AP Syllabus focus: ‘Freezing or condensing releases energy, decreasing the system’s energy while temperature stays constant during a phase change.’
Matter can change phase without changing temperature. In freezing and condensation, energy leaves the system as particles form stronger attractions, so the system’s energy decreases even though the thermometer stays steady.
What it means for energy to decrease at constant temperature
During a phase change, the average kinetic energy of particles (and therefore temperature) does not change. Instead, energy changes are tracked in the system’s potential energy associated with intermolecular attractions.
Phase change: A physical change in which a substance transitions between states (e.g., liquid to solid or gas to liquid) without changing chemical identity.
Because freezing and condensation release energy to the surroundings, they are exothermic physical processes from the system’s perspective.
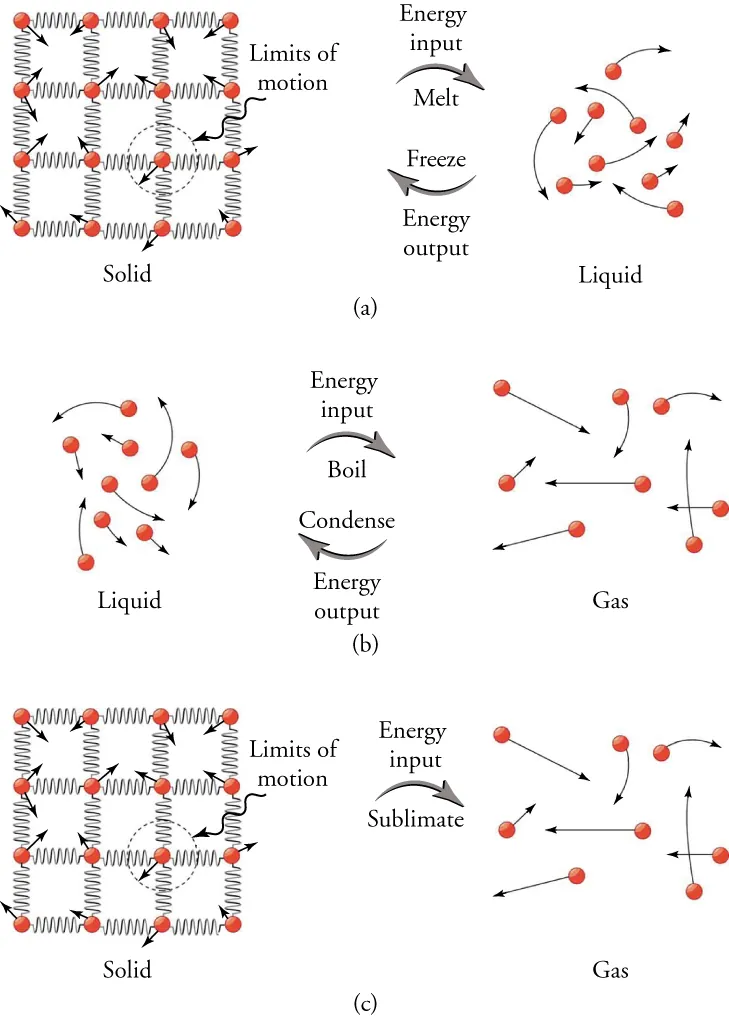
Particle-level diagram contrasting phase changes that require energy input (melting, evaporation) with those that require energy output (freezing, condensation). It emphasizes that when particles move closer and form stronger intermolecular attractions (condensation/freezing), energy must be transferred out of the system as heat. Source
Freezing (liquid → solid): energy is released
Particle-level explanation
In a liquid, particles are close together but still mobile. During freezing, particles arrange into a more ordered structure where attractions are, on average, more fully realized.
Freezing: The phase change in which a liquid becomes a solid as particles become locked into a more ordered arrangement.
Energy is released because:
Forming the solid structure increases the net strength/extent of intermolecular attractions.
The system’s potential energy decreases as particles settle into lower-energy positions relative to one another.
