AP Syllabus focus: ‘Thermodynamically favored (ΔG° < 0) means products are favored at equilibrium, so K > 1 under standard conditions.’
This topic links thermodynamics to equilibrium outcomes. By interpreting the sign of ΔG° under standard conditions, you can predict whether equilibrium lies toward products or reactants using the equilibrium constant, K.
Core idea: ΔG° predicts where equilibrium lies
What ΔG° communicates under standard conditions
ΔG° (standard Gibbs free energy change): the change in Gibbs free energy for a reaction when all species are in their standard conditions, indicating thermodynamic favorability for the standard-state process.
A reaction with ΔG° < 0 is thermodynamically favored under standard conditions, meaning the system has a tendency (at equilibrium) to contain relatively more products than reactants. This is not a statement about reaction speed; it is a statement about the equilibrium outcome that is favored.
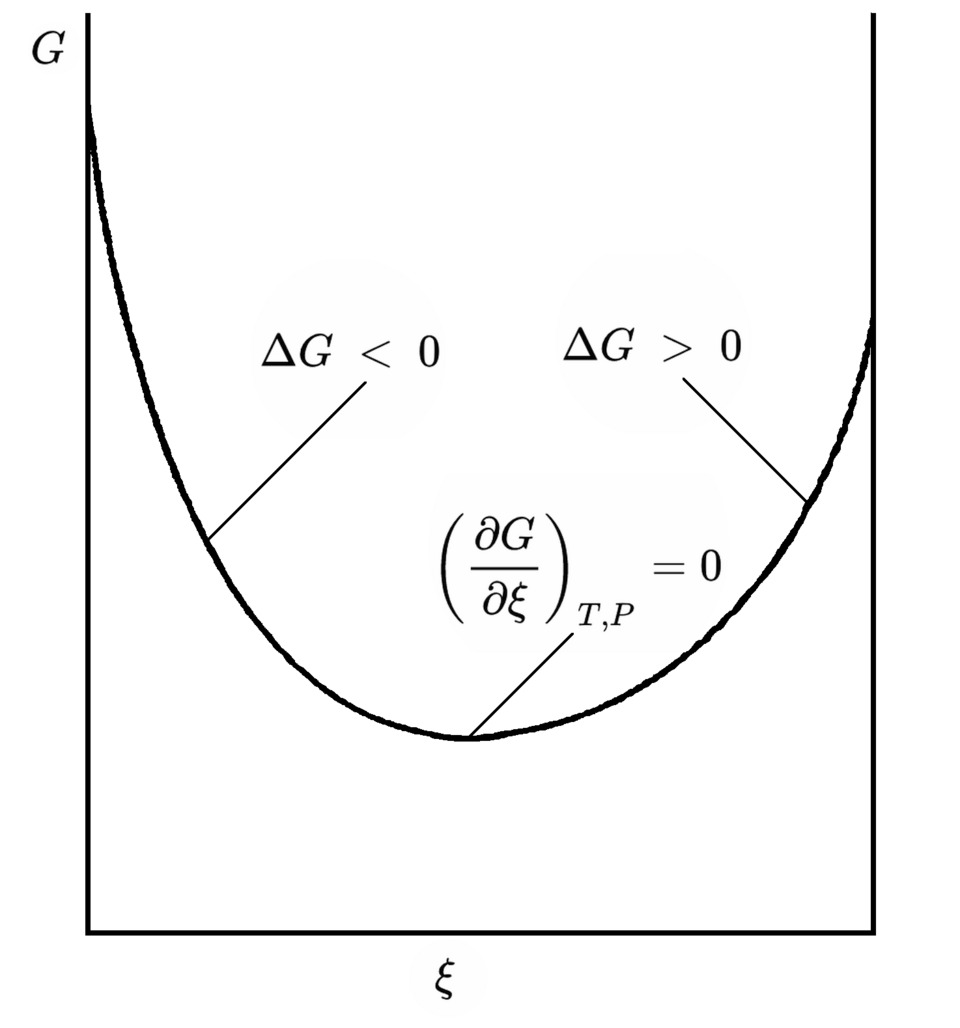
Graph of Gibbs free energy versus extent of reaction (reaction progress), illustrating that equilibrium occurs at the minimum of where . The downhill direction indicates the spontaneous shift toward equilibrium, connecting the idea of thermodynamic favorability to the equilibrium state rather than to kinetics. Source
What K communicates at equilibrium
Equilibrium constant, K: a ratio (built from equilibrium amounts of products to reactants, each raised to stoichiometric powers) that indicates the position of equilibrium for a particular reaction at a given temperature.
