Big picture
Functional groups are the key structural features that give organic molecules their characteristic physical properties and chemical reactions.
Organic compounds are classified by the functional group(s) present.
A homologous series is a family of compounds with the same functional group, same general formula, similar chemical properties, and successive members differing by .
Saturated compounds contain only C-C single bonds; unsaturated compounds contain C=C and/or C≡C.
For exam questions, always identify: functional group, homologous series, saturation, possible isomerism, and intermolecular forces.
Representations of organic compounds
Know and interconvert molecular, empirical, full structural, condensed structural, and skeletal formulas.
Skeletal formulas: each line end / vertex = a carbon atom unless another atom is shown.
In skeletal formulas, H atoms on carbon are omitted; heteroatoms like O, N, Cl, Br must be shown.
Be able to move between molecular formula ↔ structural formula ↔ skeletal formula quickly.
Use 3D models or mental visualization to check connectivity and, at HL, spatial arrangement.
Functional groups you must know
Alkanes: only C-C single bonds; general formula .
Alkenes: contain C=C; general formula .
Alkynes: contain C≡C; general formula .
Halogenoalkanes: contain C-X where X = F, Cl, Br, I.
Alcohols: contain hydroxyl group, -OH.
Aldehydes: contain terminal carbonyl, -CHO.
Ketones: contain internal carbonyl, >C=O.
Carboxylic acids: contain carboxyl group, -COOH.
Ethers: contain alkoxy linkage, R-O-R'.
Amines: contain amino group, -NH2 / -NHR / -NR2.
Amides: contain amido group, -CONH2 / -CONHR / -CONR2.
Esters: contain -COO-.
Phenyl: , derived from benzene ring.
Key distinction: carbonyl compounds include aldehydes and ketones; then related carbonyl-containing groups include carboxylic acids, esters, amides.
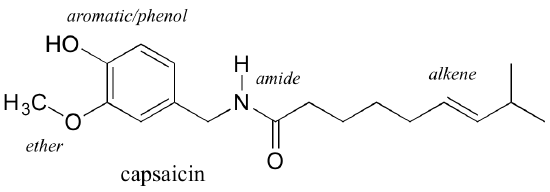
This image shows how different functional groups can be recognized within one organic molecule, reinforcing that classification depends on identifiable structural patterns. Source
Homologous series to identify
Know these series by name, formula pattern, and functional group: alkanes, alkenes, alkynes, halogenoalkanes, alcohols, aldehydes, ketones, carboxylic acids, ethers, amines, amides, esters.
Members of the same homologous series have similar chemical reactions because they share the same functional group.
Successive members have gradual changes in physical properties because molar mass and London dispersion forces increase.
When asked to identify a series, focus first on the highest-priority functional group visible.
Trends in physical properties
Within a homologous series, melting point and boiling point usually increase as chain length / molar mass increases.
Reason: larger molecules have stronger London dispersion forces.
Functional group matters because it changes the type/strength of intermolecular forces.
Compounds with O-H or N-H can show hydrogen bonding, so they often have higher boiling points than similar-sized molecules without these groups.
Branching usually lowers boiling point because it reduces surface contact between molecules.
Solubility in water depends on balance between polar functional group and non-polar hydrocarbon chain.
Small alcohols, carboxylic acids, and some amines are more water-soluble; increasing hydrocarbon chain length usually decreases solubility.
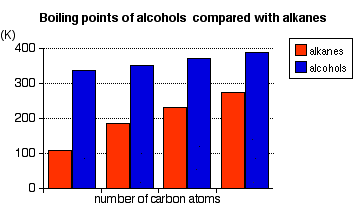
This chart shows that boiling points rise with chain length and that alcohols boil at much higher temperatures than comparable alkanes. It is useful for linking homologous series trends to intermolecular forces, especially hydrogen bonding. Source
IUPAC naming: exam method
IB requires naming of saturated or mono-unsaturated compounds with up to 6 carbons in the parent chain.
