Redox essentials
Oxidation = loss of electrons, increase in oxidation state, gain of oxygen, or loss of hydrogen.
Reduction = gain of electrons, decrease in oxidation state, loss of oxygen, or gain of hydrogen.
Redox reactions always involve both oxidation and reduction happening together.
The oxidizing agent is reduced because it accepts electrons.
The reducing agent is oxidized because it donates electrons.
In exam questions, identify redox by spotting electron transfer, oxidation state changes, or oxygen/hydrogen transfer.
Be confident with variable oxidation states, especially for transition metals and common non-metals.
Oxidation states: fast rules
Elements in their standard state have oxidation state 0.
Monatomic ions have oxidation state equal to their charge.
Group 1 metals are usually +1; Group 2 metals are usually +2.
Fluorine is always −1.
Oxygen is usually −2, but −1 in peroxides.
Hydrogen is usually +1, but −1 in metal hydrides.
The sum of oxidation states equals the overall charge on the species.
Oxidation states are a bookkeeping tool: they help track redox even when reactions are covalent, not just ionic.
Identifying what is oxidized and reduced
Oxidized species: oxidation state increases.
Reduced species: oxidation state decreases.
Oxidizing agent causes another species to be oxidized, so it is itself reduced.
Reducing agent causes another species to be reduced, so it is itself oxidized.
In combustion, oxygen is usually the oxidizing agent.
In metal displacement, the more reactive metal is usually oxidized.
Half-equations and balancing redox
Half-equations show oxidation and reduction separately.
In oxidation half-equations, electrons appear on the products side.
In reduction half-equations, electrons appear on the reactants side.
For acidic or neutral solution, balance in this order:
balance all atoms except O and H
balance O using HO
balance H using H
balance charge using e
Multiply half-equations so the number of electrons cancels, then add them.
Final equations must have atoms balanced and overall charge balanced.
Only use half-equations when they make electron transfer clearer or when asked explicitly.
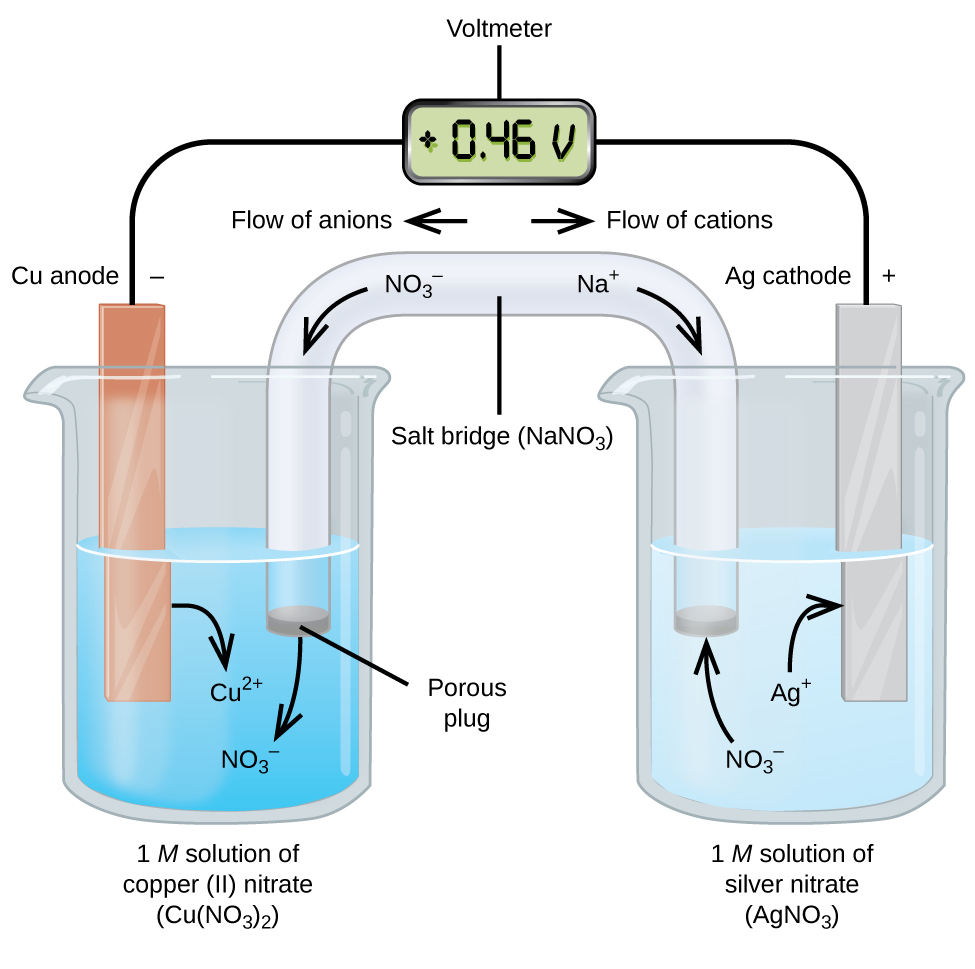
This image shows a voltaic (galvanic) cell with anode, cathode, electron flow in the wire, and ion movement through the salt bridge. It is useful for linking half-equations to the physical structure of an electrochemical cell. Source
Relative reactivity and displacement reactions
The ease of oxidation of metals can be predicted from their position in the periodic table.
More reactive metals lose electrons more easily, so they are more easily oxidized.
A more reactive metal can displace a less reactive metal ion from solution.
General pattern:
metal + metal ion solution → reaction only if the solid metal is more reactive.Example: Zn(s) + Cu(aq) → Zn(aq) + Cu(s).
For halogens, reactivity is discussed in terms of ease of reduction.
A more reactive halogen is more easily reduced and can displace a less reactive halide ion.
In data questions, use the observations given rather than memorizing a full reactivity series.
Metals and acids
Reactive metals react with dilute acids to produce hydrogen gas.
General pattern: metal + acid → salt + H.
Here, H is reduced to H, and the metal is oxidized to its ion.
This is both a redox reaction and an acid–metal reaction.
Typical half-equations:
M → M + ne
2H + 2e → H
Less reactive metals may not react with dilute acid.
Electrochemical cells: core ideas
Oxidation always occurs at the anode.
Reduction always occurs at the cathode.
Remember: AnOx, RedCat.
In any electrochemical cell, identify electrodes by the reaction occurring, not by memorizing sign only.
The sign/polarity depends on whether the cell is voltaic or electrolytic.
Voltaic (primary) cells
A voltaic cell converts chemical energy from a spontaneous redox reaction into electrical energy.
In a voltaic cell:
anode is negative
cathode is positive
Electrons flow through the external circuit from anode → cathode.
The salt bridge completes the circuit by allowing ion movement, not electron movement.
In the salt bridge:
anions move toward the anode compartment
cations move toward the cathode compartment
Typical cell set-up includes two half-cells, electrodes, electrolytes, wire, and salt bridge.

This image shows the classic Zn/Cu voltaic cell, a standard model for explaining anode, cathode, electron flow, and salt bridge ion movement. It is especially useful for visualizing why the anode is negative and the cathode is positive in a voltaic cell. Source
Secondary (rechargeable) cells
Secondary cells use redox reactions that can be reversed by applying electrical energy.
Discharge = cell acts like a voltaic cell.
Charging = reactions are forced in the reverse direction.
If given discharge half-equations, reverse them to get the charging reactions.
Know the broad comparison:
Primary cells: not rechargeable
Secondary cells: rechargeable
Fuel cells: reactants supplied continuously from outside
Electrolytic cells
An electrolytic cell converts electrical energy into chemical energy.
It drives a non-spontaneous redox reaction.
In an electrolytic cell:
anode is positive
cathode is negative
Oxidation still happens at the anode and reduction at the cathode.
Current is conducted by:
electrons in the external circuit
moving ions in the electrolyte
For a molten salt, only the ions from the salt are present, so products are easier to predict.
Electrolysis of molten salts
In a molten salt, the cation is reduced at the cathode.
The anion is oxidized at the anode.
