AP Syllabus focus: ‘Many processes that are thermodynamically favored occur only to a small extent or proceed at extremely slow rates.’
Thermodynamics tells you whether a change is energetically “allowed,” but it does not set the pace. Many reactions have a driving force yet show little observable change because the pathway is difficult or the driving force is small.
Thermodynamic favorability is not a rate prediction
A process can be thermodynamically favored and still appear to “not happen” on a human timescale. Thermodynamics addresses the initial-to-final energy comparison, while kinetics addresses the pathway taken to get there.
Thermodynamically favored (spontaneous): A process that has a negative Gibbs free energy change under the stated conditions, so the products are lower in free energy than the reactants.
Key idea: the sign and magnitude of describe the direction of the driving force, not how quickly particles reorganize to reach the lower-free-energy state.
Thermodynamics asks: “Is the destination lower in free energy?”
Kinetics asks: “How hard is it to get there?”
Why a favored process can proceed extremely slowly
The reaction pathway can involve a large barrier
Even when products are lower in free energy, reactant particles typically must pass through a high-energy arrangement as bonds stretch and break and new bonds form.
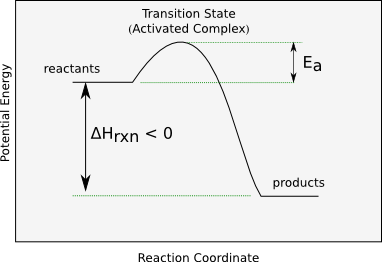
Reaction-coordinate (potential energy) diagram for an exothermic reaction, highlighting the transition state (activated complex) and the activation energy that must be overcome. The products lie lower than the reactants (thermodynamically “downhill”), yet the barrier explains why the process can still be slow. Source
Activation energy: The minimum energy barrier that must be overcome for reactants to reach the transition state and form products.
A large activation energy means:
Only a small fraction of collisions have enough energy at a given temperature.
Correct collision orientation may be rare, especially for complex molecules.
The observed rate can be so low that essentially no macroscopic change is seen.
This is why “favored” does not imply “fast”: the barrier governs how frequently successful events occur, even if the overall change lowers free energy.
Physical constraints can make the “chemistry” hard to access
Many favored processes require particles to move, mix, or reorganize before reaction events can occur.
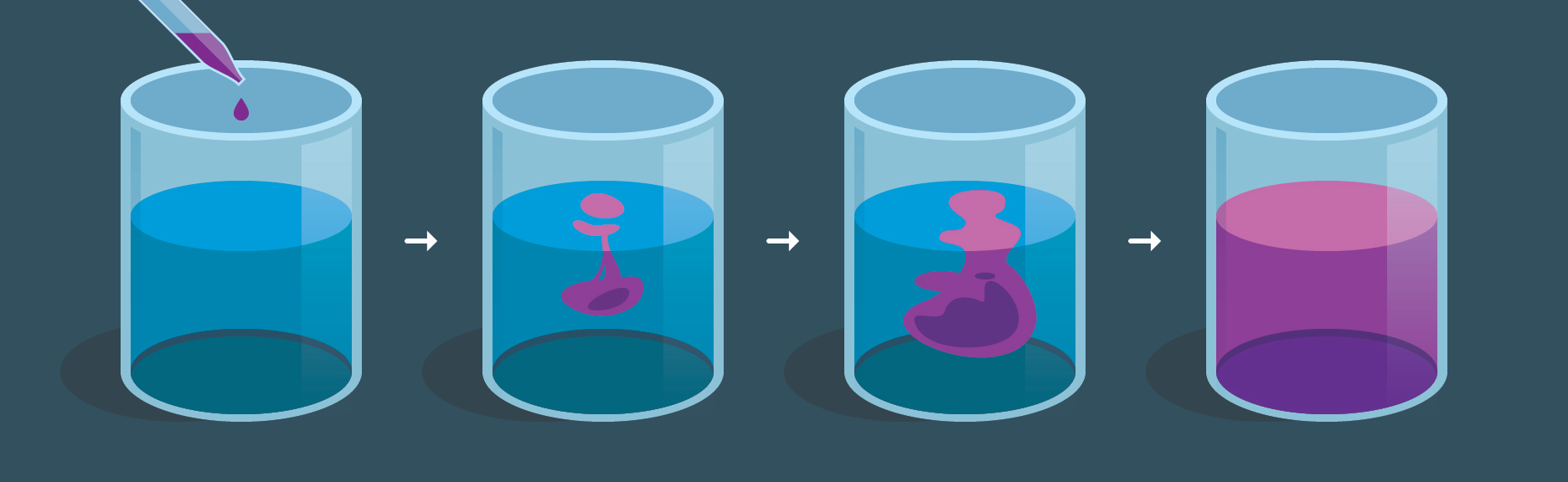
Sequence illustrating diffusion of a dye drop in water: initially localized high concentration spreads until the solution becomes uniform. This provides a concrete picture of diffusion-controlled mixing, where the rate of molecular transport (not thermodynamic driving force) can be the limiting step for how fast changes become observable. Source
These steps can be slow even if the underlying chemical change is downhill in free energy.
