Brønsted–Lowry acids and bases
Brønsted–Lowry acid = proton donor.
Brønsted–Lowry base = proton acceptor.
In aqueous chemistry, a proton can be written as or .
To identify acid/base roles in an equation, track which species loses and which species gains .
Base does not always mean alkali: an alkali is a soluble base that produces in water.
Typical example:
acts as the base.
acts as the acid.
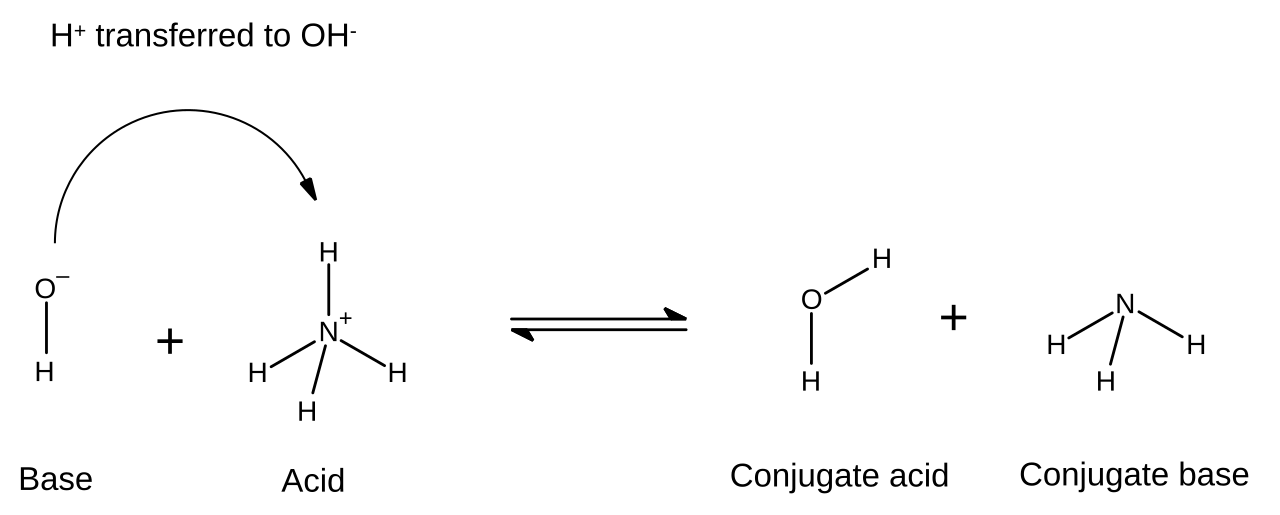
This diagram shows a proton transfer equilibrium between ammonium/ammonia and water/hydroxide, making the acid, base, conjugate acid, and conjugate base easy to identify. It is ideal for practicing how to label Brønsted–Lowry species in both forward and reverse directions. Source
Conjugate acid–base pairs
A conjugate acid–base pair consists of two species differing by one proton.
When an acid loses , it forms its conjugate base.
When a base gains , it forms its conjugate acid.
Quick rule:
acid conjugate base +
base + conjugate acid
Example: in
is one conjugate pair.
is the other conjugate pair.
Exam tip: species on opposite sides of the equation that differ by one proton are the conjugate pair.
Amphiprotic species
Some species can act as both acids and bases: these are amphiprotic.
An amphiprotic species can donate in one reaction and accept in another.
Important example: water.
As a base:
As an acid:
Other common amphiprotic ions at HL include and sometimes in broader acid–base chemistry.
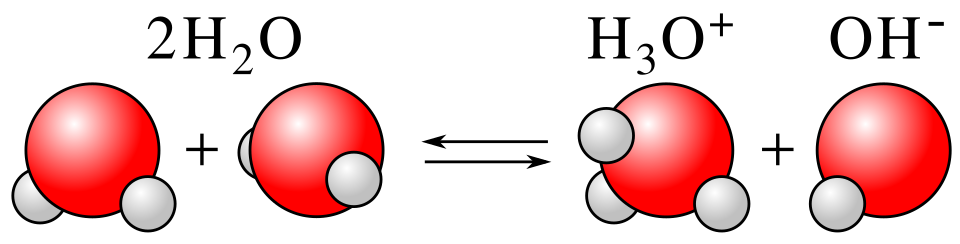
The figure shows one water molecule donating a proton to another, producing hydronium and hydroxide. It is useful for understanding why water is amphiprotic and how arises. Source
pH and hydrogen ion concentration
pH describes the hydrogen ion concentration of a solution.
