AP Syllabus focus: ‘In rate‑limiting‑step mechanisms, the rate law follows the molecularity of the slow step (the rate‑determining step).’
Chemical reactions often occur through multiple elementary steps. When one step is much slower than the others, it controls how quickly products form. In that common situation, the slow step’s molecularity determines the observable rate law.
Core idea: the slow step sets the concentration dependence
In a multistep mechanism, each elementary step has its own microscopic event (a collision or unimolecular change). If one step is the bottleneck, changing reactant concentrations mainly affects the frequency of that slow event, so the overall rate mirrors that step’s reactant pattern.
Rate-determining step (RDS)
Rate-determining step (slow step): The elementary step in a mechanism that is slowest and therefore limits the overall reaction rate.
When the slow step is rate-determining, the experimentally determined rate law is taken directly from the reactants that appear in that slow elementary step, with exponents equal to their stoichiometric coefficients in that step.
Molecularity: how many particles collide in the slow step
“Molecularity” is only defined for an elementary step (never for an overall balanced equation), because it refers to a single microscopic event.
Molecularity: The number of reacting particles (molecules, atoms, or ions) that participate as reactants in a single elementary step.
Common molecularities (and what they imply for the slow step’s rate law form):
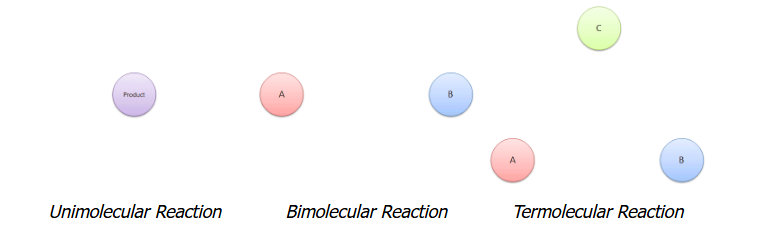
This set of diagrams illustrates unimolecular, bimolecular, and termolecular elementary steps as single microscopic events with one, two, or three reacting particles. The visuals support the rule that an elementary step’s molecularity corresponds to the total number of reactant particles involved in that step, which is why slow-step molecularity can map onto the exponents in the rate law for an elementary rate-determining step. Source
Unimolecular slow step: one reacting particle rearranges or dissociates
Bimolecular slow step: two particles collide (most common)
Termolecular slow step: three particles collide simultaneously (uncommon)
Writing the rate law from the slow elementary step
If a mechanism’s slow step is known, you translate that step’s reactant “collision stoichiometry” into the rate law.
Key rules (for a rate-limiting elementary step):
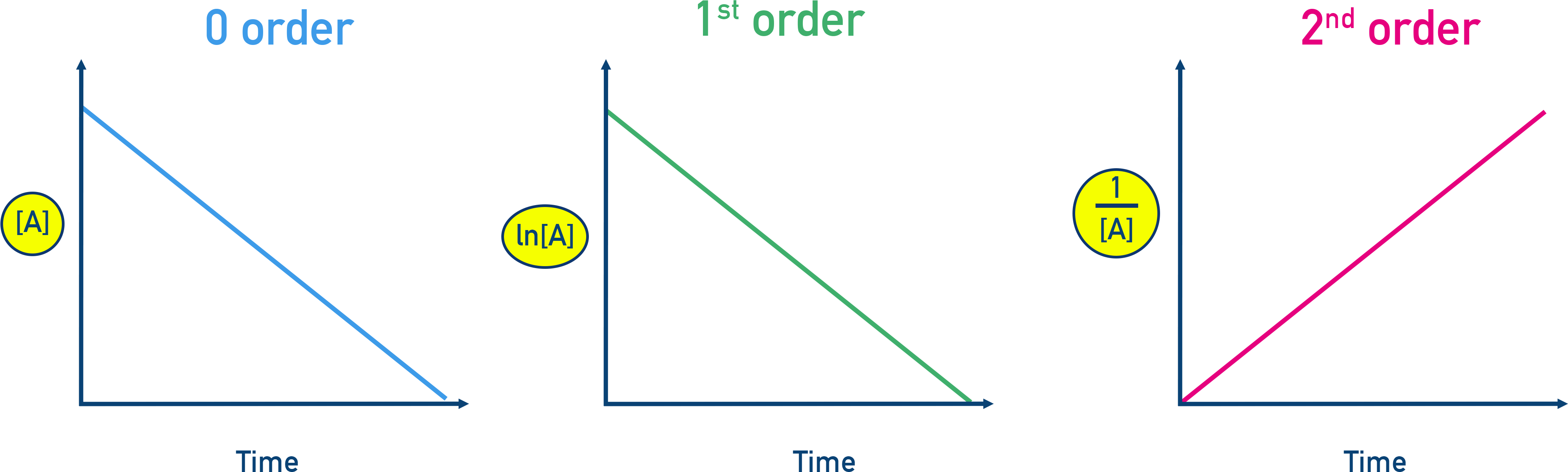
These concentration–time plots show the diagnostic linear forms used to identify reaction order from data: vs time (zero order), vs time (first order), and vs time (second order). The key idea is that the correct transformation produces a straight line, letting the slope relate to and the linearity confirm the order. Source
Use only reactants in the slow step (not products of that step).
