Nucleophiles and electrophiles
Electron-pair sharing reactions involve one species donating an electron pair and another accepting that electron pair.
A nucleophile is an electron-pair donor that forms a bond to an electrophile.
Common nucleophiles: OH⁻, CN⁻, NH₃, H₂O, halide ions, alkenes (π bond).
Nucleophiles are often negative ions or neutral molecules with a lone pair.
An electrophile is an electron-pair acceptor that forms a bond by accepting both bonding electrons from the nucleophile.
Common electrophiles: H⁺, δ⁺ carbon in C–X bonds, Br₂ (after polarization), carbocations, NO₂⁺.
Electrophiles are often positively charged or electron-deficient species.
Exam cue: identify the electron-rich species as the nucleophile and the electron-poor species as the electrophile.
Nucleophilic substitution (SL core)
In nucleophilic substitution, a nucleophile forms a new bond to carbon while another bond breaks to produce a leaving group.
Typical substrate: halogenoalkane with a polar C–X bond.
The carbon attached to the halogen is δ⁺, so it is attacked by the nucleophile.
General pattern: Nu: + R–X → R–Nu + X⁻.
A leaving group is the species that leaves with the bonding pair, usually X⁻.
Know how to use curly arrows: they show the movement of an electron pair.
At SL, you must be able to deduce equations, describe electron-pair movement, and identify nucleophile, electrophile, and leaving group.
Typical substitutions of halogenoalkanes:
R–X + OH⁻ → R–OH + X⁻
R–X + NH₃ → R–NH₂ + HX or, in two-step form, formation of an alkylammonium intermediate followed by deprotonation
R–X + CN⁻ → R–CN + X⁻
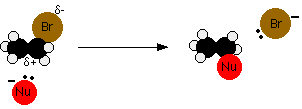
This image shows a nucleophile attacking the δ⁺ carbon of a halogenoalkane while the leaving group departs. It is useful for recognizing the core pattern of nucleophilic substitution and where to place curly arrows in exam mechanisms. Source
Heterolytic fission and curly arrows
Heterolytic fission is bond breaking where both bonding electrons go to one atom.
This produces ions, not radicals.
Example idea: R–Br → R⁺ + Br⁻ if the bonding pair goes to Br.
In nucleophilic substitution and electrophilic addition mechanisms, use full curly arrows for electron pairs.
Start the curly arrow from a lone pair, negative charge, or bond.
End the arrow at the atom or bond where the electron pair moves.
Do not use single-barbed arrows here; those are for radical mechanisms.
Exam trap: confusing heterolytic fission with homolytic fission.
Electrophilic addition of alkenes (SL core)
Alkenes are susceptible to electrophilic attack because the π bond has high electron density.
The π bond acts as a nucleophile.
Electrophilic addition means an electrophile attacks the double bond and the product has two new single bonds instead of one double bond.
You must be able to deduce equations for reactions of alkenes with:
halogens
hydrogen halides
water
Core examples:
Ethene + Br₂ → 1,2-dibromoethane
Ethene + HBr → bromoethane
Ethene + H₂O → ethanol
Bromine water is decolourized by alkenes because Br₂ adds across the C=C bond.
At SL, you are not required to memorize detailed mechanisms for these alkene additions, but you must know why alkenes react and be able to write products/equations.
This diagram shows the electron-rich π bond polarizing Br₂ and the steps leading to addition across the double bond. It is especially useful for explaining why bromine water is decolourized by alkenes. Source
Lewis acids, Lewis bases and coordination bonds
A Lewis acid is an electron-pair acceptor.
A Lewis base is an electron-pair donor.
This definition is broader than Brønsted–Lowry because it does not require proton transfer.
When a Lewis base reacts with a Lewis acid, a coordination bond forms.
A coordination bond is a covalent bond where both bonding electrons come from the same atom.
Link the ideas together:
nucleophiles = Lewis bases
electrophiles = Lewis acids
Classic examples:
NH₃ + H⁺ → NH₄⁺
H₂O + H⁺ → H₃O⁺
BF₃ + :NH₃ → F₃B←NH₃
In Lewis structures, show the donated pair and the new coordinate bond clearly.
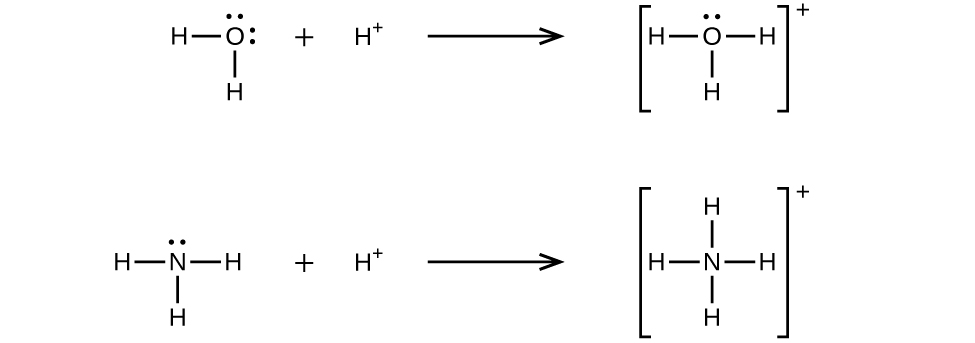
These diagrams show a Lewis base donating a lone pair into an electron-deficient Lewis acid to make a coordinate covalent bond. They are ideal for linking Lewis acid–base theory to nucleophile/electrophile behavior. Source
HL only: complex ions and ligand bonding
Ligands are Lewis bases that donate a lone pair to a transition metal cation.
