AP Syllabus focus: ‘Beer–Lambert law relates absorbance to concentration, path length, and molar absorptivity: A = εbc; ε reflects how strongly a species absorbs light at a specific wavelength.’
Beer–Lambert law is the core quantitative relationship used in spectrophotometry to connect what an instrument measures (light absorption) to how much absorbing species is present in solution, under carefully controlled conditions.
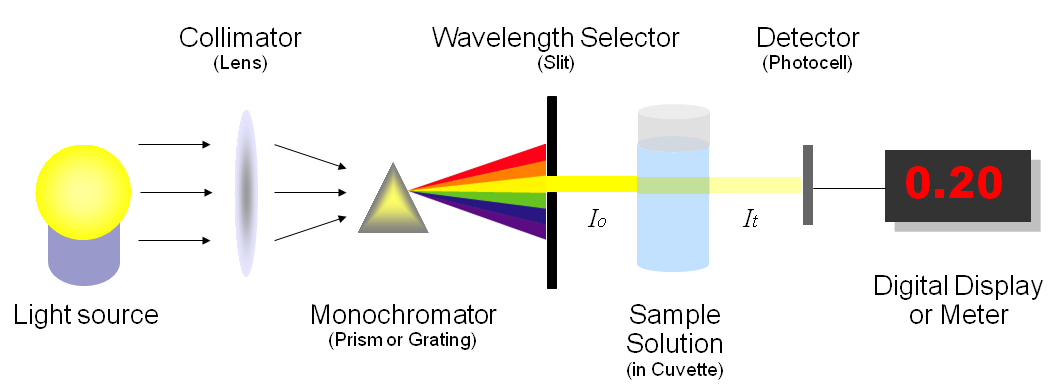
Schematic layout of a UV–Vis spectrophotometer showing the optical path from the light source through wavelength selection (monochromator/slits) to the sample cuvette and detector. This helps connect the measured signal (transmitted intensity) to absorbance as an instrument-generated quantity used for quantitative analysis. Source
Beer–Lambert Law: What It Connects
Beer–Lambert law links a macroscopic measurement, absorbance, to three factors you can control or interpret in the lab: concentration, path length, and molar absorptivity. It applies to a specific species at a specific wavelength of light.
Absorbance (A)
Absorbance (A): A unitless measure of how much light is absorbed by a sample; higher absorbance means less light is transmitted through the sample.
Absorbance is reported by spectrophotometers and is designed to scale linearly with “how many absorbers” are in the light path, making it useful for determining unknown concentrations.
Concentration (c)
Concentration (c): The amount of the absorbing species per unit volume of solution, most commonly expressed in mol/L (M) in AP Chemistry.
Only the concentration of the absorbing particles (molecules or ions that interact with the selected wavelength) belongs in Beer–Lambert law.
Path length (b)
Path length (b): The distance light travels through the sample, usually the cuvette width, commonly 1.00 cm.
A longer path length gives light more opportunity to be absorbed, increasing the measured absorbance for the same solution.
Molar absorptivity (ε)
Molar absorptivity (ε): A proportionality constant that describes how strongly a given species absorbs light at a particular wavelength; units are typically L·mol⁻¹·cm⁻¹ when is in mol/L and is in cm.
ε depends on both identity and wavelength: changing the wavelength changes ε, even for the same chemical species. This is why Beer–Lambert measurements must specify the wavelength used.
The Beer–Lambert Equation and Variable Meanings
The relationship is written with absorbance as the measured outcome and the other terms as causes of stronger or weaker absorption.
= absorbance (unitless)
= molar absorptivity at a specific wavelength (commonly L·mol⁻¹·cm⁻¹)
= path length through the sample (commonly cm)
= concentration of the absorbing species (commonly mol/L)
Because is proportional to each factor, increasing or increases proportionally (holding the other variables constant). The constant sets the overall sensitivity: a large means even small concentrations can give measurable absorbance.
Interpreting ε: “How Strongly a Species Absorbs”
In the syllabus language, ε reflects how strongly a species absorbs light at a specific wavelength. Practically:
If two species are at the same and measured with the same and wavelength, the one with larger ε produces larger A.
If the same species is measured at two different wavelengths, ε may change dramatically, so A changes even if and do not.
Conditions for Linearity (When Beer–Lambert Works Best)
Beer–Lambert law is used as a linear model under typical spectrophotometry conditions. Linearity is most reliable when:
The light is effectively monochromatic (one selected wavelength)
The solution is reasonably dilute (so particles act independently)
The sample is clear (minimal scattering from bubbles, precipitate, or fingerprints)
The chemical form of the absorber is stable (no reaction or association changing the absorbing species during measurement)
These conditions support treating as constant for that species at that wavelength, which is the key assumption behind using as a direct proxy for .
FAQ
Absorbance is defined using a ratio of intensities (transmitted vs incident), so the units cancel.
Because it is logarithmic in origin, it is reported without units even though it reflects a physical measurement.
$\varepsilon$’s units must change to keep $A$ unitless.
If $c$ is in mol/m$^3$, $\varepsilon$ must incorporate m$^3$ instead of L.
Always ensure $\varepsilon bc$ is dimensionless.
Sometimes, but only with careful controls.
You would need:
the exact wavelength,
the solvent and temperature conditions,
confidence that the absorbing chemical form is the same as the reference.
At higher concentrations, particles can interact (association, electrostatic effects) and the solution’s refractive/scattering behaviour can change.
These effects make $\varepsilon$ effectively non-constant, breaking the linear link between $A$ and $c$.
Measure $A$ for a set of standard solutions at known $c$, using a fixed $b$ and wavelength.
The proportionality between $A$ and $c$ allows $\varepsilon$ to be obtained from the gradient divided by $b$ (within the linear range).
Practice Questions
Question 1 (2 marks) State the Beer–Lambert law and identify what each symbol represents, including a typical unit for .
1 mark: Correct equation: .
1 mark: Correct identification of symbols (any three correct earns the mark) and unit for (e.g. L·mol·cm).
Question 2 (5 marks) A student measures the absorbance of a solution containing one absorbing species at a fixed wavelength using a 1.00 cm cuvette. (a) Describe how the absorbance changes if the concentration is doubled. (1 mark) (b) Describe how the absorbance changes if the path length is increased from 1.00 cm to 2.00 cm. (1 mark) (c) Explain what information is contained in and why the wavelength must be specified when quoting . (3 marks)
1 mark: Absorbance doubles (direct proportionality to ). (b)
1 mark: Absorbance doubles (direct proportionality to ). (c)
1 mark: indicates how strongly the species absorbs light (intrinsic sensitivity).
1 mark: depends on the identity of the species.
1 mark: depends on wavelength / must specify wavelength because absorption varies with wavelength.

