Balanced chemical equations
A chemical equation shows the stoichiometric ratio of reactants and products.
Always balance the equation first before doing any calculation.
Use state symbols correctly: (s) solid, (l) liquid, (g) gas, (aq) aqueous.
The coefficients in a balanced equation give the mole ratio.
Mole ratio = conversion bridge between any two substances in the equation.
You may need to deduce an equation from the reactants and products named in the question.
Core stoichiometric method
Convert the given quantity to moles first unless the quantity is already in moles.
Use the mole ratio from the balanced equation.
Convert from moles to the required final quantity.
Common conversion routes:
mass moles using
moles mass using
solution volume and concentration moles using
moles gas volume using molar volume only when conditions allow
Always check:
equation balanced
units consistent
significant figures sensible
Mass calculations
Use relative atomic masses, , from the data booklet to calculate molar mass, .
Use values to 2 decimal places where given.
Typical structure of a mass question:
calculate moles of known substance
use coefficient ratio
convert to mass of required substance
The coefficient ratio works only with moles, not directly with grams.
Do not compare masses directly unless you have converted to moles.
Gas-volume calculations
For gases under the same temperature and pressure, the coefficients can be used as volume ratios.
If gas is at STP and molar volume is needed, use the value from the data booklet.
For mixed questions, use a chain such as:
mass moles moles gas volume
Only use gas volume relationships for substances in the gas state.
Read the question carefully for temperature/pressure conditions.
Solution stoichiometry
Molar concentration means moles of solute per dm of solution.
Use square brackets for concentration, e.g. [HCl].
Key relationship: .
Volume must usually be in dm, so convert from cm by dividing by 1000.
Typical method:
find moles in solution from concentration and volume
use the mole ratio
convert to the required mass, concentration, or volume
Be alert to unit conversions between mol dm and g dm.

This image shows how a volumetric pipette and volumetric flask are used to make accurate solutions, linking directly to IB practical work involving molarity and standard solution preparation. Source
Limiting reactant and excess reactant
The limiting reactant is the reactant that is used up first.
The limiting reactant determines the theoretical yield.
The excess reactant is left over after the reaction finishes.
To identify the limiting reactant:
convert each reactant to moles
compare using the stoichiometric ratio from the balanced equation
the reactant producing the smaller amount of product is limiting
A fast method is to divide each reactant amount in moles by its coefficient; the smaller value is limiting.