K summarizes where equilibrium lies:
K > 1: equilibrium is product-favored (products predominate relative to reactants).
K < 1: equilibrium is reactant-favored.
K ≈ 1: neither side is strongly favored.
Connecting the sign of ΔG° to the size of K (standard conditions)
Under standard conditions, ΔG° and K are consistent ways to describe the same thermodynamic preference:
If ΔG° < 0, then products are favored at equilibrium, so K > 1.
If ΔG° > 0, then reactants are favored at equilibrium, so K < 1.
If ΔG° = 0, then neither direction is favored under standard conditions, so K = 1.
This connection is a key interpretation skill: ΔG° provides the thermodynamic “direction”, while K describes the equilibrium mixture that results.
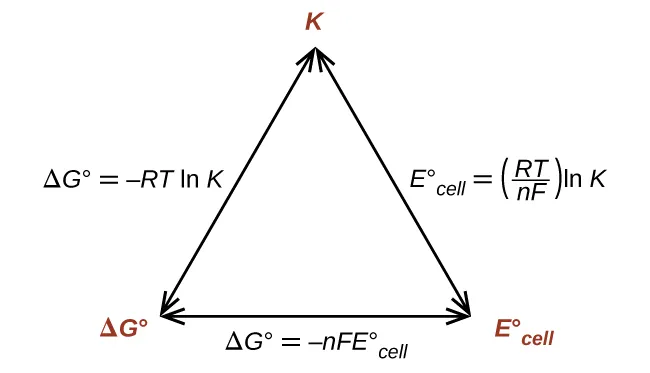
Triangle summary diagram connecting , , and (for electrochemical reactions) . The arrows encode the key relationship , making it visually obvious that negative corresponds to (product-favored equilibrium) and positive corresponds to . Source
What “favored at equilibrium” does (and doesn’t) mean
Favored refers to equilibrium composition, not completion
Even when K > 1, the reaction does not have to go to 100% products. “Product-favored” means the equilibrium ratio leans toward products, not that reactants disappear.
Likewise, K < 1 does not mean “no products form”; it means equilibrium contains proportionally more reactants than products.
Standard conditions matter for ΔG°
ΔG° is tied to standard conditions. The statement “ΔG° < 0 implies K > 1” is specifically about interpreting thermodynamic favorability and equilibrium position in the standard-state framework at that temperature.
Common interpretation checks
Always interpret ΔG° with its sign: negative = favored, positive = unfavored (under standard conditions).
Always interpret K relative to 1: greater than 1 = products favored, less than 1 = reactants favored.
Remember that both ΔG° and K are temperature-dependent; the link is made at a specified temperature under standard conditions.
FAQ
Pure solids and liquids have essentially constant “effective concentration” (activity), so they are treated as 1 in $K$ expressions.
The ΔG°–K link still holds; those phases are simply not variables that shift the value of $K$.
In non-ideal systems, $K$ is defined using activities rather than concentrations/partial pressures.
At higher concentrations or ionic strength, activity coefficients matter, so experimentally determined “$K$” based on concentration may deviate from the true thermodynamic constant.
Not necessarily.
A small negative $\Delta G^\circ$ implies $K$ is only modestly greater than 1, so appreciable reactants can remain at equilibrium.
Yes.
$K$ fixes the equilibrium ratio, but the actual equilibrium amounts also depend on starting quantities, stoichiometry, and constraints (for example, limited initial reactant).
It comes from minimising Gibbs free energy at equilibrium and expressing the reaction Gibbs energy in terms of standard-state contributions plus a term involving the logarithm of activities.
Setting the equilibrium condition leads to a constant that is identified as $K$.
Practice Questions
Under standard conditions at 298 K, a reaction has . State whether is greater than 1, less than 1, or equal to 1, and justify your choice.
States . (1)
Links to thermodynamic favourability under standard conditions. (1)
Connects “favoured” to products being favoured at equilibrium. (1)
For three reactions at the same temperature under standard conditions:
Reaction A:
Reaction B:
Reaction C:
For each reaction, state the expected sign of (negative, zero, or positive) and describe whether equilibrium is product-favoured or reactant-favoured.
Reaction A: so . (1) Product-favoured. (1)
Reaction B: so . (1) Neither favoured. (1)
Reaction C: so . (1) Reactant-favoured. (1)