Only one type of these functional groups is required at SL: halogeno, hydroxyl, carbonyl, carboxyl.
Step 1: choose the longest parent chain containing the highest-priority functional group.
Step 2: number the chain to give the functional group the lowest possible number.
Step 3: identify and number substituents.
Step 4: use the correct prefix / suffix.
Common suffixes: -ane, -ene, -yne, -ol, -al, -one, -oic acid.
Halogens are named as prefixes: fluoro-, chloro-, bromo-, iodo-.
Write numbers with hyphens and multiple substituents with di-, tri-, etc.
Straight-chain and branched-chain isomers must both be recognized and named.
Structural isomerism
Structural isomers have the same molecular formula but different connectivities.
Types you must recognize:
Chain isomers: different carbon skeleton.
Position isomers: same functional group, different position.
Functional group isomers: same molecular formula, different functional group.
Also know primary (1°), secondary (2°), and tertiary (3°) forms for alcohols, halogenoalkanes, and amines.
For alcohols / halogenoalkanes / amines:
Primary = carbon attached to the functional group is bonded to 1 other carbon.
Secondary = bonded to 2 other carbons.
Tertiary = bonded to 3 other carbons.
In questions, first check whether compounds have the same molecular formula, then compare connectivity.
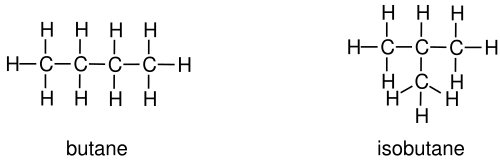
This image shows butane and isobutane as chain isomers with the same molecular formula but different carbon connectivity. It is a clean visual for distinguishing straight-chain and branched structures. Source
Checklist: can you do this?
Identify the functional group and homologous series from a displayed structure.
Convert between molecular, structural, and skeletal formulas.
Name simple organic compounds using IUPAC rules up to 6 carbons.
Explain trends in boiling point / solubility using intermolecular forces, chain length, and branching.
Recognize chain, position, and functional group isomers, including 1° / 2° / 3° examples.
HL only - stereoisomerism
Stereoisomers have the same atom connectivity but different spatial arrangement.
Cis-trans isomerism occurs when there is restricted rotation, most commonly around a C=C bond or in some cycloalkanes.
To get cis-trans isomerism in a non-cyclic alkene, each carbon of the C=C must have two different groups attached.
Cis = similar / chosen groups on the same side; trans = on opposite sides.
A chiral carbon is bonded to 4 different groups.
Chirality gives enantiomers: non-superimposable mirror images.
Enantiomers have identical ordinary physical properties except for their effect on plane-polarized light and behavior in chiral environments.
Racemic mixture = equal amounts of both enantiomers; overall optically inactive.
Be able to recognize wedge-dash formulas showing tetrahedral arrangement around a chiral carbon.
