AP Syllabus focus: ‘Changes in pH affect solubility qualitatively through Le Châtelier’s principle by shifting equilibria involving weak acid/base ions or OH−.’
Solubility is not always a fixed property: for many ionic solids, changing pH changes the concentrations of ions that participate in equilibrium. Using Le Châtelier’s principle, you can predict whether lowering or raising pH increases or decreases solubility.
Core idea: link solubility to equilibrium shifts
Le Châtelier’s principle in words
Le Châtelier’s principle: When a system at equilibrium is disturbed (e.g., by changing pH), the equilibrium shifts in the direction that reduces the disturbance.
For sparingly soluble salts, the dissolution equilibrium can shift if one of the ions is removed from solution by an acid–base reaction (or increased by it).
Removing a product drives more dissolution; increasing a product drives precipitation.
Solubility equilibrium language
= solubility product constant (unit depends on exponents)
= equilibrium molar concentration,
Even without calculating, the key prediction tool is: anything that lowers an ion concentration appearing in tends to increase solubility, and anything that raises it tends to decrease solubility.
When pH changes solubility: three common patterns
1) Salts containing a weak base anion (conjugate base of a weak acid)
These include anions like F⁻, CO₃²⁻, SO₃²⁻, S²⁻, PO₄³⁻, and many organic anions.
Lowering pH (adding H₃O⁺) protonates the anion:
This removes from solution (a “product” of dissolution).
By Le Châtelier, the solid dissolves more to replace , so solubility increases in acidic solution.
Raising pH does the opposite (less protonation), leaving more in solution, so dissolution is less favoured and solubility decreases (or precipitation becomes more likely).
2) Metal hydroxides and salts where is an ion
Many metal hydroxides are sparingly soluble:
Lowering pH consumes :
Removing shifts dissolution right, so hydroxides become more soluble in acid.
Raising pH directly increases (common ion effect), shifting left, so solubility decreases in base.
This is a prime case of the syllabus phrase “equilibria involving … OH−.”
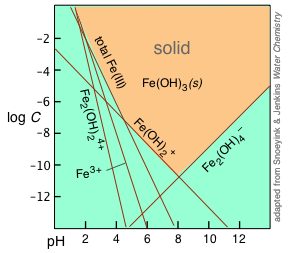
A log(concentration) vs pH speciation-style diagram for an iron(III)–hydroxide system, showing which aqueous hydroxo species dominate at different pH values. As pH changes, equilibria involving and redistribute dissolved forms, which can strongly affect whether a metal hydroxide tends to remain dissolved or precipitate. This connects the “pH consumes ” idea to a visual map of pH-dependent equilibria. Source
3) Salts containing a weak acid cation (conjugate acid of a weak base)
Some salts dissolve to give a cation that can donate a proton (often written as BH⁺), typical for ammonium or protonated amines.
Raising pH (adding base) deprotonates the cation:
This removes from solution, so dissolution shifts right and solubility increases in basic solution.
Lowering pH suppresses deprotonation (keeps more ), so solubility decreases relative to basic conditions.
A quick decision checklist (qualitative, no maths)
Identify whether pH can “consume” an ion from the solid
If an ion from the solid is a weak base, acid will consume it → acid increases solubility.
If the solid produces , acid consumes it → acid increases solubility and base decreases it.
If an ion from the solid is a weak acid (or behaves as BH⁺), base will consume it → base increases solubility.
Keep the equilibrium framing explicit
Use phrasing such as:
“Adding reduces , so the dissolution equilibrium shifts right.”
“Adding increases a product ion, so the equilibrium shifts left and precipitation is favoured.”
FAQ
Look for anions that are conjugate bases of weak acids (e.g., from HF, H$_2$CO$_3$, H$_3$PO$_4$). These readily protonate, so $[\text{anion}]$ is pH-dependent.
Cl$^-$ is the conjugate base of a strong acid (HCl), so it is negligibly basic and is not appreciably protonated; changing pH does not remove Cl$^-$ from solution.
Yes, if dissolution produces a weak-acid cation such as $\text{NH}_4^+$ (or another $\text{BH}^+$). Added $\text{OH}^-$ converts $\text{BH}^+$ to B, lowering $[\text{BH}^+]$ and pulling dissolution forward.
Explicitly state (1) which species concentration changes due to pH, (2) whether that species is a reactant or product in dissolution, and (3) the direction of the shift (right = dissolves, left = precipitates).
They can be protonated in steps (CO$_3^{2-}\rightarrow$ HCO$_3^-\rightarrow$ H$_2$CO$_3$), so lowering pH can remove the anion through multiple equilibria, often making the “acid increases solubility” effect especially strong.
Practice Questions
Q1 (3 marks) A student compares the solubility of in strongly acidic solution versus neutral water. Predict the effect of lowering pH on solubility and justify using Le Châtelier’s principle.
States solubility increases in acidic solution (1)
Correctly notes is protonated by to form (1)
Links removal of to equilibrium shifting right (more dissolution) via Le Châtelier (1)
Q2 (6 marks) A sparingly soluble salt is placed in (i) dilute nitric acid and (ii) sodium hydroxide solution. For each, predict whether solubility increases or decreases compared with pure water, and explain in terms of equilibria and Le Châtelier’s principle.
Writes/recognises dissolution: (1)
Acid case: states solubility increases (1)
Acid case: explains consumes to form water, lowering (1)
Acid case: links lowered product concentration to shift right (1)
Base case: states solubility decreases (1)
Base case: explains added increases a product/common ion, shifting left (precipitation favoured) (1)