The released energy flows to the surroundings as heat.
Why temperature stays constant during freezing
At the freezing point (for a pure substance at a given pressure), added or removed heat does not change average particle speed. Instead, energy removal is “spent” on:
reducing particle freedom of motion,
building and stabilizing the solid’s intermolecular arrangement.
So the temperature remains constant until essentially all of the liquid has solidified. After freezing is complete, further energy removal lowers the temperature of the solid.
Condensation (gas → liquid): energy is released
Particle-level explanation
A gas has particles far apart with minimal intermolecular attraction. During condensation, particles come much closer and form many more attractions, producing a liquid.
Condensation: The phase change in which a gas becomes a liquid as particles come closer together and experience significantly stronger intermolecular attractions.
Energy is released because:
Bringing particles together increases attractive interactions and lowers potential energy.
The decrease in potential energy appears as heat transferred from the system to the surroundings.
The surroundings warm up (or warm up more than they otherwise would) as they absorb this released energy.
Why temperature stays constant during condensation
At the condensation point, removing heat primarily changes how closely particles associate rather than how fast they move. As long as gas and liquid coexist:
energy removal continues to convert gas to liquid,
average kinetic energy stays essentially constant,
the measured temperature remains constant.
Once all the gas has condensed, further cooling lowers the liquid’s temperature.
Energy flow language you must use correctly
System vs. surroundings
To stay consistent with AP thermochemistry wording:
System = the substance undergoing freezing or condensation.
Surroundings = everything else that can absorb the released heat (air, container, water bath).
For both freezing and condensation:
Heat flows from the system to the surroundings.
The system’s energy decreases during the phase change.
Temperature of the system stays constant during the phase change, even though energy is leaving.
Common experimental evidence
You typically infer energy release when:
the container or nearby solution warms (or cools less than expected),
a temperature-time graph shows a plateau (constant temperature) while the phase change proceeds.
