
General strength trend for comparable molar mass:
London (dispersion) < dipole–dipole < hydrogen bonding
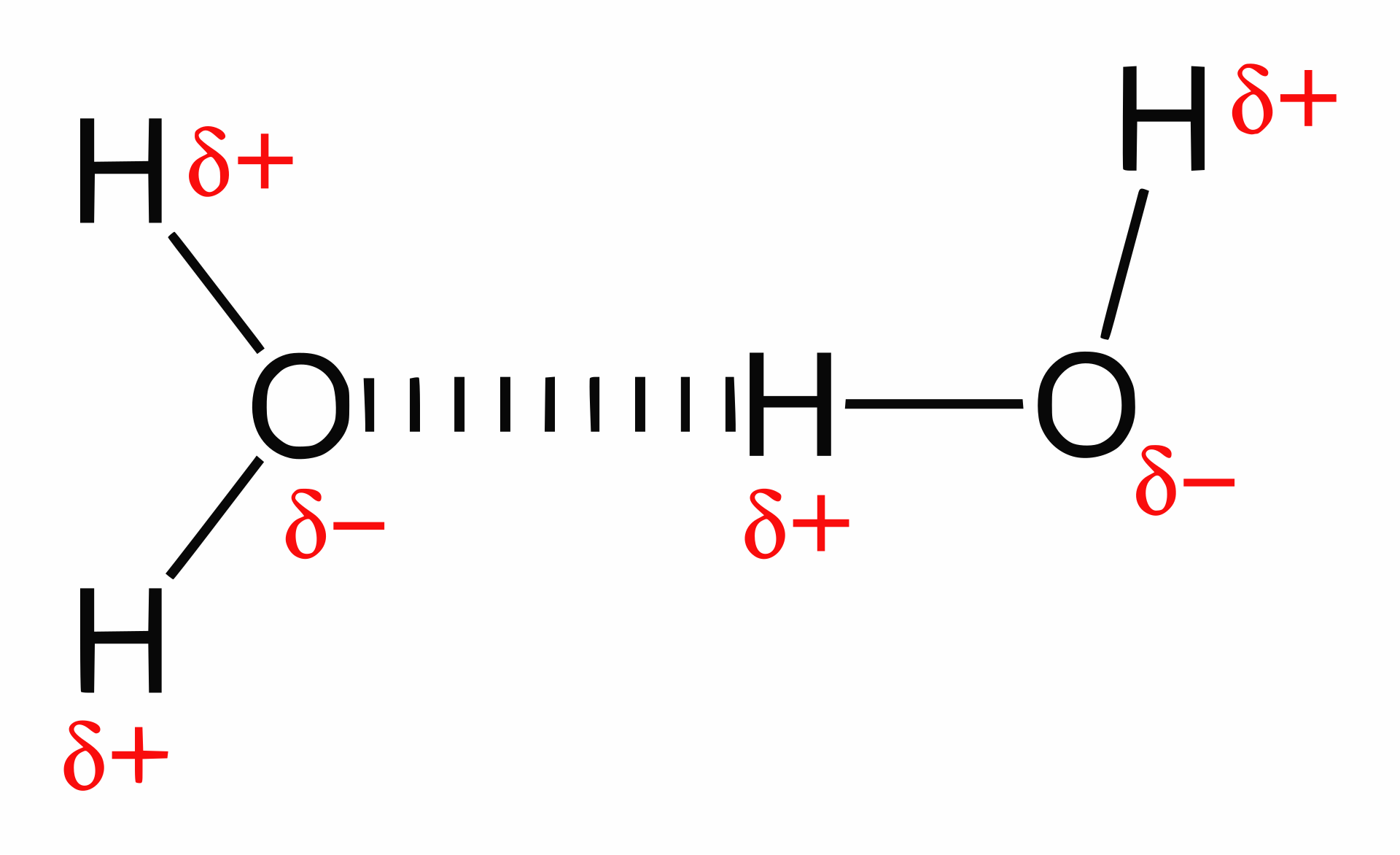
This image shows hydrogen bonds between water molecules, distinguishing them from the covalent O–H bonds inside each molecule. It is useful for explaining why hydrogen bonding gives water unusually strong intermolecular attraction. Source
How structure explains physical properties of covalent substances
Simple molecular covalent substances usually have:
low melting and boiling points because only intermolecular forces are overcome
poor electrical conductivity because they have no mobile charged particles
Covalent network substances usually have:
very high melting points because covalent bonds must be broken
hardness due to strong 3D bonding
variable conductivity depending on whether delocalized electrons are present
Volatility decreases as intermolecular forces become stronger.
