AP Syllabus focus: ‘Carboxylic acids are a common class of weak acids; strong bases (group I and II hydroxides) have very weak conjugate acids, while weak bases include ammonia, other nitrogen bases, and carboxylates.’
Recognizing common acids and bases from structure helps you quickly predict reaction direction, major species in solution, and relative strengths. AP Chemistry emphasizes a few high-utility structural “families” that appear repeatedly.
Common weak acids: carboxylic acids
Carboxylic acids contain the carboxyl group and are among the most common weak acids encountered in aqueous chemistry.
Carboxylic acid — an organic compound containing the –COOH functional group (often written R–CO2H) that can donate the –OH proton in acid–base reactions.
Key structural cues and examples:
Look for –COOH at the end of a carbon chain or attached to a ring (e.g., acetic acid, benzoic acid).
The acidic proton is on the O–H of the carboxyl group (not typically on nearby C–H bonds).
In water, carboxylic acids partially ionize, so most molecules remain as R–CO2H with a smaller amount of R–CO2− formed:
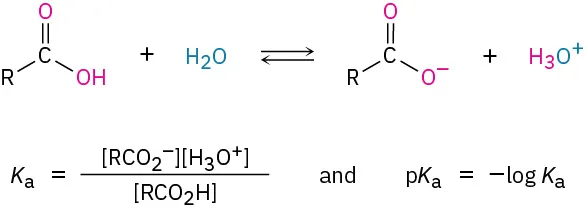
Reversible dissociation of a generic carboxylic acid in water, producing a carboxylate anion and hydronium. This visual reinforces that the reaction is an equilibrium (not complete ionization), consistent with carboxylic acids being weak acids in aqueous solution. Source
R–CO2H + H2O ⇌ R–CO2− + H3O+
How to use this on sight:
If a reactant is a carboxylic acid, expect it to behave as a proton donor toward bases (especially strong bases), but not to fully ionize like a strong acid would.
Common strong bases: Group I and Group II hydroxides
A major structural class of strong bases is metal hydroxides from Group I and (most) Group II.
Recognizing them:
Group I hydroxides: LiOH, NaOH, KOH, RbOH, CsOH
Group II hydroxides: commonly treated as strong in AP contexts (e.g., Ca(OH)2, Sr(OH)2, Ba(OH)2)
What “strong base” implies structurally in water:
These are ionic solids that dissociate essentially completely in aqueous solution to release OH−.
Their conjugate acids are the corresponding hydrated metal cations (e.g., Na+(aq)), which are very weak acids and do not meaningfully neutralize OH−. This is why the hydroxide behaves “fully basic” in water.
Quick identification tips:
If you see a formula that is “metal + OH,” especially an alkali metal, treat it as a strong base source.
For Group II, remember each formula unit can supply two OH− (e.g., M(OH)2), even when solubility is limited.
Common weak bases: ammonia and other nitrogen bases
Many weak bases in introductory acid–base chemistry are nitrogen-containing compounds that can accept a proton.
Typical examples and structural cue:
Ammonia (NH3) and amines (nitrogen with a lone pair, such as R–NH2, R2NH, R3N)
“Other nitrogen bases” can include ring nitrogens (e.g., pyridine-like structures) where a lone pair is available to bond to H+.
Characteristic behavior in water:
They partially react with water to form their conjugate acid and OH−:
B + H2O ⇌ BH+ + OH−
Because ionization is incomplete, these solutions are basic but typically not as basic as comparable concentrations of strong hydroxides.
Recognition mindset:
If nitrogen has an available lone pair (and is not locked into a strongly electron-withdrawing environment), it is a strong candidate for a weak base.
Carboxylates as weak bases (conjugate bases of carboxylic acids)
When a carboxylic acid loses its proton, it forms a carboxylate, which can act as a weak base.
