AP Syllabus focus: ‘Apply the octet rule and formal charge to decide which valid Lewis diagram best predicts molecular structure and properties when multiple nonequivalent structures are possible.’
Choosing among multiple valid Lewis diagrams is a model-selection task. You use the octet rule to check electron accounting, then use formal charge to decide which structure best represents bonding and likely properties.
Core ideas: octet rule + formal charge
Octet rule as a screening tool
Octet rule: main-group atoms tend to form bonds so each atom has 8 electrons in its valence shell (counting shared electrons in bonds).
When multiple nonequivalent Lewis diagrams are possible, first verify each candidate is internally consistent:
Lewis-style bonding diagrams for and methane, , illustrating how shared electron pairs (bonds) contribute to each atom’s stable electron count. This kind of picture helps you “screen” candidate Lewis diagrams by checking whether the bonding pattern can satisfy typical valence-shell targets (duet for H, octet for C). It directly supports the idea that octet/valence satisfaction is a first-pass filter before comparing formal charges. Source
Correct total valence electron count (including charge)
Acceptable electron placement (bonding pairs and lone pairs)
Octets on relevant atoms (especially second-period atoms like C, N, O, F, which are strongly constrained)
A diagram can be “valid” (electron-counting works) yet still be a poor model if it gives unreasonable charge separation or violates the octet rule for atoms that rarely do.
Formal charge as the selection criterion
Formal charge: a bookkeeping charge assigned to an atom in a Lewis diagram assuming equal sharing of bonding electrons.
Use formal charge to compare candidate diagrams that satisfy electron counting (and, when expected, the octet rule). It helps identify which model most plausibly represents electron distribution.
A key constraint is that the sum of all formal charges equals the overall charge on the species; if it does not, the diagram is not valid.
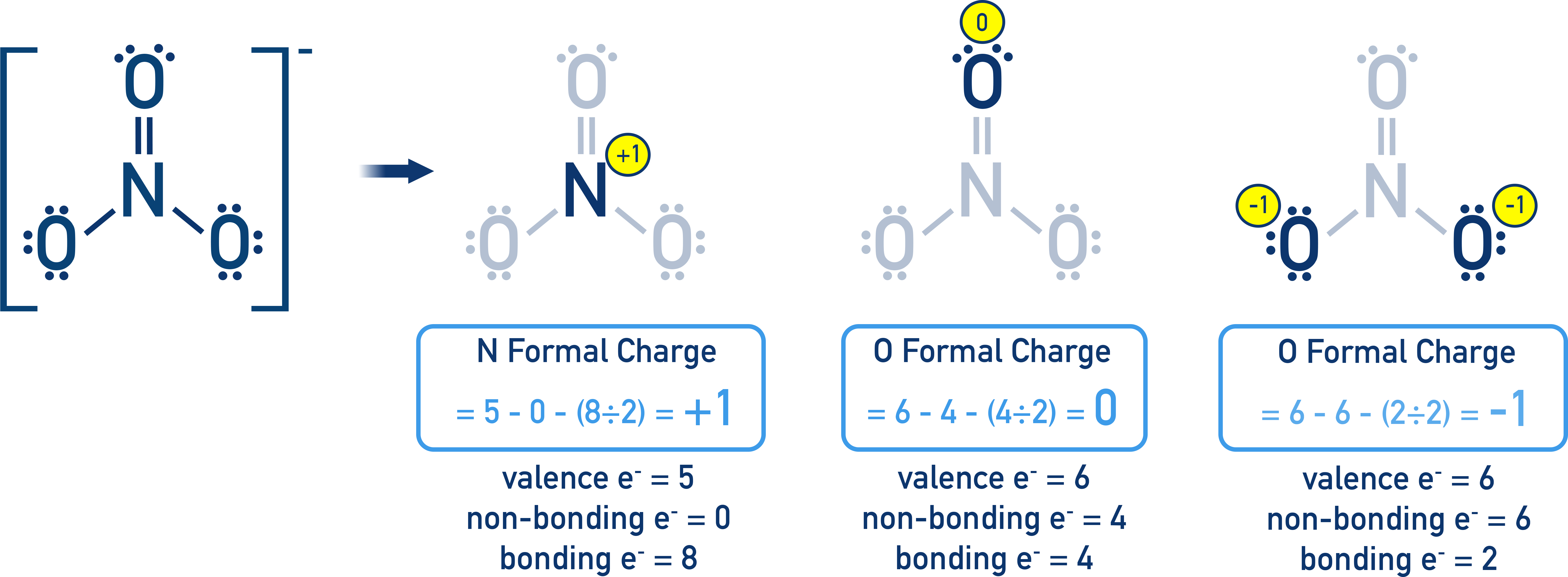
Formal-charge bookkeeping for the nitrate ion, , with each atom’s valence, nonbonding, and bonding electrons shown and the resulting formal charges labeled. The diagram highlights how formal charges distribute across nonequivalent atoms (single-bonded vs double-bonded oxygen) while still summing to the ion’s overall charge. This is a typical example of using formal charge to evaluate whether a Lewis diagram is both valid and chemically reasonable. Source
= valence electrons of the free atom (from periodic group number)
= nonbonding (lone-pair) electrons on that atom in the diagram
= bonding electrons shared in bonds to that atom (2 per single bond, 4 per double bond, etc.)
