AP Syllabus focus: ‘At equilibrium, no observable changes occur: both reactants and products are present, and the concentrations or partial pressures of all species remain constant over time.’
Chemical equilibrium is often identified by what you can measure directly in the lab. This page focuses on the macroscopic evidence that a system has reached equilibrium and what observations can be misleading.
What “macroscopic signs of equilibrium” means
Macroscopic observations involve bulk, measurable properties (like concentration, pressure, colour intensity, pH, or mass) rather than molecular-level events.
Macroscopic equilibrium: A state in which no observable changes occur because measurable properties (such as concentrations or partial pressures) remain constant over time, even though particles continue reacting.
A key idea from the syllabus is that at equilibrium:
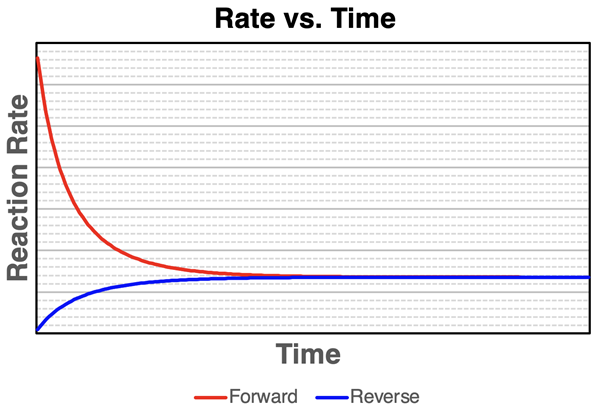
Rate–time curves for a reversible reaction showing the forward rate decreasing and the reverse rate increasing until they become equal. The point where the two rates match corresponds to equilibrium, meaning there is no net change in concentrations. This supports the interpretation that “no observable change” reflects balanced opposing processes, not the absence of reaction. Source
Both reactants and products are present
Concentrations (solutions) or partial pressures (gases) of all species remain constant over time
Observable signs you can use
Constant concentration or partial pressure over time
At equilibrium, measurements stop changing:
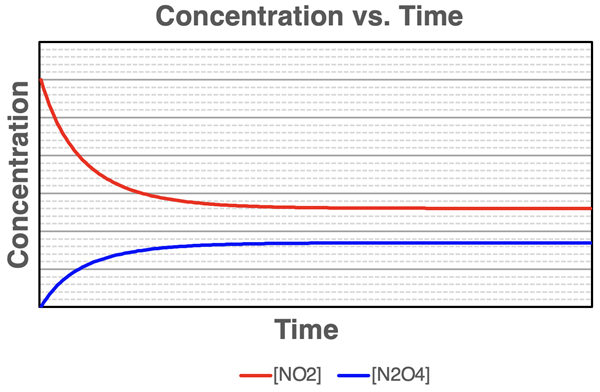
Concentration–time curves for a reversible reaction showing the reactant concentration decreasing and the product concentration increasing until both level off. The flat (time-independent) portions indicate equilibrium, where concentrations are constant even though the reaction continues microscopically. The different plateau heights reinforce that equilibrium does not require equal amounts of reactant and product. Source
In solution: repeated sampling shows constant molarities for reactants and products
In gases: a pressure sensor shows constant total pressure, and gas analysis shows constant partial pressures for each gas
Important nuance:
“Constant” means not changing with time, not “equal amounts.” Equilibrium mixtures commonly contain unequal amounts of reactants and products.
Both reactants and products are detectable
A macroscopic equilibrium mixture contains measurable amounts of:
Reactants that have not been fully consumed
Products that have formed and persist
In practice, detectability depends on sensitivity of the method (colour, conductivity, chromatography, spectroscopy), but the equilibrium idea is that neither side is absent.
No further observable changes in physical properties
Depending on the system, equilibrium can appear as steady:
Colour intensity (for coloured species)
pH (for acid–base systems)
Conductivity (if ion concentrations stabilise)
Mass in a sealed container (no net gas production escaping)
Level of precipitate or cloudiness (if dissolution/precipitation has balanced)
Conditions and common pitfalls
Equilibrium requires a closed system (for matter)
If products can escape (for example, gas leaving an open flask), the system may show continued change and may never establish a true equilibrium composition. Apparent “steady readings” can reflect experimental limitations rather than equilibrium.
“No visible change” does not always mean equilibrium
A system can look unchanged even when it is not at equilibrium:
The reaction may be very slow, so changes are too small to notice over short times
The relevant species may be colourless, giving no visual cue
Mixing delays or temperature gradients can mask changes
To claim equilibrium, observations should show constant measurements over time, not just a momentary lack of visible activity.
Phase equilibria can look static
Many equilibria involve phase changes (for example, evaporation/condensation) where liquid level or appearance may remain steady. The macroscopic sign is still the same: no net change in measurable amounts of each phase over time.
FAQ
It depends on temperature, concentrations/pressures, and the reaction mechanism.
Some systems equilibrate in milliseconds; others take hours. “Equilibrium” is about constancy over time, so the timescale must be compared to how frequently you measure.
Common tools include gas pressure sensors, pH meters, conductivity probes, and spectrophotometers.
For gases, constant partial pressures (not just total pressure) give stronger evidence of equilibrium.
Colour can track a specific species directly, whereas mass in a closed container is often constant regardless of reaction progress.
A constant colour over time is more directly tied to constant concentration of a coloured component.
Equilibrium does not mean completion.
It means the measurable amounts stop changing, so leftover reactant is expected unless the equilibrium lies extremely far toward products.
Sometimes you can observe a near-steady state, but true equilibrium for reactions involving gases or volatile components generally requires a closed system.
Open systems can lose matter, preventing constant composition from being maintained.
Practice Questions
Q1 (1–3 marks) A sealed container holds a reversible reaction that produces a brown gas. After some time, the brown colour intensity becomes constant. State two macroscopic observations that support the conclusion that the system is at equilibrium.
Colour intensity remains constant over time (1)
Both reactants and products are present/detectable in the container (1)
Concentrations or partial pressures of all species are constant over time (1)
Q2 (4–6 marks) In a sealed vessel for , measurements show that after 10 minutes the total pressure remains constant, and analysis shows constant partial pressures of and . Explain, using macroscopic criteria, why this indicates equilibrium, and give one reason why “constant total pressure” alone could be insufficient evidence.
States that at equilibrium there are no observable changes in composition (1)
Links equilibrium to constant partial pressures/concentrations of all species over time (1)
Notes both and are present at equilibrium (1)
Explains that constant total pressure alone may hide changing composition (e.g., decreasing while increasing with no net pressure change under some conditions/measurement limits) (1)
Mentions experimental limitation or need for time-series confirmation (1)

