HL only: Photoelectric effect
Photoelectric effect = emission of photoelectrons from a metal surface when electromagnetic radiation shines on it.
Key evidence for the particle nature of light: electrons are emitted only if each photon has enough energy, not because of total wave intensity.
Threshold frequency = minimum frequency needed to eject electrons.
Work function = minimum energy needed to remove an electron from the metal.
Einstein’s photoelectric equation: .
Since photon energy is , emission occurs only when .
If , no electrons are emitted, however intense the light is.
Increasing frequency increases the maximum kinetic energy of emitted electrons.
Increasing intensity above threshold increases the number of emitted electrons/current, not .
Common extension: stopping potential satisfies .
Features not explained by classical wave theory: threshold frequency, instant emission, and depends on frequency not intensity.
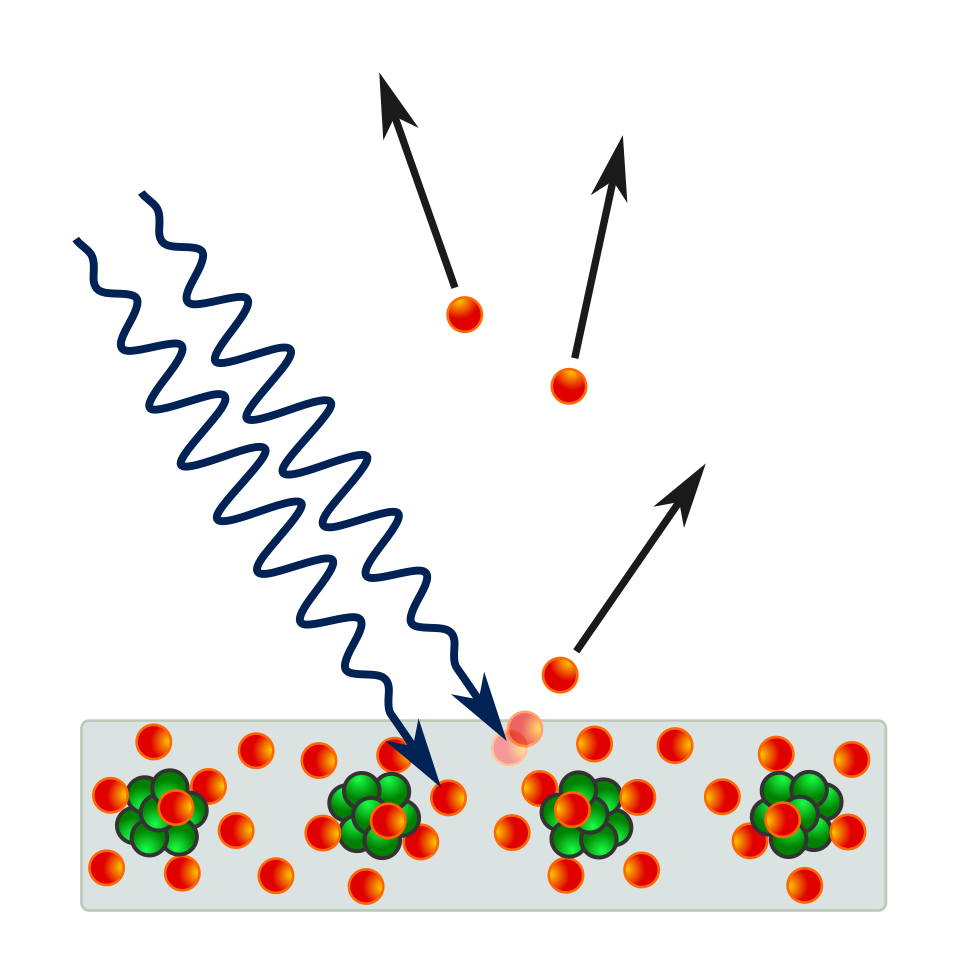
This diagram shows incoming light ejecting electrons from a solid surface. It is useful for visualizing why the effect supports the particle model of light: photons transfer energy directly to electrons. The emitted electrons represent photoelectrons leaving the metal. Source
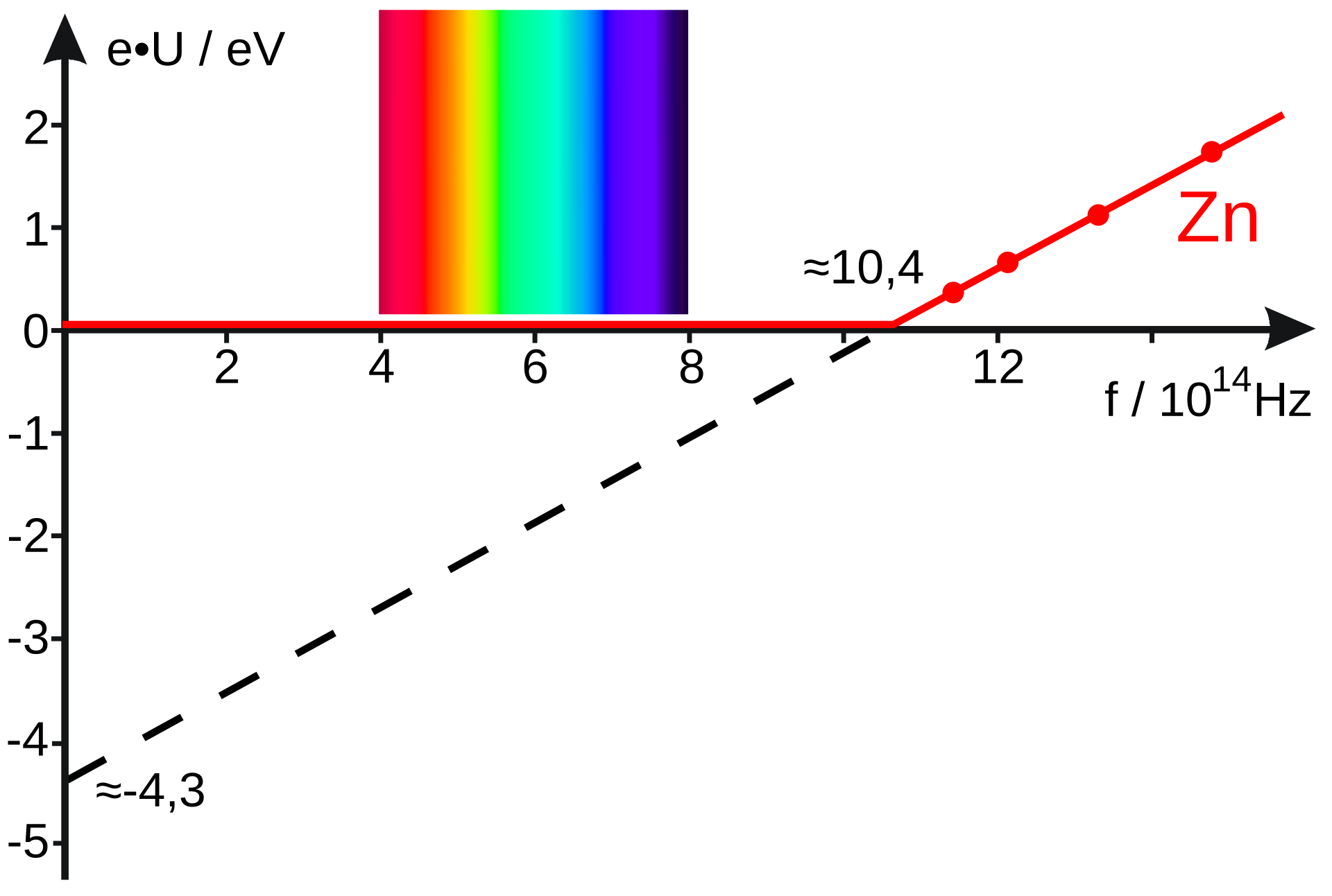
This figure is useful for the exam because it links threshold frequency, work function, and the linear dependence of maximum kinetic energy on frequency. The graph helps students interpret how Planck’s constant and the work function can be obtained experimentally. It also reinforces that intensity changes current, not the gradient of the graph. Source
HL only: Matter waves and particle diffraction
Diffraction of particles is evidence for the wave nature of matter.
