AP Syllabus focus: ‘Recognize limitations of Lewis structure models, especially for species with an odd number of valence electrons; interpret what the model can and cannot represent.’
Lewis structures are essential electron-bookkeeping models, but they are simplified. AP Chemistry expects you to recognise when a Lewis diagram is an adequate representation and when it necessarily fails, especially for odd-electron species.
What Lewis structures can and cannot represent
What the model represents
A Lewis structure is a 2-D sketch showing:
Atom connectivity (which atoms are bonded)
Valence electrons placed as bonding pairs and lone pairs
This model is best at tracking valence-electron counts and proposing plausible bonding patterns.
Core limitations of Lewis structures
Lewis structures can be “valid” yet still incomplete or misleading because they do not directly represent:
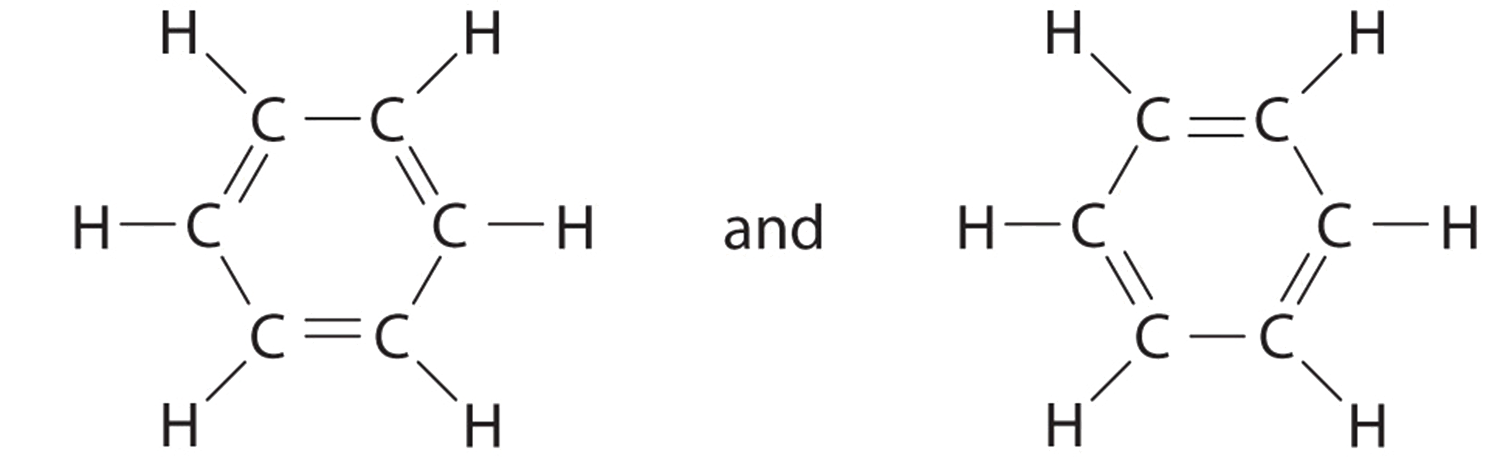
Benzene resonance representations, contrasting alternating single/double-bond Lewis drawings with the common “circle in a hexagon” shorthand for delocalized electrons. This diagram reinforces that a single Lewis structure can imply unequal bond types even when all C–C bonds are experimentally equivalent. Source
Three-dimensional geometry (bond angles and spatial arrangement are not encoded in the drawing)
Orbital overlap and bond strength (a line does not show how effective overlap is)
Electron delocalisation (electrons spread over multiple atoms may be forced into a single-bond/double-bond choice)
Real bond order and bond length trends in delocalised systems (a single drawing can imply unequal bonds where experiments show equivalence)
Magnetic properties (paired vs unpaired electrons) unless you explicitly track whether an electron is unpaired
When you are asked what a Lewis model “can and cannot represent,” focus on these mismatches between the sketch and real electron behaviour.
Odd-electron species (radicals) as a key Lewis-model limit
Why odd-electron counts break the usual pattern
Many students implicitly assume “all electrons pair up into bonds or lone pairs.”
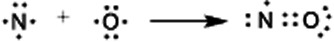
Lewis dot depiction of nitric oxide, NO, highlighting that one electron remains unpaired (a radical) even after assigning bonding pairs and lone pairs. This is a concrete example of why an odd total valence-electron count cannot be fully paired in a Lewis picture. Source
That assumption fails for species with an odd number of total valence electrons, because pairing all electrons is impossible.