The metal ion acts as a Lewis acid.
Complex ions form by coordination bond formation between ligands and metal ions.
To deduce the charge on a complex ion:
add the metal ion charge
add the charges of all ligands
Common neutral ligands: H₂O, NH₃.
Common negative ligands: Cl⁻, CN⁻.
Example: in [Cu(NH₃)₄]²⁺, NH₃ is neutral, so charge stays 2+.
Example: in [Fe(CN)₆]⁴⁻, each CN⁻ is –1, so metal oxidation state must balance to give overall 4−.
HL only: SN1 vs SN2 nucleophilic substitution
Primary halogenoalkanes usually react by SN2.
Tertiary halogenoalkanes usually react by SN1.
Secondary halogenoalkanes can react by either mechanism.
SN2 is one-step and concerted: bond making and bond breaking happen simultaneously.
SN1 is two-step: first the leaving group departs, then the nucleophile attacks the carbocation.
SN2 rate equation: rate = k[halogenoalkane][nucleophile].
SN1 rate equation: rate = k[halogenoalkane].
SN2 shows stereospecific inversion because the nucleophile attacks from the opposite side.
Exam cue: primary = SN2, tertiary = SN1 is the key first decision.
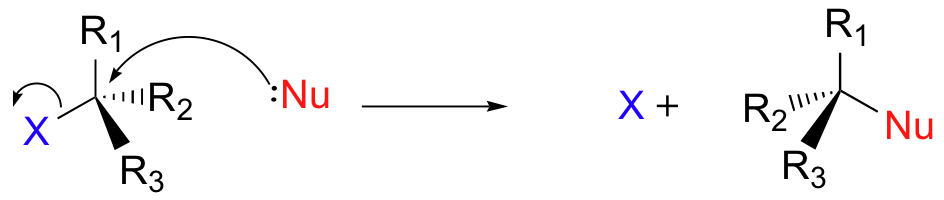
These images compare the one-step SN2 pathway with the two-step SN1 pathway via a carbocation intermediate. They are useful for linking mechanism, rate equation, and stereochemical outcome. Source
HL only: leaving group strength and substitution rate
The rate of nucleophilic substitution depends partly on the identity of the leaving group.
Better leaving groups form more stable ions and break away more easily.
For halogenoalkanes, a common trend is I > Br > Cl as leaving groups.
Therefore, substitution is generally fastest for iodoalkanes and slowest for chloroalkanes (with comparable structures).
Link to bonding: a weaker C–I bond is easier to break than a stronger C–Cl bond.
Exam wording often asks you to explain rate differences using bond strength and leaving-group ability.
HL only: electrophilic addition mechanisms
Symmetrical alkenes react with halogens, water, and hydrogen halides by electrophilic addition.
The first step is attack of the π bond on an electrophile.
For unsymmetrical alkenes, the major product depends on the stability of the carbocation intermediate.
More stable carbocations are favored: tertiary > secondary > primary.
This explains why one product is usually formed in greater amount than another.
In exam answers, explain the major product by saying it forms via the more stable carbocation.
HL only: electrophilic substitution of benzene
Benzene reacts by electrophilic substitution, not addition.
Reason: benzene is stabilized by delocalized π electrons; addition would destroy aromatic stability.
General pattern:
ring attacks E⁺
a positive intermediate forms
H⁺ is lost and aromaticity is restored
You need to describe the mechanism for reaction of benzene with a charged electrophile, E⁺.
Key exam phrase: substitution preserves aromaticity; addition would remove it.
A standard example is nitration of benzene using an electrophile such as NO₂⁺.
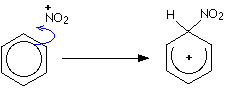
This mechanism shows benzene attacking an electrophile and then losing H⁺ to restore aromaticity. It is the clearest visual summary of why benzene undergoes electrophilic substitution rather than addition. Source
Exam patterns and quick comparisons
Nucleophile = electron-pair donor; electrophile = electron-pair acceptor.
Nucleophilic substitution is most associated with halogenoalkanes.
Electrophilic addition is most associated with alkenes.
Electrophilic substitution is most associated with benzene.
Heterolytic fission makes ions; homolytic fission makes radicals.
Curly arrows show electron-pair movement; fish-hook arrows show single-electron movement.
Lewis base = nucleophile and Lewis acid = electrophile is a high-value linking idea.
Checklist: can you do this?
Identify the nucleophile, electrophile, and leaving group in a reaction.
Draw curly arrows correctly for electron-pair movement and distinguish them from fish-hook arrows.
Deduce products/equations for nucleophilic substitution and electrophilic addition reactions.
Explain why alkenes undergo addition but benzene undergoes substitution.
For HL, decide whether a halogenoalkane reaction is more likely SN1 or SN2, and justify it with mechanism and rate law.