AP Syllabus focus: ‘Relate the number of electrons transferred to amounts of reactants and products using balanced redox stoichiometry.’
Electrolysis problems are stoichiometry problems in which electrons serve as the counting bridge between electrical charge and chemical change. Mastery comes from writing correct half-reactions, then using coefficients to convert between mol and mol of substance produced or consumed.
The central idea: electrons as the “limiting reagent”
In electrolysis, oxidation and reduction occur simultaneously at different electrodes.
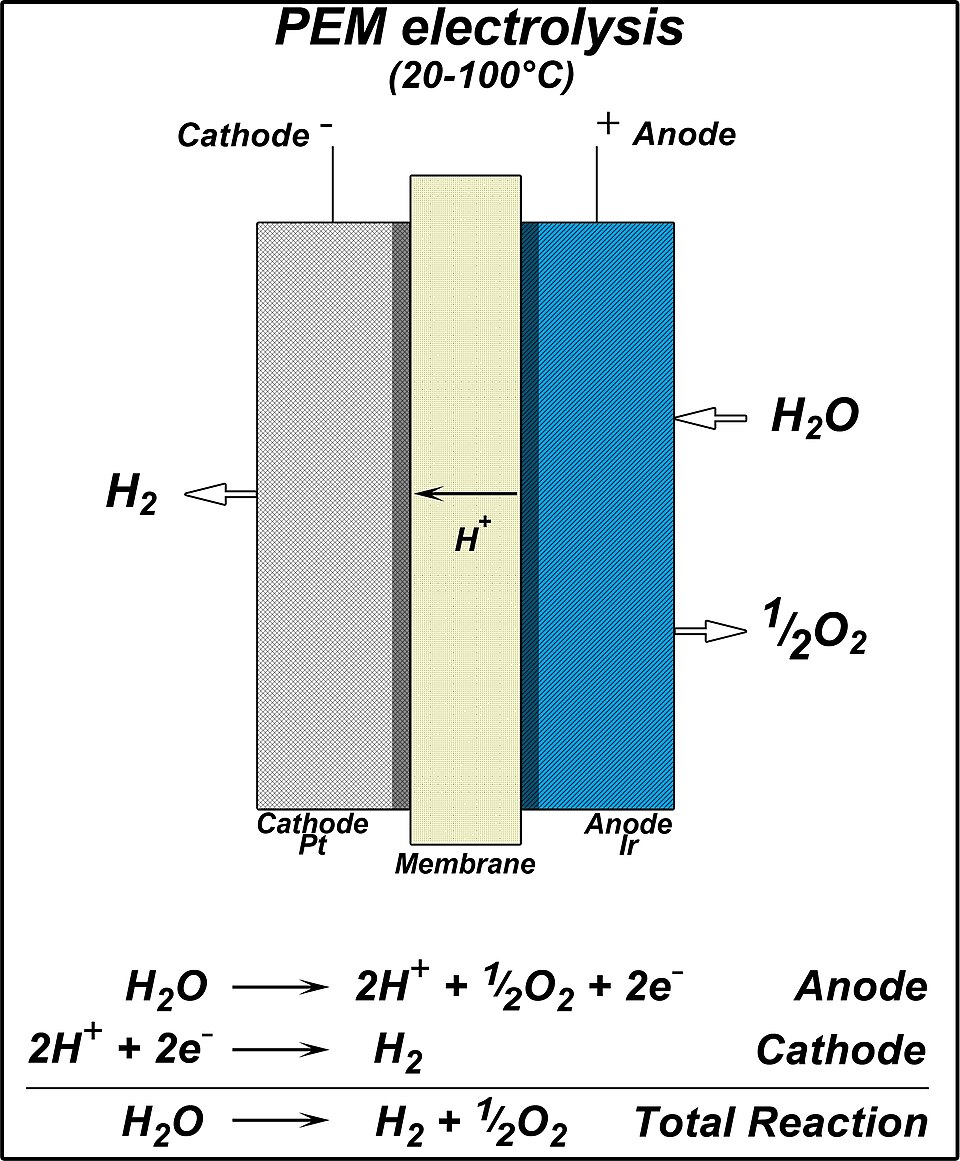
Diagram of a proton-exchange-membrane (PEM) electrolyzer illustrating separated anode and cathode half-reactions with charge transport through the circuit and membrane. The visual makes it explicit that electrons produced at the anode are consumed at the cathode, which is the physical basis for consistent electron accounting in redox stoichiometry. Source
The balanced half-reactions explicitly show how many electrons are transferred per mole of a species, and those coefficients control all mole relationships.
Faraday constant (): The charge carried by 1 mole of electrons, approximately .
Even if a problem starts with current and time, the chemistry step is always: convert to moles of electrons, then use the balanced redox stoichiometry to relate electrons to chemical amounts.