Common rate-limiting physical factors include:
Diffusion limits (particles must migrate through solution, solids, or membranes)
Phase boundaries (reactions between solids and gases or between immiscible liquids occur only at interfaces)
Low surface area of a solid reactant (fewer reactive sites exposed)
Viscosity or restricted motion (slower transport, fewer effective collisions)
Some favored processes require nucleation or restructuring
Certain changes (especially involving solids, crystallization, precipitation, or bubble formation) may require formation of an initial “seed” of the new phase. Even if the final state is favored, the early stages can be slow because forming the first stable clusters is difficult.
Nucleation can be the slow step before rapid growth becomes possible.
A system may persist in a metastable state until disturbed (shaking, scratching glass, adding a seed crystal).
Why a favored process can occur only to a small extent
The syllabus also highlights that favored processes may proceed “only to a small extent.” This happens when the driving force is small or when forward progress is increasingly opposed as products accumulate.
Qualitatively, limited extent is more likely when:
The decrease in free energy from reactants to products is slight, so there is not a strong push toward products.
The system can readily undergo the reverse change, leading to a mixture where neither side dominates strongly.
Conditions restrict progress (closed containers, limited contact between reactants, or competing processes consuming reactants).
In practice, you can observe a reaction that is thermodynamically allowed but shows:
only partial conversion,
small measurable changes,
or a final mixture that still contains significant reactants.
What you can and cannot infer from observations
Observable slowness is evidence about kinetics, not thermodynamic direction.
If little change is seen, you cannot conclude the reaction is unfavorable; it may be thermodynamically favored but slow.
Increasing temperature, increasing surface area, improving mixing, or adding a catalyst can increase rate without changing whether the overall process is thermodynamically favored.
A catalyst changes the pathway (lower barrier), not the relative free energies of reactants and products, so it does not “make” an unfavorable process favorable; it only helps a favorable process occur measurably (or helps an unfavorable one reach its nonfavored equilibrium faster).
FAQ
“Spontaneous” refers to thermodynamic direction, not a guarantee of observable progress.
A reaction can be downhill in free energy but trapped behind a large barrier, so the timescale for noticeable change may be hours, years, or longer.
A metastable system sits in a local, temporary minimum of free energy.
It can persist until a disturbance supplies a route over a barrier (for example, a scratch, a seed crystal, or a shock), after which change can proceed rapidly.
Early stages may be limited by nucleation or by creating reactive surface area.
Once a new phase forms or more interface becomes available, the rate can increase sharply even though the overall thermodynamic driving force is unchanged.
No. $\Delta G$ describes how far downhill the reaction is overall.
Rate is controlled mainly by the height of the barrier along the pathway; a reaction can be strongly downhill but still slow if the barrier is large.
You can change conditions that affect rate without changing the basic chemistry, such as:
increasing surface area (powdering a solid)
improving contact between phases (stirring, bubbling)
modest heating to increase collision energy
If the reaction then proceeds measurably, the original lack of change was consistent with kinetic limitation rather than thermodynamic impossibility.
Practice Questions
In one sentence, explain why a thermodynamically favoured reaction may show no observable change at room temperature.
States that thermodynamics predicts feasibility (driving force), not speed (1)
States that rate depends on the pathway/energy barrier (activation energy) (1)
Links large barrier to very slow rate / negligible observable change on the timescale (1)
A reaction has under the conditions used, but after stirring for several minutes there is little visible change.
(a) Give two kinetic or physical reasons (not thermodynamic ones) that could account for the lack of observable change.
(b) Explain, in terms of the reaction pathway, what effect adding a catalyst would have on the speed of the reaction and why this does not change the sign of .
(a) Any two valid reasons, 1 mark each (2 max):
large activation energy / high barrier
poor mixing or slow diffusion
small surface area / limited interface between phases
orientation/steric requirements make effective collisions rare
nucleation required before rapid change occurs
(b) Catalyst provides an alternative pathway with a lower activation energy (1)
Lower barrier increases the rate by increasing the fraction of successful events per unit time (1)
depends on initial and final states, so changing the pathway does not change the sign of (1)
States catalyst does not alter relative free energies of reactants and products (1)