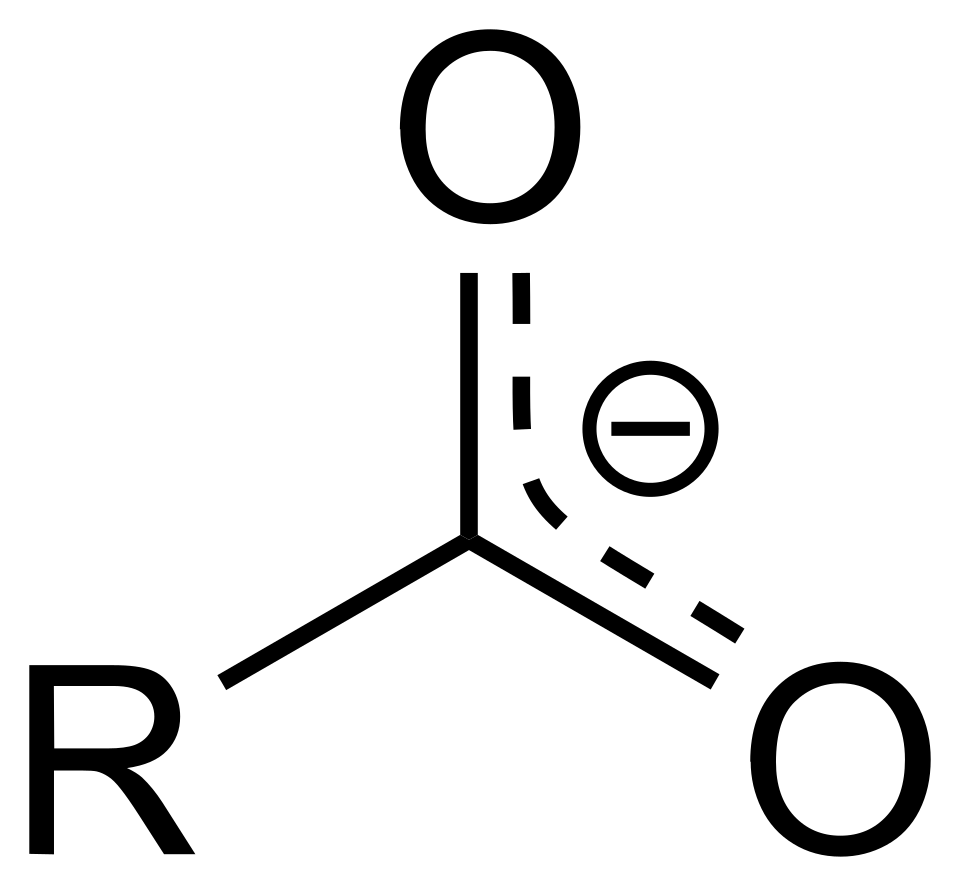
Resonance-hybrid depiction of a carboxylate ion, showing the negative charge delocalized over both oxygen atoms and the two C–O bonds having partial double-bond character. This resonance stabilization is why carboxylate conjugate bases are relatively weak bases compared with bases that localize charge on a single atom. Source
Carboxylate — the conjugate base of a carboxylic acid, with the general formula R–CO2− (often present as an ionic salt such as sodium acetate, Na+ R–CO2−).
Practical structural examples:
Acetate: CH3CO2−
Benzoate: C6H5CO2−
What to expect in water:
Carboxylates can accept a proton from water to a small extent, producing some OH−:
R–CO2− + H2O ⇌ R–CO2H + OH−
Because they are weak bases, salts containing carboxylates can make solutions slightly basic.
Fast classification checklist (structure → behavior)
Contains –COOH: classify as a weak acid (carboxylic acid family).
Metal hydroxide with Group I or II metal: classify as a strong base source (releases OH− readily).
Nitrogen with an available lone pair (ammonia/amine-like): classify as a weak base.
R–CO2− present (often as a salt): classify as a weak base (carboxylate family).
FAQ
Carboxylic acids form carboxylate ions whose negative charge is spread over two oxygens, whereas alkoxides localise charge mainly on one oxygen.
This difference makes deprotonation of –COOH far more favourable than deprotonation of a typical alcohol.
Check whether the lone pair is part of an aromatic sextet or strongly delocalised into a nearby carbonyl.
If it is delocalised, basicity drops; if it is localised and accessible, the nitrogen is more likely to behave as a base.
Their hydroxide is strong when dissolved, but some have limited solubility.
So they can be “strong bases” chemically yet produce less OH− than expected if only a small amount dissolves.
Carboxylates are stabilised anions, so they have less driving force to grab a proton from water compared with OH−.
As a result, they generate only a modest amount of OH− in aqueous solution.
Such molecules can undergo internal acid–base behaviour, often existing largely in a zwitterionic form.
Which form dominates depends on the relative tendencies of the groups to gain/lose H+ and the solution conditions.
Practice Questions
(2 marks) State whether each species is most likely a weak acid, weak base, or strong base in water: (i) CH3CO2H, (ii) NH3, (iii) Ba(OH)2.
(i) weak acid (1) (ii) weak base (1) (iii) strong base (1) [max 2; award any two correct]
(5 marks) A student mixes aqueous sodium acetate (NaCH3CO2) with water.
(a) Identify the Brønsted–Lowry base present. (1)
(b) Write an equation showing how this base reacts with water. (2)
(c) State whether the resulting solution is acidic, basic, or neutral, and justify using the species formed. (2)
(a) CH3CO2− (1)
(b) CH3CO2− + H2O ⇌ CH3CO2H + OH− (2: correct reactants/products and correct charges/equilibrium)
(c) Basic; formation of OH− (or increased [OH−]) (2: “basic” + justification tied to OH− production)

