AP Syllabus focus: ‘For ideal gases, macroscopic properties are related by the ideal gas law, PV = nRT, connecting pressure, volume, temperature, and amount of gas.’
Gases are often modelled with a simple mathematical relationship that links measurable bulk properties. Mastering how to use lets you predict how changing one gas property affects the others under idealized conditions.
The Ideal Gas Model and State Variables
An ideal gas is a simplified model in which gas particles are treated as point-like and in constant random motion, with no attractions and perfectly elastic collisions. In this model, the gas is fully described by four state variables: pressure, volume, temperature, and amount.
Ideal gas law: A relationship for an ideal gas that connects pressure, volume, temperature, and moles through .
This law is used to relate macroscopic measurements (like a lab-measured pressure and volume) to the microscopic count of particles through moles.
Core Equation and Meaning of Each Quantity
The ideal gas law is the central tool for connecting gas properties.
= pressure of the gas (commonly atm, kPa, or Pa)
= volume of the gas (commonly L or m)
= amount of gas in moles (mol)
= ideal gas constant (value depends on units used for and )
= absolute temperature (K)
A key requirement is unit consistency: the numerical value chosen for must match the units used for and , and must be in Kelvin.
Temperature must be Kelvin
Because is proportional to average particle kinetic energy in this model, temperature is measured on an absolute scale.
Convert with
Never use Celsius directly in
Choosing a compatible value of
Common pairings include:
for in atm and in L
for energy relationships, noting
Using to Relate Gas Properties
The strength of is that it directly links how one property must respond when another changes, as long as the sample behaves ideally.
Solving for an unknown state variable
You can rearrange algebraically depending on what is unknown:
Moles: (connects measured and to amount of gas)
Pressure: (pressure increases with more moles or higher temperature; decreases with larger volume)
Volume: (volume expands as temperature or moles increase; compresses as pressure increases)
Temperature: (relates thermal conditions to measured and for a known amount)
Reasoning about proportional changes (qualitative)
When the amount of gas is fixed, the ideal gas law implies:
At constant , is inversely proportional to (compressing a gas raises its pressure)
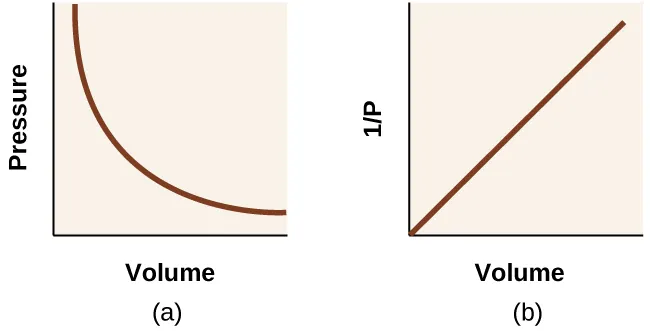
These graphs illustrate Boyle’s law for a fixed amount of gas at constant temperature. Plotting versus produces a hyperbola, while plotting versus linearizes the same inverse relationship and makes trends easier to read quantitatively. Together, they reinforce that remains constant when and are held constant. Source
At constant , is directly proportional to (heating a sealed rigid container raises pressure)
At constant , is directly proportional to (heating a balloon increases volume if pressure stays near constant)
When the container conditions are fixed, changing has predictable effects:
At constant and , increasing increases (more particles colliding with the walls)
High-Utility Workflow and Common Pitfalls
Efficient setup steps
Identify knowns and unknown; write the target rearranged form first
Convert all quantities to compatible units before substituting
Track significant figures based on measured inputs
Common errors to avoid
Using Celsius instead of Kelvin (systematic and large error)
Mixing units (e.g., in kPa with in L·atm·mol·K)
Forgetting that is the gas volume (not necessarily container volume if gas is collected differently)
FAQ
$R$ is the same constant expressed in different unit systems.
Common forms include:
$0.082057\ \text{L·atm·mol}^{-1}\text{·K}^{-1}$
$8.314\ \text{Pa·m}^3\text{·mol}^{-1}\text{·K}^{-1}$
Yes.
Rearrange to find moles, then use $M=\dfrac{m}{n}$, where $m$ is the measured mass. This requires accurate $P$, $V$, and $T$ data.
Combine $n=\dfrac{m}{M}$ with $PV=nRT$ to obtain $d=\dfrac{PM}{RT}$, where $d=\dfrac{m}{V}$.
This is useful for comparing densities at different $P$ and $T$.
$^\circ\text{C}$ to K: add 273.15
L to m$^3$: divide by 1000
kPa to Pa: multiply by 1000
It is most reasonable at relatively low pressure and high temperature, where particle volume and attractions have minimal effect on measured $P$ and $V$.
Practice Questions
(2 marks) State the ideal gas law and name the four macroscopic variables it relates.
(1)
Identifies , , , and (1)
(5 marks) A sample of gas has , , and . Calculate the amount of gas in moles, using .
Converts to Kelvin: (1)
Converts to Pa: (1)
Converts to m: (1)
Uses with correct substitution (1)
Correct final with appropriate rounding/units (1)