Matter exhibits wave–particle duality: particles can show wave behaviour, and light can show particle behaviour.
de Broglie wavelength: .
For non-relativistic particles, , so .
Larger momentum means smaller wavelength and therefore less noticeable diffraction.
Electrons, neutrons and other particles can diffract if their wavelength is comparable to atomic spacing.
In a diffraction/scattering experiment, minima and maxima are predicted using the particle’s de Broglie wavelength.
Exam idea: if a particle beam gives a diffraction pattern, it is behaving as a wave.
Link to prior wave topic: use diffraction/interference conditions exactly as for waves once is known.

This diagram shows the Davisson–Germer setup, where an electron beam is directed at a crystal and the scattered intensity is measured at different angles. It is a classic illustration of particle diffraction and supports the wave nature of matter. Use it to connect crystal spacing with de Broglie wavelength. Source

This photograph shows concentric diffraction rings produced by electrons. The ring pattern is direct evidence that electrons can behave like waves and undergo diffraction. It is especially useful for recognizing experimental evidence rather than just theory. Source
HL only: Compton scattering
Compton scattering is additional evidence for the particle nature of light.
A photon collides with an electron, transfers some energy and momentum, and is scattered.
After scattering, the photon has a longer wavelength and therefore lower energy.
The wavelength shift is:
Here, is the scattering angle, is the electron mass, and is the speed of light.
If , then .
Maximum shift occurs at , so .
The fact that wavelength changes by a definite amount after a collision supports the idea that light carries momentum and behaves like particles.
You do not need to derive the Compton formula.
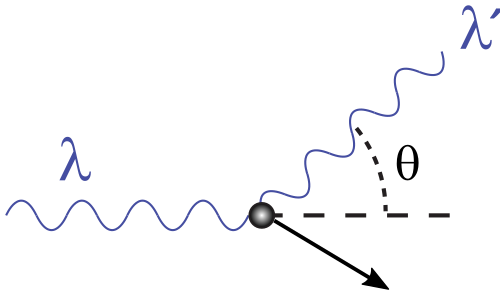
This schematic shows an incoming photon scattering from a target electron and leaving in a new direction. It helps visualize why the scattered photon has reduced energy and increased wavelength. The diagram is ideal for linking the scattering angle to wavelength shift in the Compton equation. Source
Wave–particle duality: what each experiment proves
Photoelectric effect light behaves as particles (photons).
Compton scattering light behaves as particles with energy and momentum.
Electron/particle diffraction matter behaves as waves.
The best conclusion is not “light is only a particle” or “matter is only a wave”.
Correct conclusion: quantum objects show wave–particle duality; the observed behaviour depends on the experiment.
Be ready to state which experiment gives which evidence.
Exam focus and common traps
Do not say brighter light ejects electrons if ; frequency must exceed threshold.
Do not confuse intensity with photon energy: photon energy depends on frequency.
Do not say de Broglie wavelength depends on charge; it depends on momentum.
Do not forget that a longer wavelength means a lower photon energy because .
Do not claim Compton scattering is wave evidence; it is particle evidence for light.
In explanations, always link observation to model: threshold/instant emission photons; diffraction pattern wave behaviour.
Checklist: can you do this?
Explain why the photoelectric effect cannot be explained by classical wave theory.
Use and, where needed, .
Calculate a de Broglie wavelength from a particle’s momentum.
Interpret diffraction/scattering evidence as support for the wave nature of matter.
Use and interpret the Compton shift equation and state what it shows about light.