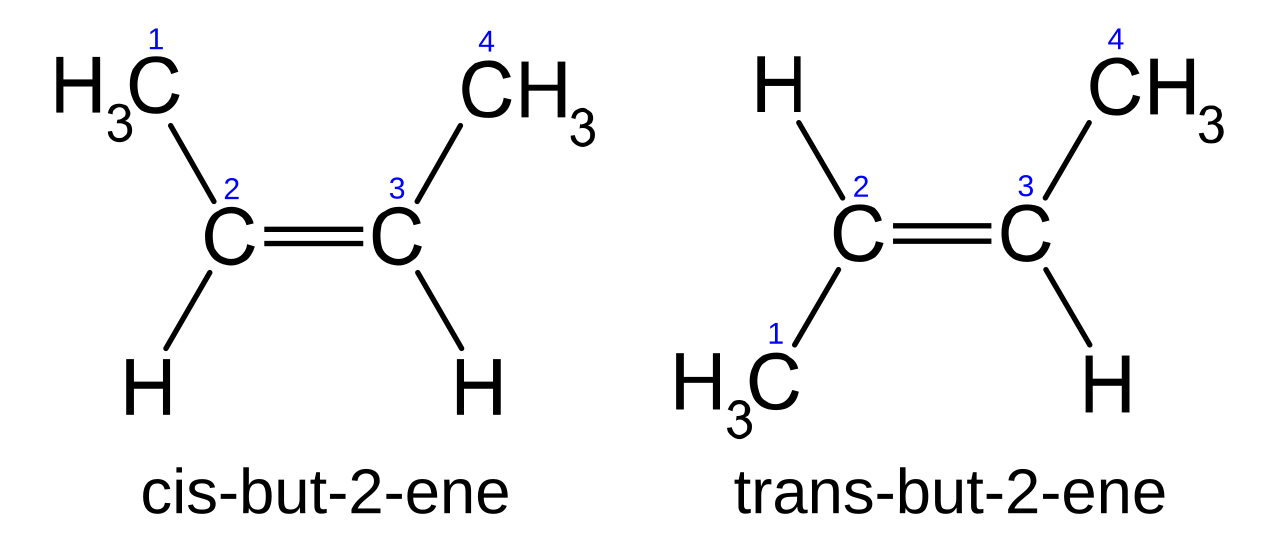
This image shows how identical connectivity can still give different spatial arrangements across a double bond. It is useful for spotting when cis-trans isomerism is possible in alkenes. Source
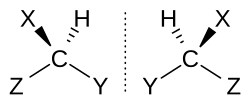
These diagrams illustrate a chiral carbon bonded to four different groups, giving rise to non-superimposable mirror images. They are helpful for visualizing why wedge-dash drawings matter at HL. Source
HL only - spectroscopy for structure determination
Mass spectrometry (MS): identifies molecular ion and useful fragmentation patterns.
The molecular ion peak gives the relative molecular mass.
Fragments help infer structural features; use the data booklet fragment values rather than memorizing extra ones.
Infrared (IR): used to identify bond types / functional groups from the functional group region.
Key IR idea: look for broad / sharp absorptions characteristic of groups such as O-H, N-H, C=O, C=C, C-H.
NMR: gives the number of different proton environments.
Chemical shift tells you the type of environment.
Integration gives the relative number of H atoms in each environment.
Splitting: know singlet, doublet, triplet, quartet.
Use the rule as the high-yield pattern guide: a proton set is split by neighboring H atoms into peaks.
The strongest HL skill is combining molecular formula + MS + IR + NMR to deduce one structure.
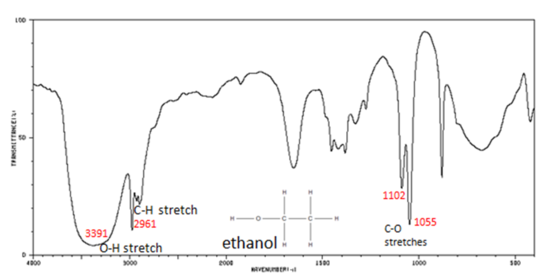
This chart shows where common functional groups absorb in the IR spectrum, especially diagnostic regions such as O-H and C=O. It is useful for rapid HL identification of the main bonds present in an unknown organic molecule. Source
High-yield exam traps
Do not confuse aldehyde with ketone: aldehyde is terminal, ketone is internal.
Do not confuse ether with ester: ester has plus O, ether has O only.
Alcohol and carboxylic acid both contain O-H, but acids also contain .
A molecular formula alone does not prove one structure; always check for isomers.
In naming, number from the end nearest the highest-priority functional group, not the nearest branch.
In HL spectra questions, never rely on one technique alone; combine all evidence.
One-line memory bank
Functional group controls chemistry; carbon chain controls size effects.
Same functional group = homologous series; same formula + different connectivity = structural isomers.
Longer chain -> stronger dispersion forces -> higher boiling point.
Hydrogen bonding -> unusually high boiling point / higher water solubility.
HL structure determination = use MS, IR, and NMR together.