Formal charge is not the same as partial charge, but diagrams with more reasonable formal charges usually better predict connectivity, bond order placement, and qualitative polarity.
How to choose the “best” Lewis diagram
A practical decision hierarchy
When you have multiple nonequivalent valid Lewis diagrams, prefer the one that best satisfies these ideas (roughly in this order):
Octets satisfied for atoms that follow the octet rule strongly (especially second period)
Small formal charges on atoms (minimise magnitude of FC across the structure)
Least charge separation (avoid placing + and − charges on adjacent atoms unless unavoidable)
Negative formal charge on the more electronegative atom, and positive on the less electronegative atom, when charges must appear
Appropriate overall charge distribution for ions (e.g., anions often place negative FC on electronegative atoms like O, N, halogens)
Interpreting what “best predicts structure and properties” means
The preferred Lewis diagram is the one most likely to correspond to:
More realistic bonding patterns (where multiple bonds are placed)
More plausible bond order assignments (single vs double vs triple placement)
More chemically reasonable reactive sites (atoms bearing significant formal charge)
Better qualitative predictions of polarity and intermolecular interactions (via charge placement trends)
Formal charge comparisons are model-based: they do not measure energy directly, but they correlate with stability and plausibility for many main-group molecules and polyatomic ions.
Common pitfalls to avoid
Mixing up overall charge with a single atom’s formal charge; always check the sum of FCs.
Declaring a diagram “best” solely because it has all octets, while ignoring extreme formal charges.
Forgetting that the “best” model is chosen among valid diagrams; an invalid electron count or impossible octet arrangement disqualifies a candidate immediately.
Treating formal charge as the same as real electron distribution; it is a selection tool, not an experimental charge measurement.
FAQ
Formal charge is a bookkeeping tool based on equal sharing, whereas partial charges come from unequal sharing (polarity) and electron density.
A structure with “better” formal charges often aligns with expected partial charge trends, but they need not match numerically.
Some species cannot be drawn without charges while maintaining correct electron counts and octets.
In such cases, choose the diagram with the smallest charge magnitudes and place $-$ on more electronegative atoms.
More electronegative atoms stabilise extra electron density more effectively.
So, among valid options, diagrams with $-$ on atoms like O, F, Cl are typically more chemically reasonable than $-$ on C or B.
After assigning FC to each atom, add them.
The sum must equal the overall charge (e.g., $-1$ for an anion). If not, the diagram is not a valid Lewis model.
Yes, occasionally.
Then prioritise octet satisfaction (especially for second-period atoms) and the placement of multiple bonds to avoid unlikely patterns (e.g., excessive bonding on atoms that typically form fewer bonds).
Practice Questions
(3 marks) Two nonequivalent Lewis diagrams are proposed for :
A: \ce{^-C#N-O}
B:
Calculate the formal charge on each atom in A and B, and state which diagram is preferred using formal charge and electronegativity.
1 mark: Correct FCs for A: C = , N = , O = .
1 mark: Correct FCs for B: C = , N = , O = .
1 mark: B preferred because it minimises placing negative charge on less electronegative C; negative FC on O (more electronegative) is more reasonable.
(6 marks) Three nonequivalent Lewis diagrams are proposed for :
A: \ce{N#N-O}
B:
C: \ce{N-N#O}
For each diagram, determine the formal charge on each atom and identify the best model using the octet rule and formal charge criteria.
1 mark: A FCs: first N = , central N = , O = .
1 mark: B FCs: first N = , central N = , O = .
1 mark: C FCs: first N = , central N = , O = .
1 mark: Identifies A as best (smallest magnitudes and least unreasonable separation).
1 mark: Uses electronegativity argument: negative FC on O is favourable.
1 mark: Notes octets are satisfied in A and B, but C has highly unfavourable FC pattern.