Example: molten NaCl
cathode: Na + e → Na
anode: 2Cl → Cl + 2e
Overall: 2NaCl(l) → 2Na(l) + Cl(g)
Key exam point: in molten salts, water is absent, so there is no competition from water.
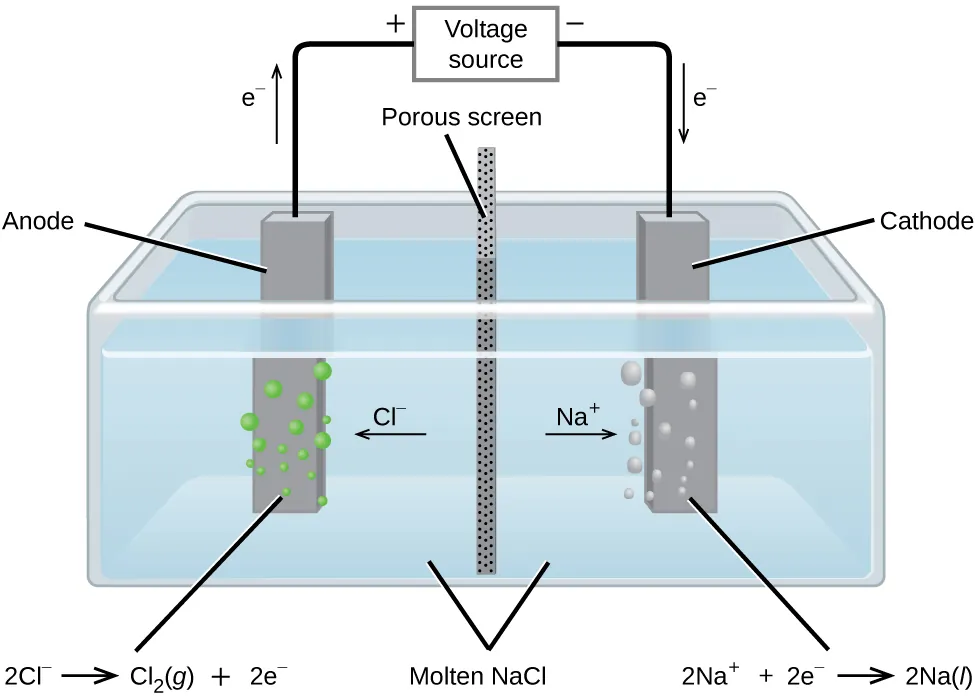
The diagram shows an electrolytic cell for molten sodium chloride, including the power source, anion/cation movement, and products at each electrode. It is ideal for comparing electrolysis with a voltaic cell. Source
Organic oxidation
Organic oxidation usually means more bonds to oxygen and/or fewer bonds to hydrogen.
Primary alcohols oxidize in two steps:
primary alcohol → aldehyde
aldehyde → carboxylic acid
Secondary alcohols oxidize to ketones.
Tertiary alcohols are not oxidized under similar conditions.
You may be asked to deduce equations showing only the functional group change.
Example patterns:
RCHOH → RCHO → RCOOH
RCHOH → RCO
Distinguish apparatus:
distillation helps remove the aldehyde as it forms, limiting further oxidation
reflux keeps products in the flask, allowing further oxidation to the carboxylic acid
Specific oxidizing agents and full mechanisms are not required.

This image summarizes the organic redox patterns required in the syllabus: primary alcohols can form aldehydes then carboxylic acids, secondary alcohols form ketones, and tertiary alcohols resist oxidation. It is a strong revision visual for quick product prediction. Source
Organic reduction
Organic reduction usually means fewer bonds to oxygen and/or more bonds to hydrogen.
Carboxylic acids can be reduced to primary alcohols via an aldehyde intermediate.
Ketones are reduced to secondary alcohols.
In these reductions, hydride ions act as the reducing species.
Typical pattern:
RCOOH → RCHO → RCHOH
RCO → RCHOH
Focus on deducing products and recognizing oxidation vs reduction from structural change.
Reduction of unsaturated compounds
Addition of hydrogen to alkenes and alkynes is a reduction.