Converting between electrons and chemical amounts
The key quantitative link is the mole ratio embedded in the half-reaction coefficients.
= moles of electrons transferred, mol
= total charge passed, C
= Faraday constant,
Once is known (or given), treat like a reactant/product in a balanced equation: coefficients give mol-to-mol conversion factors.
Building the correct stoichiometric map
1) Identify what is being oxidised and reduced
Electrolysis always includes:
A reduction half-reaction (electrons on the reactant side)
An oxidation half-reaction (electrons on the product side)
The question may specify electrode products (e.g., “metal plates,” “gas evolves”), or it may provide the half-reactions directly. Your stoichiometry must match the stated chemical change.
2) Use a balanced half-reaction to link to the target species
For any half-reaction of the form
(reduction), or
(oxidation)
the coefficient means:
mol are transferred per 1 mol of the species paired with that electron coefficient (as written)
This lets you form conversions like:
for reduction products
for oxidation reactants
3) Decide whether to use a single half-reaction or the combined redox equation
Use a single half-reaction when the question asks about material formed at one electrode (common in electrolysis). Use the overall balanced redox only when the question asks for net chemical change in the cell.
Important constraint: when combining half-reactions, you may multiply entire half-reactions to cancel electrons, but do not change subscripts, phases, or charges.
Stoichiometry patterns that appear repeatedly
Metals plating (cation reduction)
For a metal ion reduced to metal(s), the electron coefficient equals the ionic charge:
So, mol correspond to 1 mol metal deposited. If electrons are known, the deposited moles are proportional to .
Halogen formation (halide oxidation)
Halide oxidation typically produces diatomic halogen:
So, 2 mol correspond to 1 mol produced, and also to 2 mol consumed.
Water oxidation/reduction (common in aqueous electrolysis)
Water-based half-reactions often involve multiple electrons, so coefficients matter greatly:
Hydrogen formation often uses 2 per
Oxygen formation often uses 4 per
For stoichiometry, you do not need the mechanism—only the balanced electron count in the half-reaction provided or implied.
Keeping track of “amounts”: what can (and cannot) be inferred
Moles scale with electrons: doubling doubles the theoretical moles of products, assuming 100% current efficiency.
Charge conservation across electrodes: the same total moles of electrons that leave the anode must arrive at the cathode, so the electron bookkeeping must match between half-reactions.
Do not add electrons across electrodes unless forming the net reaction: electrons are an accounting tool; they cancel only when oxidation and reduction are combined properly.
High-frequency errors to avoid
Forgetting to multiply by the electron coefficient (e.g., assuming 1 mol makes 1 mol product)
Using the wrong half-reaction (e.g., applying a cathode ratio to an anode product)
Canceling electrons without multiplying an entire half-reaction consistently
Mixing “moles of electrons” with “charge” without using (units must guide each conversion step)
FAQ
Ignore spectators for the mole link unless the question asks about them.
Use only the electron coefficient-to-target-species coefficient ratio. For example, if $O_2$ is produced with $4e^-$, then $n(O_2)=n(e^-)/4$ regardless of how many $H^+$ also appear.
It refers to the same pool of electrons moving through the external circuit.
You may calculate moles at either electrode from the same $n(e^-)$, but you must use the correct half-equation ratio for that electrode’s product.
Only when asked for the net chemical change (overall reactants and products).
If the task is “amount deposited at the cathode” or “amount of gas at the anode,” using the single relevant half-equation is usually cleaner and avoids algebraic cancellation errors.
Avoid them by multiplying the entire half-equation by an integer first.
Stoichiometric mole ratios come from coefficients; keeping them whole-numbered reduces mistakes when converting between $n(e^-)$ and moles of substance.
The main hidden assumption is 100% current efficiency (all electrons drive the intended half-reaction).
If side reactions occur, the actual amount produced will be lower; some problems may state a percentage efficiency, in which case multiply theoretical moles by the efficiency as a decimal.
Practice Questions
(2 marks) Aluminium is produced by electrolysis. The cathode half-equation is . If mol of electrons pass through the circuit, how many moles of can form?
Uses the ratio (1 mark)
Calculates mol (1 mark)
(5 marks) An electrolytic cell produces copper metal at the cathode and oxygen gas at the anode. The half-equations are: Cathode:
Anode:
If mol of electrons are transferred, determine: a) the moles of deposited (2 marks)
b) the moles of produced (2 marks)
c) the moles of consumed (1 mark)
(a) Uses (1 mark); mol (1 mark)
(b) Uses (1 mark); mol (1 mark)
(c) Recognises consumed equals produced in a 1:1 mole ratio from the cathode half-equation, so mol (1 mark)