Equations you must use confidently:
Lower pH = higher = more acidic.
Higher pH = lower = more basic/alkaline.
A change of 1 pH unit means a tenfold change in .
Be careful with calculator use: use 10 to the power of minus pH, not just the negative sign.
Practical methods:
Universal indicator / pH paper = estimate pH.
pH probe / meter = precise pH measurement.
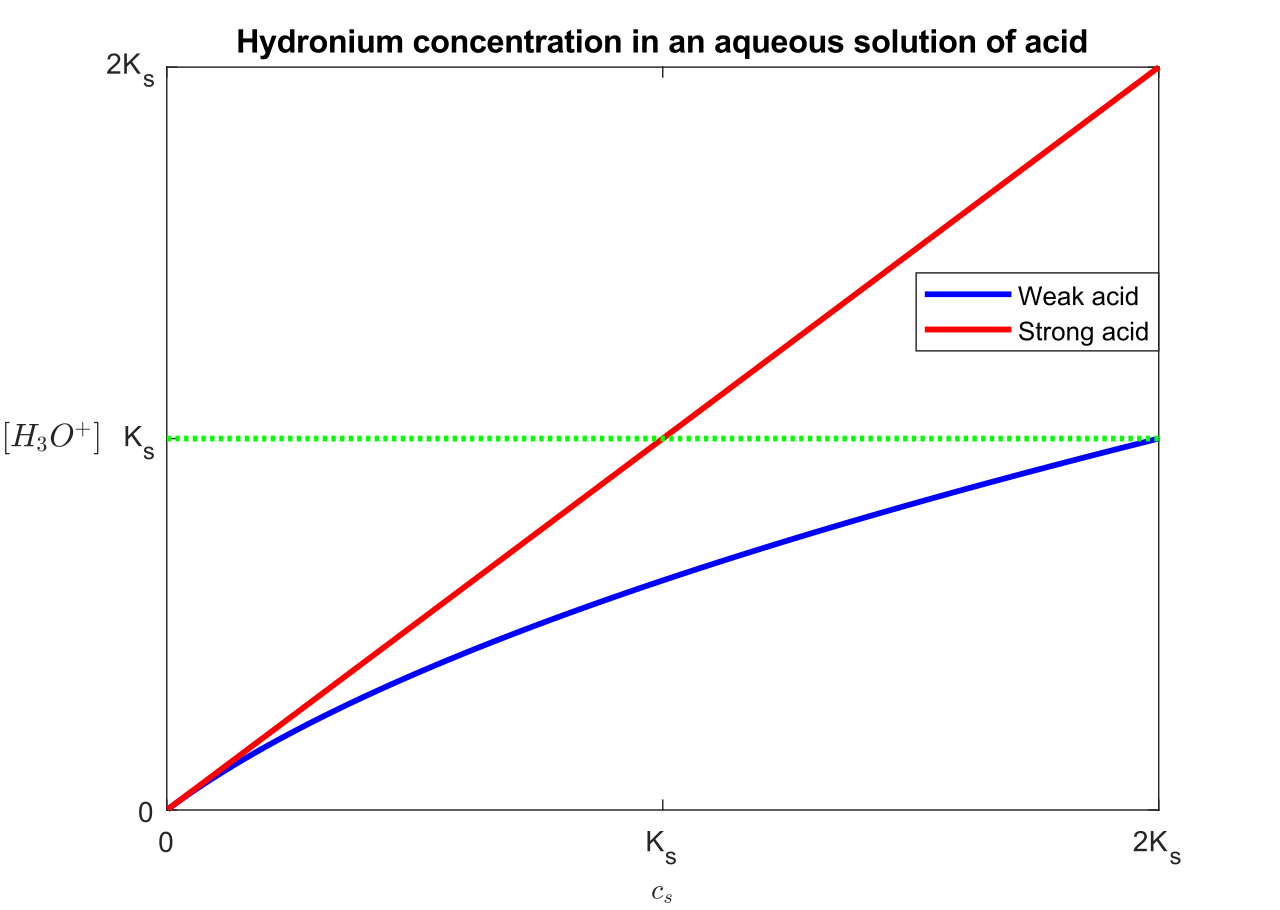
This visual represents how hydronium ion concentration changes in acidic solution, helping students connect the abstract idea of to particle-level meaning. It supports the logarithmic definition of pH. Source
Water ionization and
Water undergoes self-ionization:
or
The ion product constant of water is:
At 298 K, .
