AP Syllabus focus: ‘Increasing the concentrations of buffer components while keeping their ratio constant keeps pH the same but increases the buffer’s capacity to neutralize added acid or base.’
Buffers are designed to keep pH nearly constant during small acid/base disturbances. This page focuses on why pH depends on a ratio of buffer components, while buffer capacity depends on their absolute concentrations.
The key comparison: pH vs buffer capacity
pH is controlled by a concentration ratio
A typical buffer contains a weak acid and its conjugate base (or a weak base and its conjugate acid). For an acid buffer, the pH is set primarily by the ratio , not by the absolute size of each concentration.
Buffer capacity: the amount of strong acid or strong base a buffer can neutralise before its pH changes by a significant amount.
Keeping the ratio constant means both components are scaled up or down together (for example, doubling both and ).
= acidity of the solution (unitless)
= for (unitless)
= molar concentration of conjugate base (M)
= molar concentration of weak acid (M)
If and are both multiplied by the same factor, the fraction is unchanged, so the pH stays the same (assuming ideal behaviour).
Buffer capacity is controlled by total “inventory” of acid/base
Even if two buffers have the same ratio (and thus the same starting pH), they can behave very differently when challenged. Buffer capacity increases when the actual concentrations (and therefore moles present) increase, because there is more material available to react with added or .
Capacity is a stoichiometric idea: added strong acid or base is consumed mainly by the buffer components, and the pH shift occurs because their ratio changes.
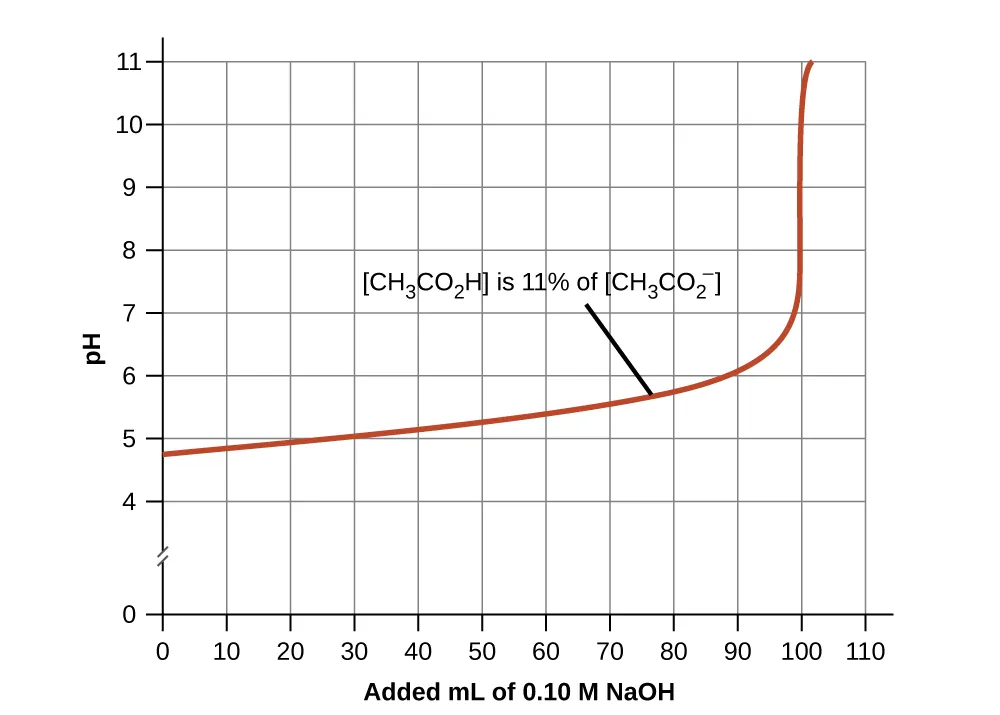
Titration curve for an acetic acid/acetate buffer during addition of strong base (NaOH). The gently sloping portion corresponds to the effective buffering region, where pH changes slowly because both and are present in appreciable amounts. The sharp rise indicates the buffer capacity has been exceeded and the pH becomes much more sensitive to added base. Source
Why increasing concentration increases capacity (at constant ratio)
What happens when strong acid is added
For an acid buffer , added is consumed mostly by the conjugate base:
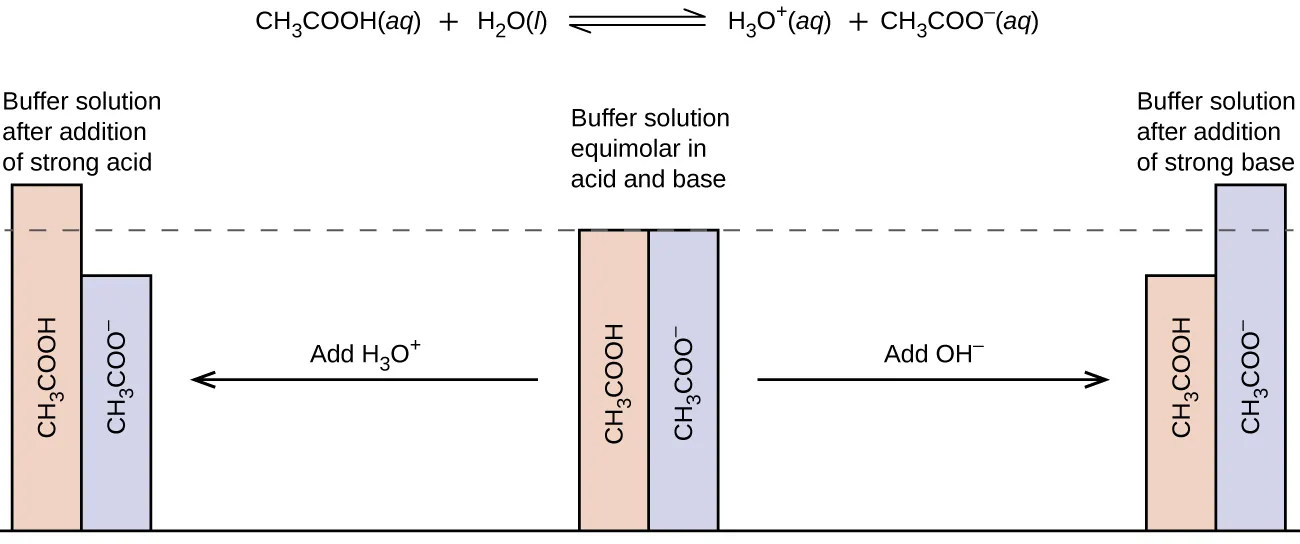
Schematic “before vs after” diagram for an acetic acid/acetate buffer showing how the conjugate-pair amounts shift when strong acid or strong base is added. Adding acid converts to , while adding base converts to . The key takeaway is that the buffer’s pH responds to how these changes alter the ratio , not just the presence of the components. Source
This decreases and increases
The ratio decreases, so pH falls
A more concentrated buffer has larger initial amounts of and , so the same added amount of produces a smaller fractional change in the ratio, causing a smaller pH change.
