AP Syllabus focus: ‘Use coefficients and formulas to map symbols to particles and relative amounts, keeping the particulate model consistent with the equation.’
Chemical equations are compact “maps” of reacting particles. This page focuses on decoding symbols into particle-level meaning and connecting those particle ratios to moles and other quantity statements without changing the chemistry.
Connecting symbols to particles and quantities
Symbols that control particle identity and count
A chemical formula tells you what a particle is made of, while numbers in front of formulas tell you how many of those particles are involved in the reaction “set” represented by the equation.
Coefficient: The whole number written before a chemical formula that indicates the relative number of particles (molecules, formula units, or moles) participating.
Key symbol-to-particle links:
Coefficient (front number) → counts particles as groups (e.g., 2 H₂O means two water molecules).
Subscript (inside formula) → counts atoms/ions within one particle (e.g., H₂O has 2 H atoms per molecule).
Parentheses and subscripts → multiply internal groups (e.g., Ca(OH)₂ has 2 O and 2 H per formula unit).
State symbols ((s), (l), (g), (aq)) → indicate physical form; they affect the particulate picture (separated ions in aqueous solution vs packed ions in a solid).
Coefficients create particle ratios (the particulate “recipe”)
In any balanced equation, coefficients define the simplest whole-number ratio of reacting particles.
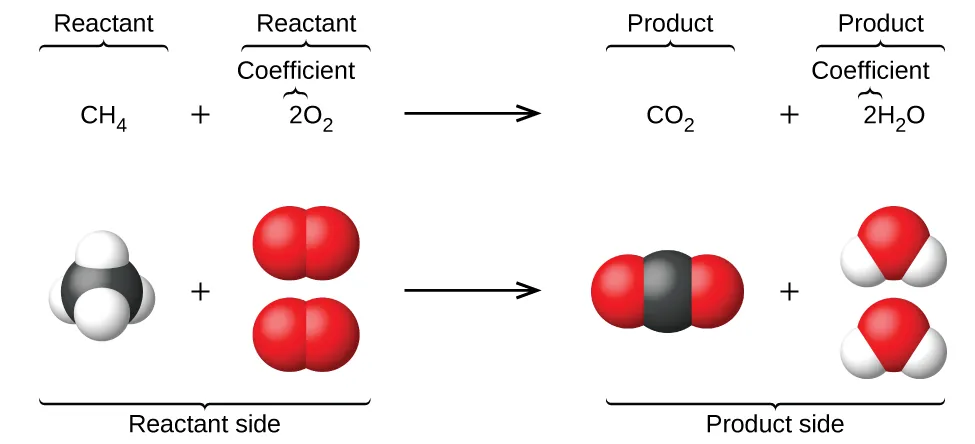
Balanced equations encode particle ratios, not just symbols. This diagram shows the reaction alongside space-filling models, making it clear that the coefficient multiplies whole molecules while subscripts describe atoms within each molecule. Source
A correct particulate model must reflect that ratio while also showing the correct composition of each particle from its formula.
To keep the model consistent:
Use coefficients to set how many of each particle appear.
Use formulas to set what each particle contains (types and counts of atoms).
Ensure the drawn particles can be rearranged to match products without creating or destroying atoms.
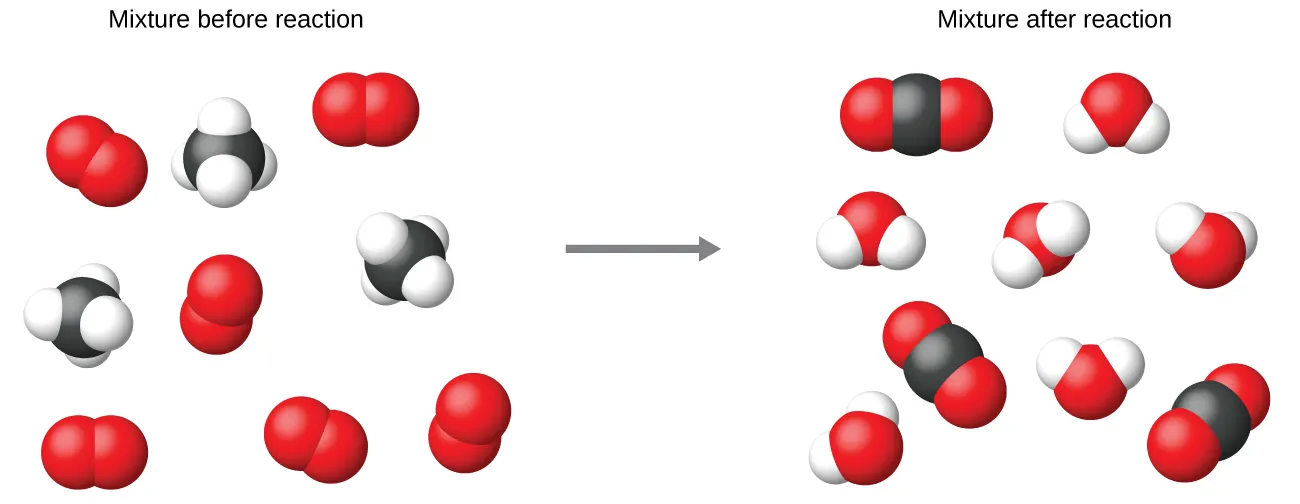
A particulate model should conserve each type of atom even as bonds are rearranged. This before/after particle diagram illustrates that the same total numbers of atoms are present on both sides, while the molecules are regrouped into products in the stoichiometric ratio implied by the balanced equation. Source
Because coefficients are relative, the same equation can be pictured at different scales:
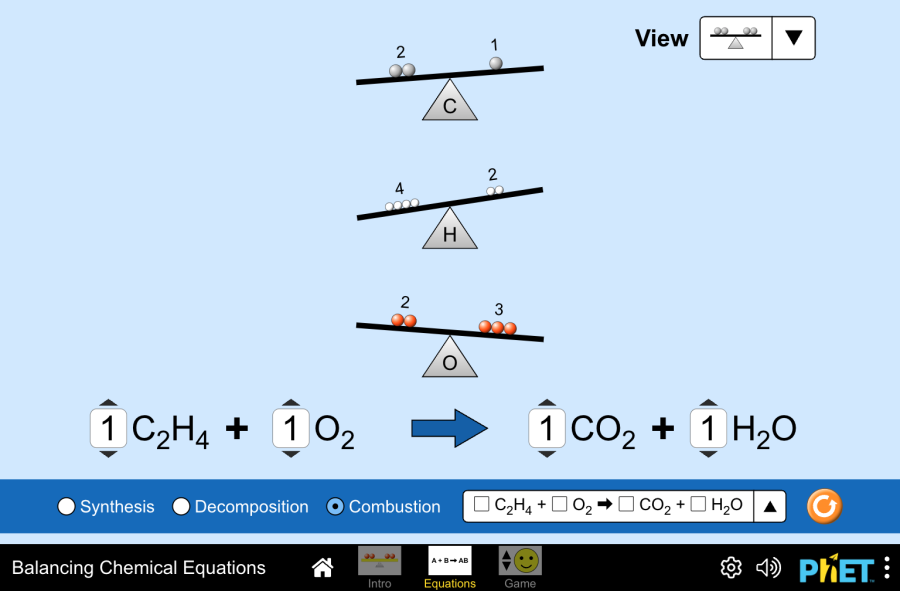
Interactive balancing visuals make the “relative scale” idea concrete: changing coefficients scales the number of whole particles while preserving the simplest ratio. In the PhET Balancing Chemical Equations simulation, you adjust coefficients (not subscripts) until atom counts match on both sides, reflecting conservation at the particulate level. Source
“1 set” of the reaction (using the written coefficients)
2 sets, 10 sets, or any multiple, as long as the ratios are unchanged
Important mapping idea: coefficients apply equally well to molecules, formula units, and moles. The particulate model should therefore match both:
a microscopic count statement (number of particles), and
