What this topic is about
Radicals are species with an unpaired electron.
They are highly reactive because an unpaired electron makes the species unstable.
This topic focuses on free-radical substitution of alkanes with halogens.
Core exam idea: how radicals form, how chain reactions continue, and why mixtures of products form.
Radicals: definition, notation, and identification
A radical is a molecular entity with an unpaired electron.
Radicals can be atoms, molecules, cations, or anions.
Represent radicals with a dot: Cl•, CH3•.
In mechanisms, use single-barbed arrows (fish-hook arrows) to show the movement of one electron.
Do not use a full curly arrow for radical steps; full curly arrows show electron pairs, not single electrons.
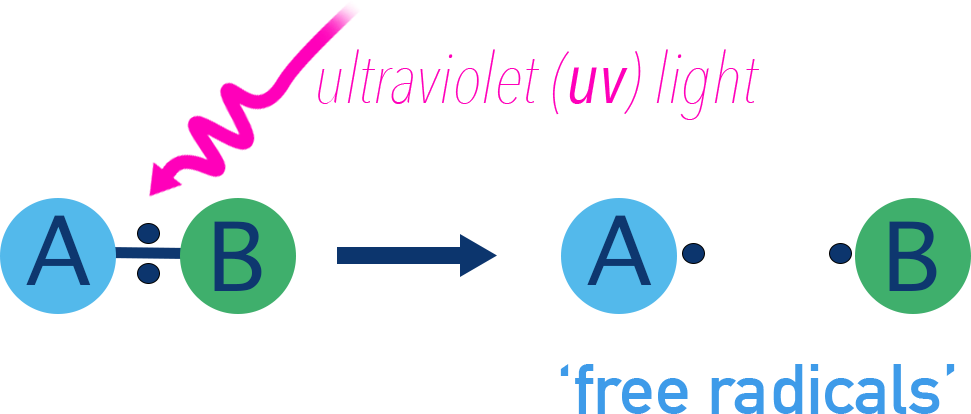
This image shows homolytic fission, where a covalent bond breaks evenly so that each atom takes one electron, forming two radicals. It is ideal for revising both radical notation and single-electron arrow notation. Source
How radicals are formed: homolytic fission
Homolytic fission = breaking a covalent bond so that each atom gets one electron.
Usually requires UV light or heat.
This is the initiation step in a chain reaction.
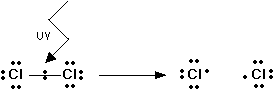
Example with chlorine:
Key exam wording:
Bond breaks homolytically
One electron goes to each atom
Two radicals are formed
Halogens such as and are common examples.
Why alkanes undergo substitution only under UV light or heat
Alkanes are relatively unreactive because C–C and C–H bonds are strong.
These bonds are also essentially non-polar, so alkanes are not easily attacked.
A halogen molecule must first produce radicals before reaction can continue.
Therefore, UV light / heat is needed to start the reaction.
Exam phrase: alkanes are stable because of strong C–C and C–H bonds and low polarity.
Free-radical substitution of alkanes: overview
General reaction: an H atom in an alkane is replaced by a halogen atom.
Example:
This is a substitution reaction because one atom/group is replaced by another.
It proceeds by a chain reaction with three stages:
Initiation
Propagation
Termination
This page shows the full free-radical substitution chain mechanism for methane and chlorine. The summary equations are especially useful for memorising the three stages and the correct radical products. Source
Initiation step
Initiation = radicals are first generated.
Example:
This starts the chain because a reactive chlorine radical is produced.
In definitions, mention homolytic fission and UV light / heat.
Propagation steps
Propagation steps keep the chain going.
A radical reacts and produces another radical.
For methane chlorination:
Why this matters:
The first step uses a chlorine radical.
The second step regenerates a chlorine radical.
Therefore the process continues as a chain reaction.
Exam wording: a radical is consumed and another radical is produced.
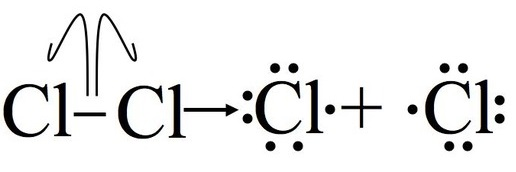
These diagrams show the stepwise chain mechanism for methane chlorination, including initiation and propagation. They are useful for seeing how one radical is regenerated, which is the key reason the reaction continues. Source
Termination steps
Termination happens when radicals combine to form a non-radical product.
This removes radicals from the system, so the chain stops.
Typical examples:
Exam wording: two radicals combine, so no new radical is formed.
Common trap: can be made in both propagation and termination; identify the step from whether a new radical is produced.
Why a mixture of products forms
Radical substitution of alkanes often gives a mixture of products.
Once a first substitution has occurred, further substitution can happen.
Example for methane chlorination:
, , , can all form.
Different radicals can also combine in different ways during termination.
Therefore, free-radical substitution is usually not a good method for making a single pure product.
Exam phrase: radical substitution with alkanes produces a mixture of products because further substitution can occur.
Exam focus: what IB wants you to explain with equations
Identify and represent radicals correctly.
Explain homolytic fission of halogens using equations.
Write initiation, propagation, and termination equations for reactions between alkanes and halogens.
Explain why alkanes are relatively stable before reaction starts.
State why the reaction gives a mixture of products.
Checklist: can you do this?
Define a radical and represent it correctly with a dot.
Write an initiation equation showing homolytic fission of a halogen under UV light.
Write propagation and termination steps for methane/chlorine or a similar alkane/halogen reaction.
Explain why propagation is a chain reaction and why termination stops it.
Explain why radical substitution produces a mixture of products.
High-yield mistakes to avoid
Do not confuse homolytic fission with heterolytic fission.
Do not use full curly arrows instead of single-barbed arrows in radical mechanisms.
Do not call termination a propagation step; propagation must regenerate a radical.
Do not forget the condition UV light or heat for initiation.
Do not say alkanes are completely unreactive; they are relatively unreactive and react under the correct conditions.
One-minute exam summary
Radicals have an unpaired electron and are highly reactive.
They are formed by homolytic fission under UV light or heat.
Radical substitution of alkanes with halogens is a chain reaction with initiation, propagation, termination.
Propagation keeps the chain going because one radical forms another radical.
Termination stops the chain because radicals combine and no new radical is produced.
A mixture of products forms because further substitution and different radical combinations are possible.