Solubility rule of thumb: like dissolves like.
Polar substances tend to dissolve in polar solvents
Non-polar substances tend to dissolve in non-polar solvents
Stronger IMFs generally give higher boiling points and lower volatility.
Chromatography and
Chromatography separates mixture components based on their different attractions to the stationary phase and the mobile phase.
Stronger attraction to the stationary phase → substance moves less.
Stronger attraction to the mobile phase → substance moves further.
Retardation factor:
values have no units.
Same substance should have the same only under the same conditions.
Exam skills:
calculate
compare values
interpret separation quality
relate movement to intermolecular forces with stationary/mobile phases

This page explains how is calculated in TLC/paper chromatography and how it is used to compare components of a mixture. It helps link the practical calculation directly to relative attraction for stationary and mobile phases. Source
Checklist: can you do this?
Draw Lewis structures for molecules and ions, including lone pairs, multiple bonds, and species with an incomplete octet.
Predict electron-domain geometry and molecular shape for species with up to 4 electron domains, and explain bond-angle changes caused by lone pairs.
Determine bond polarity and molecular polarity using electronegativity plus shape.
Identify and compare intermolecular forces, then use them to explain boiling point, volatility, conductivity, and solubility.
Calculate and interpret values from chromatography data.
HL only: resonance and delocalization
Resonance structures exist when there is more than one possible position for a double bond.
The real structure is a hybrid, not switching between drawings.
Delocalization means electrons are spread over more than two atoms.
Be able to deduce resonance structures for molecules and ions.
Benzene, C₆H₆, is the key example:
six carbons in a hexagonal ring
all C–C bonds are the same length
electrons are delocalized around the ring
this explains its extra stability and why it is less reactive than a normal alkene
HL only: expanded octets and formal charge
Some atoms can have an expanded octet, especially species with 5 or 6 electron domains around the central atom.
Be able to draw Lewis structures and predict shapes for 5-domain and 6-domain species.
Know the extra VSEPR electron-domain geometries:
5 domains → trigonal bipyramidal
6 domains → octahedral
Formal charge helps choose the preferred Lewis structure.
Use: formal charge = valence electrons − non-bonding electrons − 1/2(bonding electrons)
Preferred structures usually minimize:
magnitude of formal charges
charge separation
Negative formal charge is usually more favourable on the more electronegative atom.
HL only: sigma bonds, pi bonds and hybridization
Sigma bond () = head-on overlap of orbitals; electron density lies along the bond axis.
Pi bond () = sideways overlap of p orbitals; electron density lies above and below the bond axis.
Single bond = 1
Double bond = 1 + 1
Triple bond = 1 + 2
Hybridization = mixing of atomic orbitals to form new bonding orbitals.
Required types:
sp → linear, 180°
sp² → trigonal planar, 120°
sp³ → tetrahedral, 109.5°
Be able to link:
Lewis structure
number of electron domains
molecular geometry
hybridization
Quick mapping:
2 domains → sp
3 domains → sp²
4 domains → sp³
Exam traps to avoid
Do not confuse intramolecular covalent bonds with intermolecular forces.
Do not decide molecular polarity from bond polarity alone; always check shape.
Remember: multiple bonds count as one electron domain in VSEPR.
Hydrogen bonding only occurs when H is bonded to N, O or F.
In chromatography, use the distance to the centre of the spot, and divide by the distance travelled by the solvent front.