a macroscopic count statement (number of moles)
From particles to moles (linking symbols to measurable amounts)
Mole: The amount of substance containing exactly elementary entities (such as molecules, atoms, ions, or formula units).
A balanced equation’s coefficients are simultaneously:
particle ratios (microscopic)
mole ratios (macroscopic)
The bridge between these interpretations is Avogadro’s constant.
= number of particles (entities)
= amount in moles (mol)
= Avogadro’s constant,
When translating symbols to quantities, be explicit about the entity:
For covalent substances: entities are usually molecules (e.g., CO₂ molecules).
For ionic solids: entities are formula units (e.g., NaCl units), not molecules.
For aqueous ionic compounds: entities are often ions (e.g., Na⁺(aq), Cl⁻(aq)) if the equation is ionic.
Consistency checks for a correct symbol-to-particle translation
Use these quick checks to ensure the particulate model matches the equation:
Ratio check: particle counts match the coefficient ratio.
Composition check: each particle’s internal atom counts match its formula.
Conservation check: total atoms of each element match on both sides after rearrangement.
Charge check (when ions are shown): total charge is the same before and after.
Common errors to avoid
Treating a subscript as if it changes the number of molecules (it changes atoms within one molecule).
Drawing diatomic elements incorrectly (e.g., showing O instead of O₂ when the formula is O₂).
Using coefficients to change atom counts inside a molecule (coefficients multiply whole particles, not subscripts).
Mixing scales (e.g., reactant picture shows “3 sets” while product picture shows “1 set”).
Forgetting that (aq) often implies separated ions in particle diagrams, not intact ionic “molecules.”
FAQ
Use the formula type: covalent compounds are discrete molecules; ionic solids are formula units. If the equation shows ions (especially with (aq)), draw ions.
Because a mole is a fixed number of entities, scaling particle counts by a factor scales mole amounts by the same factor; ratios stay identical.
Typically as separated, solvated ions or molecules dispersed in water. Keep ions apart and use the ionic charge shown in the symbol.
Ask: “Does this number change how many particles I have (coefficient) or how many atoms are inside one particle (subscript)?” Then re-check ratios.
They don’t change ratios, but they change what the particles look like (packed lattice for (s), widely spaced for (g), separated ions for many (aq) species).
Practice Questions
For the balanced equation , state what the coefficient 2 indicates for and for \text{H}_2\text{O in a particulate interpretation.
States 2 means two molecules (or particles) react (1)
States 2 means two molecules (or particles) are formed (1)
A student draws a particulate diagram for . Explain how the symbols in the equation determine (i) the identity of each particle and (ii) the relative numbers of each particle shown.
Identifies as one formula unit containing 1 Ca, 2 O, 2 H (1)
States and are ions as separate particles in solution (1)
Uses coefficient 2 to require twice as many ions as ions (1)
Links subscripts/parentheses to ion production: two hydroxide groups per formula unit become two (1)
Notes consistency requirement: atoms (and overall charge) are conserved in the diagram (1)