Radical (odd-electron species): a species containing at least one unpaired valence electron, often arising from an overall odd total valence-electron count.
Odd-electron species highlight a limitation: the Lewis model can place the unpaired electron, but it does not explain why that placement is energetically preferred, nor does it capture how that unpaired electron may be distributed over multiple atoms.
How to draw and interpret radicals in Lewis form
When constructing a Lewis diagram for an odd-electron species:
Count total valence electrons carefully (including charge)
Build a reasonable skeleton and add electrons to satisfy octets where possible
Accept that one atom will have an unpaired electron and may have an incomplete octet
Use a single dot to show the unpaired electron (not a pair)
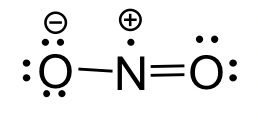
Lewis structure of nitrogen dioxide, NO, showing one unpaired electron as a single dot and the resulting octet-rule exception. This example is useful for practicing electron counting and for recognizing when a Lewis diagram must include an unpaired electron rather than forcing full pairing. Source
Interpretation expectations on AP-style questions typically include:
Identifying that the species must contain an unpaired electron
Recognising that a central atom may not achieve a full octet in that drawing
Stating that the Lewis picture is a simplified representation of electron distribution
What Lewis cannot tell you about radical behaviour
A Lewis structure with one dot does not, by itself, provide:
The stability of the radical (some radicals are much more stabilised than others)
Whether the unpaired electron is localised on one atom or delocalised across several atoms
The actual set of equivalent bonding arrangements that can exist in the real molecule/ion
So, for radicals, Lewis structures are best treated as electron-accounting sketches rather than literal pictures of where the unpaired electron “sits” at all times.
Other warning signs that the Lewis model is strained
Even when the electron count is even, Lewis structures can signal their own limits when you encounter:
Incomplete octets (particularly for some electron-deficient central atoms)
Seemingly competing “reasonable” drawings that predict different bond patterns (indicating the drawing is not uniquely determined by simple rules)
In these cases, the correct AP-level move is to acknowledge the model limitation and avoid over-claiming precise bonding details from a single sketch.
Using Lewis structures responsibly on the AP Exam
Keep Lewis structures in their proper role:
Use them to count valence electrons and propose connectivity
Use them to identify lone pairs and the possibility of unpaired electrons
Do not claim that a single Lewis diagram uniquely determines true bond equivalence, electron distribution, or magnetic behaviour without additional reasoning
FAQ
Use electron counting and octet completion to find plausible placements, then choose a structure that avoids extreme charge separation.
When multiple placements seem plausible, the Lewis model alone may be insufficient; the best answer is often to acknowledge ambiguity.
Sometimes yes (the unpaired electron can be on an atom that still has eight electrons around it), but many common radicals necessarily leave one atom short of an octet.
Odd-electron species can force compromises between octets and charge separation.
If charge placement seems contradictory, that often signals the Lewis diagram is an imperfect model rather than the species being impossible.
Paramagnetism (attraction to a magnetic field) indicates at least one unpaired electron.
This connects to radicals, but magnetism itself is not directly encoded unless you track electron pairing.
A single drawing can localise bonding, implying unequal bonds.
In reality, electron density (including an unpaired electron) may be spread over more than one bond, producing averaged, more similar bond lengths.
Practice Questions
(1–3 marks) Nitric oxide, NO, has 11 valence electrons. (a) Draw a Lewis structure for NO showing all valence electrons. (b) State one limitation of this Lewis structure model for NO.
Correct total of 11 valence electrons shown (1)
One unpaired electron correctly indicated (1) (b)
One valid limitation stated (e.g., does not show the true distribution/delocalisation of the unpaired electron, or is a 2-D bookkeeping model) (1)
(4–6 marks) Consider NO and NO. Both are based on N and O atoms. (a) Determine the total number of valence electrons in NO and in NO. (b) Explain, using Lewis-structure reasoning, why NO is an odd-electron species but NO is not. (c) Give one reason a single Lewis structure can be misleading for bonding in NO.
NO: 17 valence electrons (1)
NO: 18 valence electrons (1) (b)
NO has an odd total, so it must contain an unpaired electron (1)
NO has an even total, so electrons can all be paired in bonds/lone pairs (1) (c)
One valid reason (e.g., Lewis structures may force one N–O single and one N–O double even if electron density/bonding is not confined to one arrangement) (1)