Reduction lowers the degree of unsaturation.
Examples:
alkene + H → alkane
alkyne + H → alkene / alkane depending on extent of hydrogenation
In questions, identify this both as hydrogenation and reduction.
Checklist: can you do this?
Assign oxidation states quickly and correctly, including peroxides and hydrides.
Identify the oxidized species, reduced species, oxidizing agent, and reducing agent.
Balance half-equations and combine them into a full redox equation in acidic/neutral solution.
Predict products at the anode and cathode for voltaic cells, electrolytic cells, and molten salt electrolysis.
Deduce organic oxidation/reduction products and choose between reflux and distillation.
HL only — standard electrode potentials
The standard hydrogen electrode (SHE) is assigned E = 0.00 V by convention.
Standard electrode potentials are listed as reduction potentials.
A more positive E means the species is more easily reduced.
A more negative E means the reverse process, oxidation, is more favorable.
Use tables of standard reduction potentials to compare oxidizing and reducing strength.
If necessary, reverse a half-equation when considering oxidation, but remember the table value is still quoted for the reduction direction.
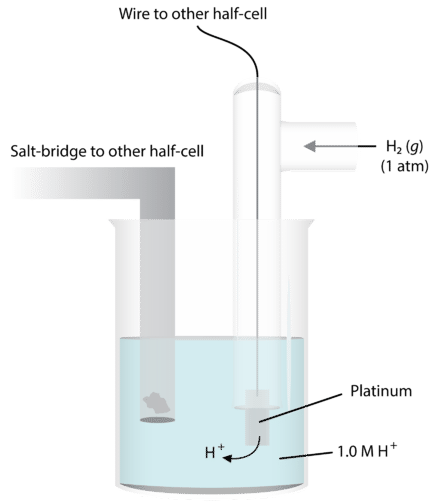
This image shows the reference half-cell used to define E = 0.00 V, including the Pt electrode, H gas, and H solution. It helps students connect the definition of standard electrode potential to a real apparatus. Source
HL only — cell potentials and spontaneity
Calculate standard cell potential using: E = E − E.
Use reduction potentials directly from the table; do not change signs first.
If E > 0, the reaction is spontaneous in the forward direction.
If E < 0, the forward reaction is non-spontaneous.
The reverse reaction would then be spontaneous.
Cell potential links to Gibbs energy by: ΔG = −nFE.
If E is positive, then ΔG is negative.
HL only — electrolysis of aqueous solutions
In aqueous electrolysis, there can be competing reactions because water may also be oxidized or reduced.
Products depend on the relative ease of discharge, using standard electrode potentials.
At the cathode, either the dissolved cation or water/H may be reduced.
At the anode, either the anion or water may be oxidized.
Be especially ready for questions on aqueous NaCl and aqueous CuSO.
You must use the data booklet values plus any information given about concentration or electrode type.
HL only — electroplating
Electroplating uses an electrolytic cell to coat an object with a thin layer of metal.
The object being plated is the cathode.
The plating metal is usually the anode.
The electrolyte contains ions of the plating metal.
Cathode half-equation: metal ion + electrons → metal coating.
Anode half-equation: metal → metal ion + electrons.
Common reasons: corrosion resistance, appearance, and surface protection.
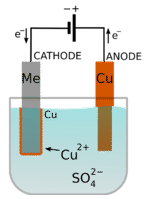
This image shows how an electrolytic cell is arranged for electroplating, including the coating metal, object to be plated, and metal-ion electrolyte. It is useful for memorizing which electrode is the cathode in plating questions. Source
Exam traps to avoid
Do not confuse oxidizing agent with the species that is oxidized.
Do not assume the anode is always positive; in voltaic cells it is negative, in electrolytic cells it is positive.
Do not forget that oxidation occurs at the anode in all electrochemical cells.
In E calculations, use reduction potentials from the table exactly as given.
In aqueous electrolysis, do not ignore possible reactions involving water.
Tertiary alcohols do not oxidize under the same conditions as primary and secondary alcohols.
Hydrogenation of an alkene is both an addition reaction and a reduction reaction.