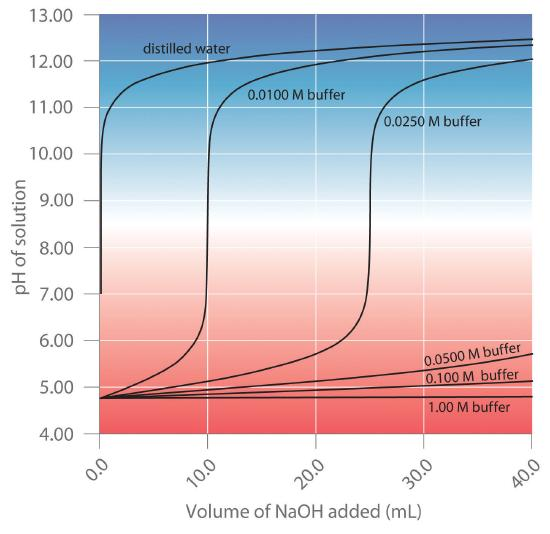
Comparison plot showing how pH responds to the same addition of strong base for buffers of different total concentrations. As concentration increases, the pH change per added increment becomes smaller, demonstrating greater buffer capacity even when the buffer ratio (and initial pH) are held constant. This is the graphical expression of “more inventory” of and resisting ratio change. Source
What happens when strong base is added
Added is consumed mostly by the weak acid:
This decreases and increases
The ratio increases, so pH rises
Again, when both components are more concentrated (but in the same ratio), the same added moles of change the ratio by a smaller percentage, so the pH is more stable.
What “constant ratio” really implies in practice
Same ratio → same pH (initially)
Two buffers with the same and the same will have essentially the same initial pH, even if one is 10× more concentrated.
Higher concentrations → greater resistance to pH change
At constant ratio, increasing concentration increases capacity because:
There are more moles of the conjugate pair available per litre
The neutralisation reactions can proceed further before the ratio is noticeably altered
The buffer can tolerate larger additions of strong acid/base before leaving its effective buffering region
Limits to the “same pH” claim
In AP Chemistry, the ratio model is the key idea. In real solutions, very high concentrations can introduce non-ideal effects (activities), but the syllabus-level takeaway remains: pH depends on ratio; capacity depends on concentration.
FAQ
Use a mole-based check: compare the moles of $HA$ and $A^-$ present to the moles of strong acid/base you plan to add.
A rough guideline: if added moles are a small fraction (for example, <5–10%) of the relevant buffer component, the pH change is typically small.
Ideally, no: dilution lowers both $[HA]$ and $[A^-]$ by the same factor, leaving $\frac{[A^-]}{[HA]}$ unchanged.
In very dilute buffers, water autoionisation and measurement limits can make deviations more noticeable.
Because pH depends on the ratio at a snapshot in time, but capacity is about how much reaction must occur before that ratio changes appreciably.
More concentration means more moles of each component to absorb added $H_3O^+$ or $OH^-$.
Capacity depends on total moles present, not just molarity.
Same molarity, larger volume $\rightarrow$ more total moles $\rightarrow$ higher total capacity
Same total moles, larger volume $\rightarrow$ lower molarity but similar total neutralisation ability
Non-ideal solution behaviour can matter: ion–ion interactions change effective concentrations (activities).
This can shift measured pH slightly even when analytical concentrations preserve $\frac{[A^-]}{[HA]}$.
Practice Questions
Q1 (2 marks) Two buffers have the same at C. Buffer 1 is M in each component; Buffer 2 is M in each component. Compare (i) their initial pH and (ii) their buffer capacity.
Initial pH is the same for both because is the same (1)
Buffer 2 has greater capacity because both components are more concentrated (more moles available to neutralise added acid/base) (1)
Q2 (5 marks) A student prepares two buffers using the same conjugate pair . Buffer X contains mol and mol . Buffer Y contains mol and mol . Each buffer is made up to L. Both are then challenged with mol of strong acid (). (a) State which buffer shows the smaller pH decrease. (1 mark) (b) Explain using the change in the ratio . (2 marks) (c) State whether the initial pH values of X and Y are the same or different, and why. (2 marks)
(a) Buffer Y (1)
(b) Adding converts to ; the ratio decreases (1)
(b) The same mol causes a smaller fractional (percentage) change in the ratio in Y because initial moles are larger (1)
(c) Same initial pH (1)
(c) Because both have the same (here, 1.0), so Henderson–Hasselbalch gives the same pH (1)