The exponent on each concentration equals the coefficient of that species in the slow step.
The rate constant corresponds to that slow step at the given temperature.
= reaction rate (typically )
= rate constant for the rate-determining step (units depend on overall order)
= molar concentrations of reactant species in the slow step ()
= stoichiometric coefficients of and as reactants in the slow step (unitless)
This approach explains why many experimental rate laws have small integer orders: they often reflect a unimolecular or bimolecular rate-determining elementary event.
What this does (and does not) mean for overall stoichiometry
A frequent misconception is that the rate law should match the coefficients in the overall balanced equation. For multistep mechanisms, that is generally false.
What you can safely infer in the slow-step-controlled case:
The rate law matches the slow step, not the overall equation.
The overall order (sum of exponents) matches the slow step’s total molecularity (e.g., bimolecular slow step → second order overall), as long as the slow step contains only species whose concentrations track the initial reactants.
What you should not assume:
That the overall reaction must be elementary.
That changing any reactant in the overall equation must affect the rate; only species appearing in the slow step necessarily do.
Intermediates in the slow step: a practical warning
Sometimes a proposed slow step includes an intermediate as a reactant. Writing the slow-step rate law would then include that intermediate’s concentration, but intermediates are usually not directly measurable in typical rate experiments. In AP Chemistry, this is a sign you cannot stop at “read off the slow step” unless the intermediate concentration can be expressed in terms of measurable reactants (the algebraic elimination methods are handled elsewhere).
Operationally, for this subsubtopic, the take-home is:
Using molecularity of the slow step works cleanly when the slow step’s reactants are measurable reactants (or otherwise known concentrations).
If not, the mechanism may require additional reasoning beyond the molecularity read-off.
How experimental data connect to the slow-step molecularity idea
When kinetics data show:
Doubling a reactant concentration doubles the rate → consistent with first power in the slow-step-derived rate law
Doubling a reactant concentration quadruples the rate → consistent with second power (e.g., two of that species in the slow step)
These patterns support (but do not absolutely prove) a particular slow-step molecularity, because different mechanisms can sometimes produce similar overall rate laws. Still, on the AP exam, if the slow step is specified as the rate-determining elementary step, you are expected to write the rate law directly from its molecularity.
FAQ
Not always.
If the slow step’s reactants are the same as the measurable reactants, then yes, the overall order matches the slow step. If the slow step involves an intermediate, the observed overall order may differ once the intermediate is eliminated.
No.
Molecularity counts reacting particles in an elementary step. The smallest possible molecularity is unimolecular (1). “Zero order” is an experimental rate-law observation, not a molecularity.
A three-particle simultaneous collision is statistically unlikely.
Even if three species are needed overall, mechanisms typically proceed via consecutive bimolecular steps because two-body encounters are far more probable in solution or gas phases.
You generally cannot with certainty.
Without experimental kinetics (or additional information), “slow” versus “fast” is an assumption in the proposed mechanism. On AP questions, the slow step is usually stated explicitly.
It can.
A catalyst may introduce a new pathway where the rate-determining step involves different reactant particles (often including the catalyst). The slow step’s molecularity in the catalysed pathway then dictates the new rate law form.
Practice Questions
(2 marks) The slow (rate-determining) step of a mechanism is . Write the rate law in terms of and .
(1)
Correct first-order dependence on both and (or identifies bimolecular overall order) (1)
(5 marks) A proposed mechanism has a slow step (slow).
(a) State the molecularity of the slow step. (1)
(b) Write the rate law predicted from this slow step. (2)
(c) An experiment finds that doubling (holding constant) increases rate by a factor of 4. Explain how this supports the predicted rate law. (2)
(a) Termolecular (1)
(b) (2): correct form (1), correct exponents (1)
(c) Links factor of 4 to second order in : doubling gives rate increase (2): states relationship (1), clear explanation (1)

