AP Syllabus focus: ‘Some processes require considering both ΔH° and ΔS° to judge favorability; examples include freezing water and dissolving sodium nitrate.’
Predicting whether a process is thermodynamically favoured often requires weighing two competing tendencies: lowering energy and increasing dispersal. When enthalpy and entropy “agree,” predictions are easy; when they compete, temperature becomes crucial.
Why both ΔH° and ΔS° matter
A process can be pulled in two different directions:
Enthalpy change, ΔH° reflects energy released or absorbed as bonds and intermolecular attractions change.
Entropy change, ΔS° reflects how dispersed matter and energy become among available microstates.
Favourability depends on the combined effect, not on ΔH° or ΔS° alone.
Gibbs free energy change (ΔG°): a state function that combines enthalpy and entropy to indicate thermodynamic favourability under standard conditions; ΔG° < 0 corresponds to a thermodynamically favoured process.
Even if a process is exothermic (ΔH° < 0), it may be opposed by a decrease in entropy (ΔS° < 0). Likewise, an endothermic process (ΔH° > 0) can still be favoured if it produces a sufficiently large entropy increase (ΔS° > 0).
The relationship that forces you to consider both
The reason both terms must be considered is that entropy is “weighted” by temperature.
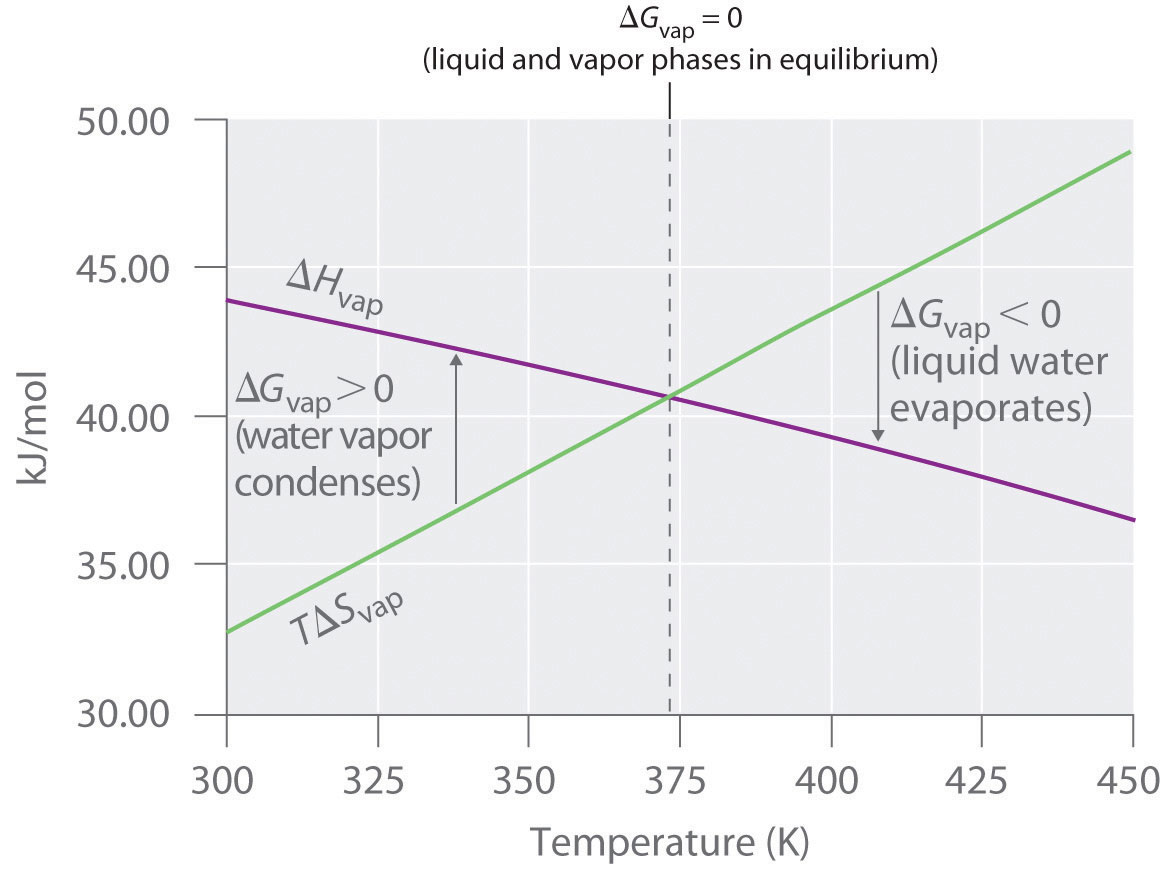
This plot compares and as temperature changes, highlighting the temperature where and therefore . It makes the “temperature weighting” idea concrete: as increases, the contribution grows in magnitude and can overtake the enthalpy term, changing which direction is thermodynamically favored. Source
= Gibbs free energy change, kJ·mol
= enthalpy change, kJ·mol
= temperature, K
= entropy change, kJ·mol·K
A key interpretation for this subtopic is qualitative:
When ΔH° and ΔS° have the same sign, they reinforce each other (favoured or unfavoured across temperatures).
When ΔH° and ΔS° have opposite signs, they compete, so you cannot decide favourability without considering temperature.