Classifying solutions:
acidic:
neutral:
basic:
In neutral water at 298 K:
mol dm
therefore pH = 7.
Common exam move: use to calculate the missing ion concentration first, then convert to pH or pOH if needed.
Strong vs weak acids and bases
Strong acids/bases ionize almost completely in water.
Weak acids/bases ionize partially in water.
Strength is about extent of ionization, not concentration.
Concentrated vs dilute describes how much solute is dissolved, not how fully it ionizes.
Acid–base equilibria lie in the direction of the weaker conjugate acid–base pair.
Strong acids commonly treated in IB include , , and .
Group 1 hydroxides are strong bases.
For equal concentration:
strong acid has lower pH than weak acid.
strong base has higher pH than weak base.
Neutralization and salts
Neutralization = acid + base salt + water.
You must be able to write equations for acids reacting with:
metal oxides
metal hydroxides
carbonates
hydrogencarbonates
General patterns:
acid + metal oxide salt + water
acid + metal hydroxide salt + water
acid + carbonate salt + water + carbon dioxide
acid + hydrogencarbonate salt + water + carbon dioxide
Bases in this topic include ammonia, amines, soluble carbonates, and hydrogencarbonates.
You should be able to identify the parent acid and parent base from a salt.
Example: comes from and .
Example: comes from and .
Strong acid–strong base titration curves
IB expects the general shape of the pH curve for a strong acid–strong base neutralization.
Key features to interpret:
initial pH
gradual rise before equivalence
steep vertical section near equivalence
equivalence point at pH 7 (for strong acid–strong base)
The equivalence point is the stoichiometric point where the amount of acid equals the amount of base according to the balanced equation.
For monoprotic titrations, use mole ratio 1:1 when appropriate.
Exam trap: end point is where the indicator changes colour; equivalence point is the true stoichiometric point.
Checklist: can you do this?
Identify the acid, base, conjugate acid, and conjugate base in any Brønsted–Lowry equation.
Calculate from pH, and pH from , without calculator mistakes.
Use to decide whether a solution is acidic, neutral, or basic.
Distinguish clearly between strong vs weak and concentrated vs dilute.
Sketch and interpret a strong acid–strong base titration curve, including equivalence point and end point.
HL only — pOH, , , salt hydrolysis, indicators and buffers
pOH equations:
At 298 K: .
/ describe acid strength; / describe base strength.
larger / smaller = stronger acid
larger / smaller = stronger base
For a conjugate pair: .
Salt hydrolysis affects pH.
ions from strong acid + strong base give ~neutral solution
makes solution acidic
and make solution basic
is amphiprotic, so analyse carefully
You must be able to write hydrolysis equations for ions in salts.
pH curves for all four monoprotic combinations:
strong acid–strong base
weak acid–strong base
strong acid–weak base
weak acid–weak base
Key HL curve features:
buffer region in weak acid/base titrations
at half-equivalence, or
equivalence point depends on the salt formed
Indicators are weak acids/bases whose conjugate forms have different colours.
Choose an indicator whose end point range overlaps the equivalence point pH.
Buffer solutions resist pH change when small amounts of acid or alkali are added.
acidic buffer = weak acid + conjugate base
basic buffer = weak base + conjugate acid
Buffer pH depends on:
the or value
the ratio of acid/base to conjugate partner
Dilution changes buffer capacity much more than buffer pH.