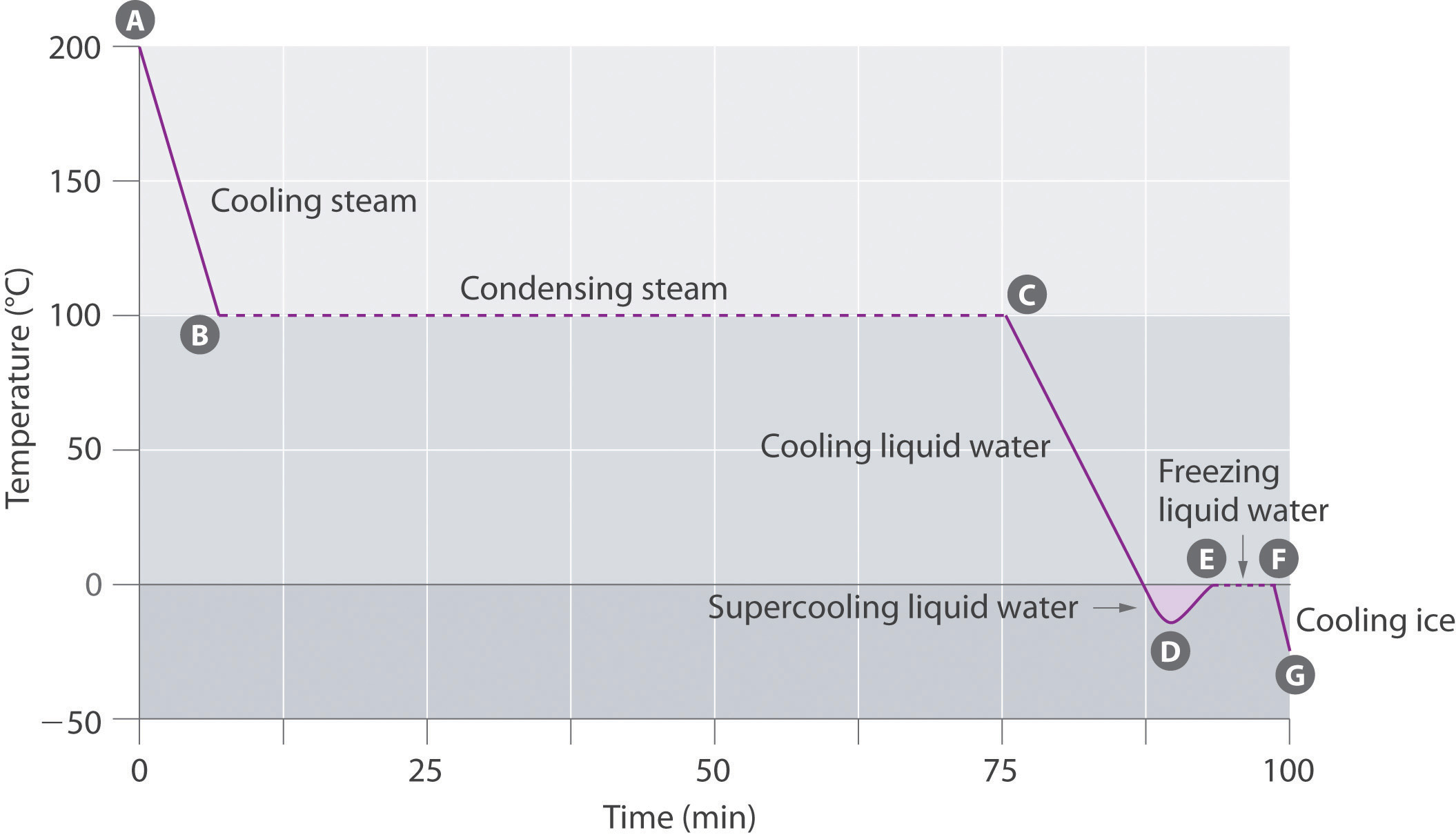
Cooling curve for water showing temperature vs. time as steam cools, condenses at 100 °C (plateau), then liquid water cools and eventually freezes near 0 °C (plateau). The horizontal regions indicate phase changes where energy is released but the temperature remains constant because the energy change is tied to intermolecular attractions rather than particle speed. Source
FAQ
Supercooling can occur when a liquid cools below its normal freezing point without forming a solid.
It happens when nucleation sites are absent; once crystallisation starts, latent energy release can drive the temperature back towards the freezing point.
Condensation is favoured where the surface cools nearby air below its dew point.
The surface also provides nucleation sites, making it easier for molecules to cluster into a liquid film.
Impurities typically disrupt crystal formation, lowering the freezing point and broadening the freezing process.
For condensation, impurities can act as nucleation sites, sometimes making droplets form more readily at a given humidity.
No new chemical bonds are formed; the substance’s identity is unchanged.
The energy change is associated with changes in intermolecular attractions (e.g., hydrogen bonding, dipole–dipole forces), not intramolecular covalent bonds.
This is deposition (gas to solid), which can occur when conditions favour immediate crystal formation.
Low temperatures and suitable surfaces can allow water molecules to lock into a solid lattice without first forming a liquid layer.
Practice Questions
(2 marks) When water vapour condenses to liquid water on a cold surface, state whether heat is absorbed or released by the system and whether the system’s energy increases or decreases.
Heat is released to the surroundings / released by the system (1)
System’s energy decreases (1)
(5 marks) Explain, in terms of particle motion and intermolecular forces, why the temperature of a pure substance remains constant while it freezes. Include where the energy goes and what energy change occurs within the system.
Temperature relates to average kinetic energy / particle speed (1)
During freezing, average kinetic energy stays constant, so temperature stays constant (1)
Particles form a more ordered arrangement and intermolecular attractions become stronger/more extensive (1)
Potential energy of the system decreases (1)
Energy is released from the system to the surroundings as heat during the phase change (1)