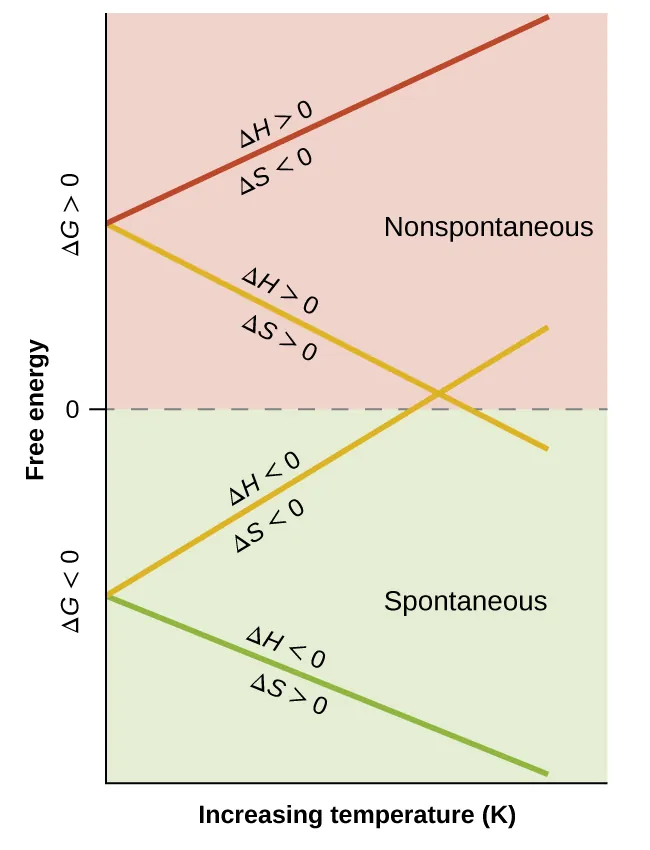
This graph shows how the sign of depends on temperature for the four possible sign combinations of and . The two “competing-sign” cases cross at a characteristic temperature, illustrating why temperature can flip a process from nonspontaneous to spontaneous (or vice versa). Source
Example 1: Freezing water (why “cold” matters)
Freezing liquid water to solid ice typically has:
ΔH° < 0: heat is released as stronger hydrogen-bonding patterns form in the solid.
ΔS° < 0: the system becomes more ordered as molecules occupy more fixed positions.
Because both terms are negative, the two contributions oppose each other in :
The enthalpy term (ΔH° < 0) favours freezing.
The entropy term (−TΔS° becomes positive when ΔS° < 0) opposes freezing, and its magnitude grows with T.
So, freezing is favoured only when temperature is low enough that the enthalpy benefit outweighs the entropy penalty; at higher temperatures, melting is favoured instead.
What students should be able to say from signs alone
Freezing is not “guaranteed” by being exothermic.
The entropy decrease makes freezing increasingly unfavourable as temperature increases.
Example 2: Dissolving sodium nitrate (why “endothermic” can still happen)
Dissolving NaNO₃(s) in water is commonly observed to cool the solution, indicating:
ΔH° > 0 overall (net absorption of heat), often because separating ions and reorganising water can outweigh ion–dipole attractions formed.
ΔS° > 0 overall (more dispersal): ions spread through the solvent, increasing the number of possible arrangements.
Here, the signs are opposite:
The enthalpy term (ΔH° > 0) opposes dissolution.
The entropy term (−TΔS° is negative when ΔS° > 0) favours dissolution, increasingly so at higher T.
Thus, dissolution can be thermodynamically favoured even when it is endothermic, provided the entropy increase (especially when multiplied by temperature) is large enough.
What students should be able to say from observations
Cooling during dissolution signals ΔH° > 0, but does not by itself mean the process is unfavoured.
The driving force can be an entropy increase that dominates in .
FAQ
Two effects compete: dispersal of solute into many positions increases entropy, while hydration shells can decrease entropy locally. The net ΔS° depends on which dominates for that solute and conditions.
If energy required to separate ions in the lattice plus solvent reorganisation exceeds energy released from ion–dipole interactions, the overall ΔH° becomes positive.
Ice forms a more constrained hydrogen-bond network with fewer accessible microstates for water molecules, which corresponds to a decrease in entropy.
No. “Standard” refers to standard states (e.g., 1 bar, 1 M, pure solids/liquids). Temperature can be specified separately; ΔH° and ΔS° are temperature-dependent in principle.
Heat capacities change with phase and temperature, so ΔH° and ΔS° can shift. Over wide ranges, this can move the balance point where the entropy and enthalpy terms swap dominance.
Practice Questions
Question 1 (3 marks) For each process, state the signs of ΔH° and ΔS°, and explain briefly why both must be considered to judge thermodynamic favourability: (a) Freezing water at constant pressure. (b) Dissolving sodium nitrate in water (solution cools).
ΔH° < 0 (1)
ΔS° < 0 (1)
Must consider both because entropy decrease opposes freezing and the term depends on temperature (1)
(b)
ΔH° > 0 (1)
ΔS° > 0 (1)
Must consider both because endothermicity opposes dissolution but entropy increase can drive it (especially as increases) (1)
Question 2 (6 marks) A student claims: “If a process is endothermic, it cannot be thermodynamically favoured.” Use dissolving NaNO₃ and freezing water to evaluate this claim using ΔH°, ΔS°, and the expression . Your answer should include how temperature affects the competition between terms.
Correctly states claim is false (1)
For NaNO₃ dissolution: identifies ΔH° > 0 and ΔS° > 0 (1)
Explains that is negative and can outweigh positive ΔH°, making ΔG negative (1)
Notes higher temperature increases the magnitude of the entropy contribution for NaNO₃ (1)
For freezing: identifies ΔH° < 0 and ΔS° < 0 (1)
Explains that becomes positive and can oppose freezing more at higher temperature, so freezing is favoured only at sufficiently low temperature (1)