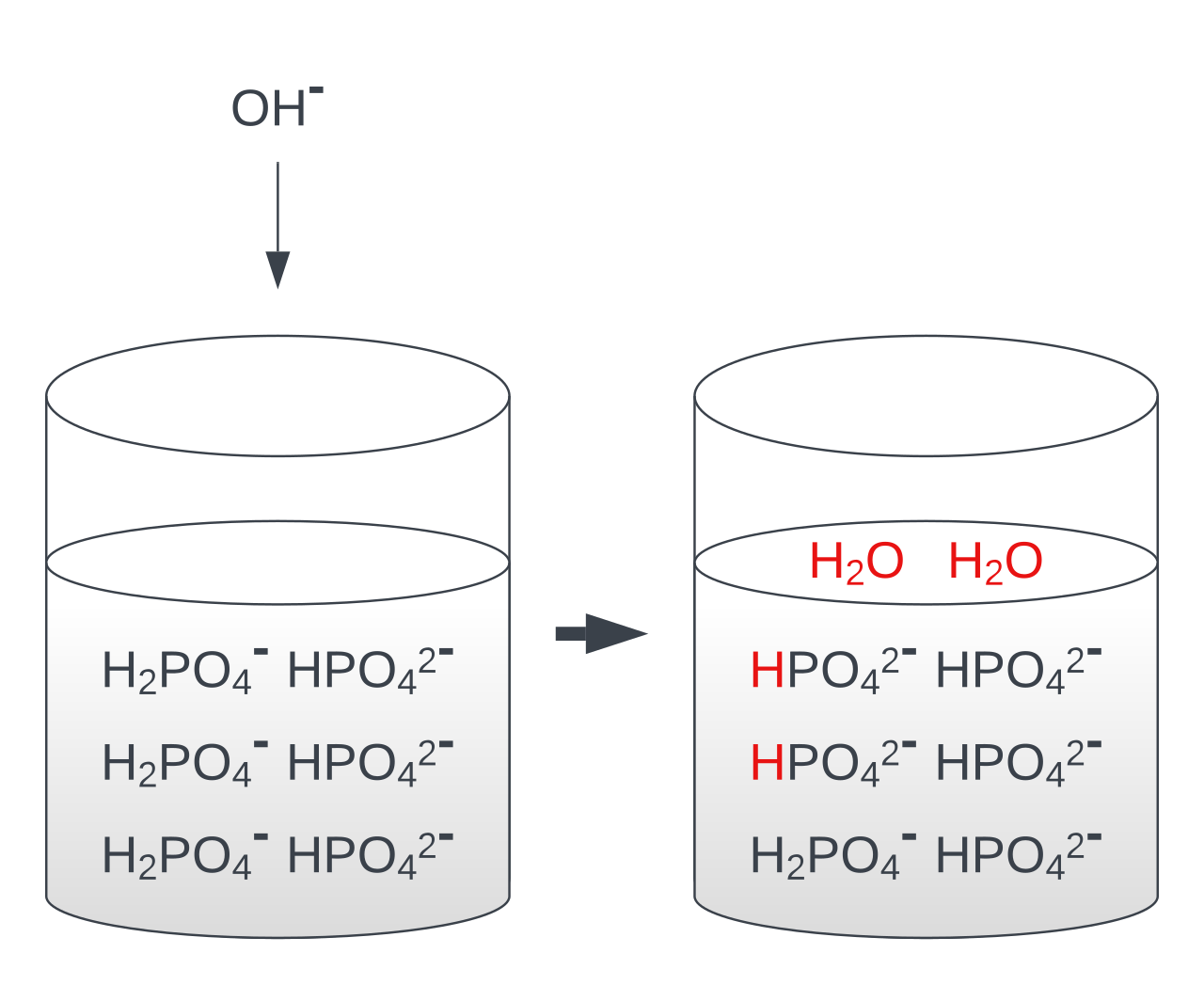
This illustration shows how a basic buffer contains a weak base and its conjugate acid, and how added acid or base is removed by equilibrium reactions. It is a strong visual aid for explaining why buffers resist pH change. Source
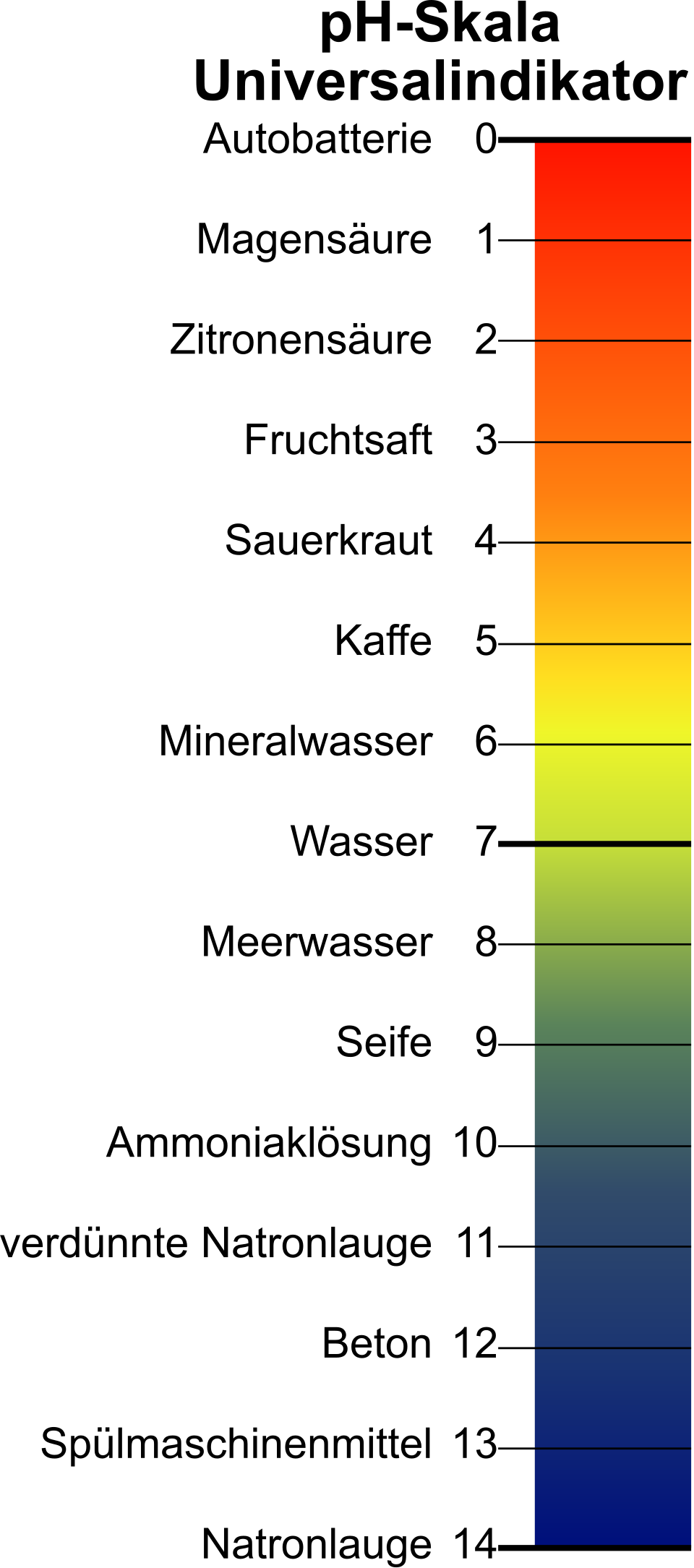
The image shows the colour changes of universal indicator across the pH scale, linking indicator colour to acidity and alkalinity. It is useful when revising estimated pH values, indicator ranges, and practical identification of acidic, neutral, and basic solutions. Source
Ultra-short exam reminders
Conjugate pairs differ by exactly one proton.
Strong means fully/almost fully ionized; concentrated means high amount dissolved.
Acidic solutions have higher than .
For strong acid–strong base titrations, equivalence point = pH 7.
At HL, always check whether a question is really about equilibrium, hydrolysis, indicator choice, or buffer action.