Once the limiting reactant is known, ignore the excess reactant for the yield calculation.
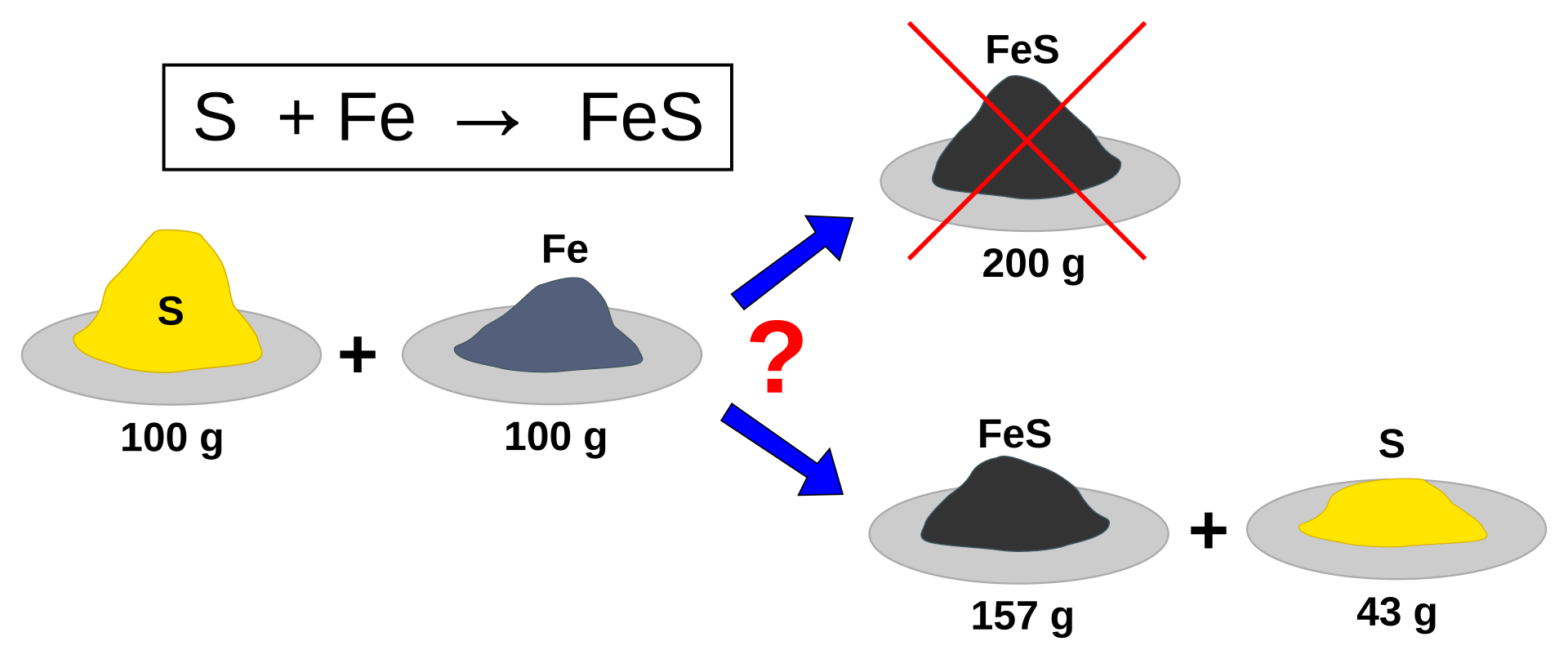
This diagram illustrates the idea of a limiting reagent by showing that one reactant is consumed first and therefore fixes the maximum amount of product. It is a clean visual reminder that stoichiometry depends on particle ratios, not on whichever mass looks smaller. Source
Theoretical yield, experimental yield, percentage yield
Theoretical yield = maximum amount of product predicted from the limiting reactant.
Experimental yield = amount of product actually obtained in the lab.
Percentage yield measures how successful the reaction was.
Formula:
Percentage yield is usually less than 100% because of:
incomplete reaction
side reactions
product loss during transfer, filtration, drying, or purification
A value greater than 100% usually suggests impurities, wet product, or measurement error.
Atom economy
Atom economy measures the efficiency of a reaction in terms of how many atoms from the reactants end up in the desired product.
High atom economy means less waste and is important in green chemistry.
A reaction can have high percentage yield but poor atom economy.
Percentage yield tells you how well the reaction worked in practice; atom economy tells you how much waste the reaction design creates.
Addition reactions often have high atom economy because most or all atoms enter the desired product.
In exam questions, identify the desired product carefully before using the formula from the data booklet.

This diagram compares reactions with high and low atom economy, making the idea of useful product versus waste products very easy to see. It is especially helpful for explaining why atom economy is about reaction design, not lab technique. Source
Common exam traps
Using an unbalanced equation.
Applying the coefficient ratio directly to mass or volume when you should first convert to moles.
Forgetting to convert cm to dm in solution questions.
Using the wrong reactant instead of the limiting reactant.
Confusing theoretical yield with experimental yield.
Mixing up percentage yield and atom economy.
Forgetting that gas-volume ratios only apply when gases are under the same conditions.
Checklist: can you do this?
Balance a chemical equation with correct state symbols.
Calculate reacting mass, gas volume, concentration, or moles using a mole ratio.
Identify the limiting reactant and the excess reactant from data.
Calculate theoretical yield, percentage yield, and atom economy.
Interpret whether a process is efficient in terms of both yield and waste.
Exam quick strategy
1. Balance the equation.
2. Convert the given data to moles.
3. Use the coefficient ratio.
4. Convert to the quantity required.
5. Check for limiting reactant, units, and significant figures.
One-line summary
Stoichiometry is the use of a balanced equation and mole ratios to calculate how much substance reacts or forms.