AP Syllabus focus: ‘When path length and wavelength are held constant, absorbance is proportional only to the concentration of absorbing molecules or ions in the light path.’
Absorbance measurements connect what a detector sees (light intensity) to how many absorbing particles are in solution. The key idea is that, under controlled conditions, more absorbers in the beam path means more light absorbed.
Core idea: more absorbers → more absorption events
In a spectrophotometer, a beam of (nearly) single-wavelength light passes through a solution.
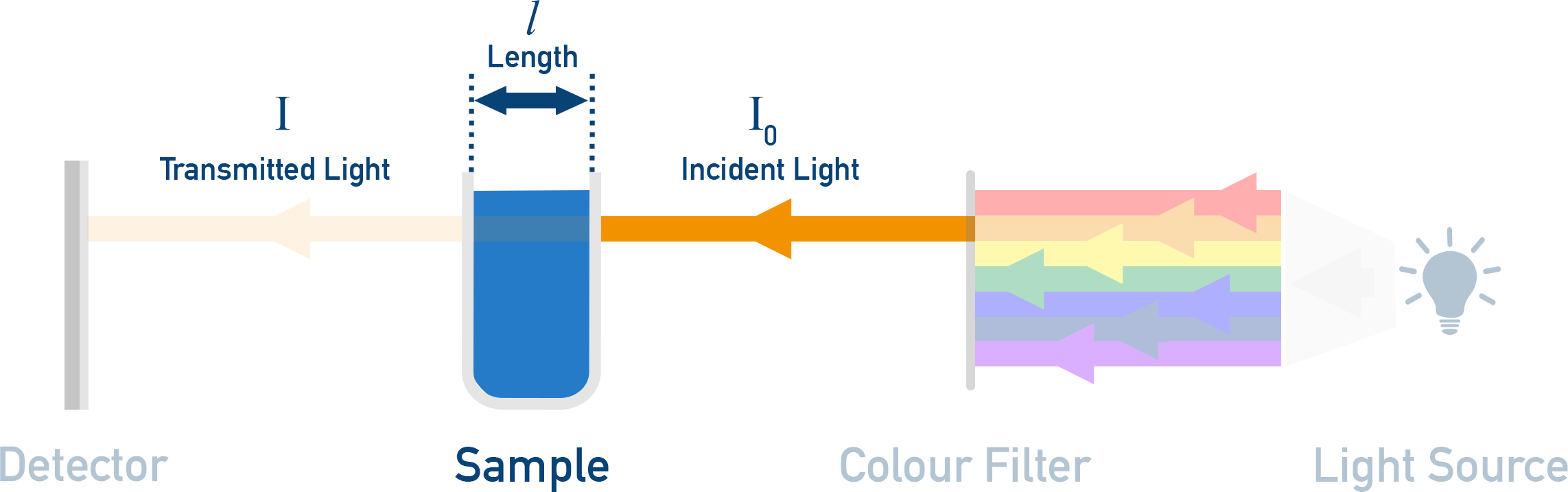
Simplified spectrophotometer schematic showing incident light passing through a sample of fixed path length and emerging as transmitted light at the detector. The color filter represents selecting a (nearly) single wavelength so that absorbance changes can be attributed to concentration rather than wavelength. Labels reinforce the experimental controls (wavelength, path length) that underlie Beer–Lambert proportionality. Source
Particles that can absorb that wavelength remove photons from the beam.
If concentration increases, there are more absorbing molecules/ions per unit volume.
For a fixed beam cross-section, that means more absorbers in the light path at any moment.
More absorbers leads to a higher probability that photons are absorbed as they travel through the sample, so the transmitted intensity decreases and the measured absorbance increases.
Absorbance as the measured quantity
Absorbance (A): A logarithmic measure of light attenuation by a sample; higher absorbance corresponds to less transmitted light at the selected wavelength.
Using absorbance (rather than raw transmitted intensity) is useful because it makes the relationship with concentration linear under typical AP Chemistry conditions (dilute solutions, well-behaved optics).
Why holding wavelength and path length constant matters
The proportionality in the syllabus statement depends on controlling two experimental factors:
Wavelength held constant: a species’ ability to absorb light depends strongly on wavelength. If wavelength changes, the “strength” of absorption changes even at the same concentration.
Path length held constant: a longer path means photons encounter more absorbing particles overall, even if concentration is unchanged. Standard cuvettes commonly keep path length fixed so concentration is the only changing factor.
Beer–Lambert relationship (the proportionality statement in equation form)
= absorbance (unitless)
= path length through the sample (cm)
= concentration of absorbing species (mol·L)
= molar absorptivity at the chosen wavelength (L·mol·cm)
When and the wavelength are held constant, is constant for that species at that wavelength. The equation then reduces conceptually to , matching the syllabus requirement: absorbance depends only on concentration of absorbers in the light path.
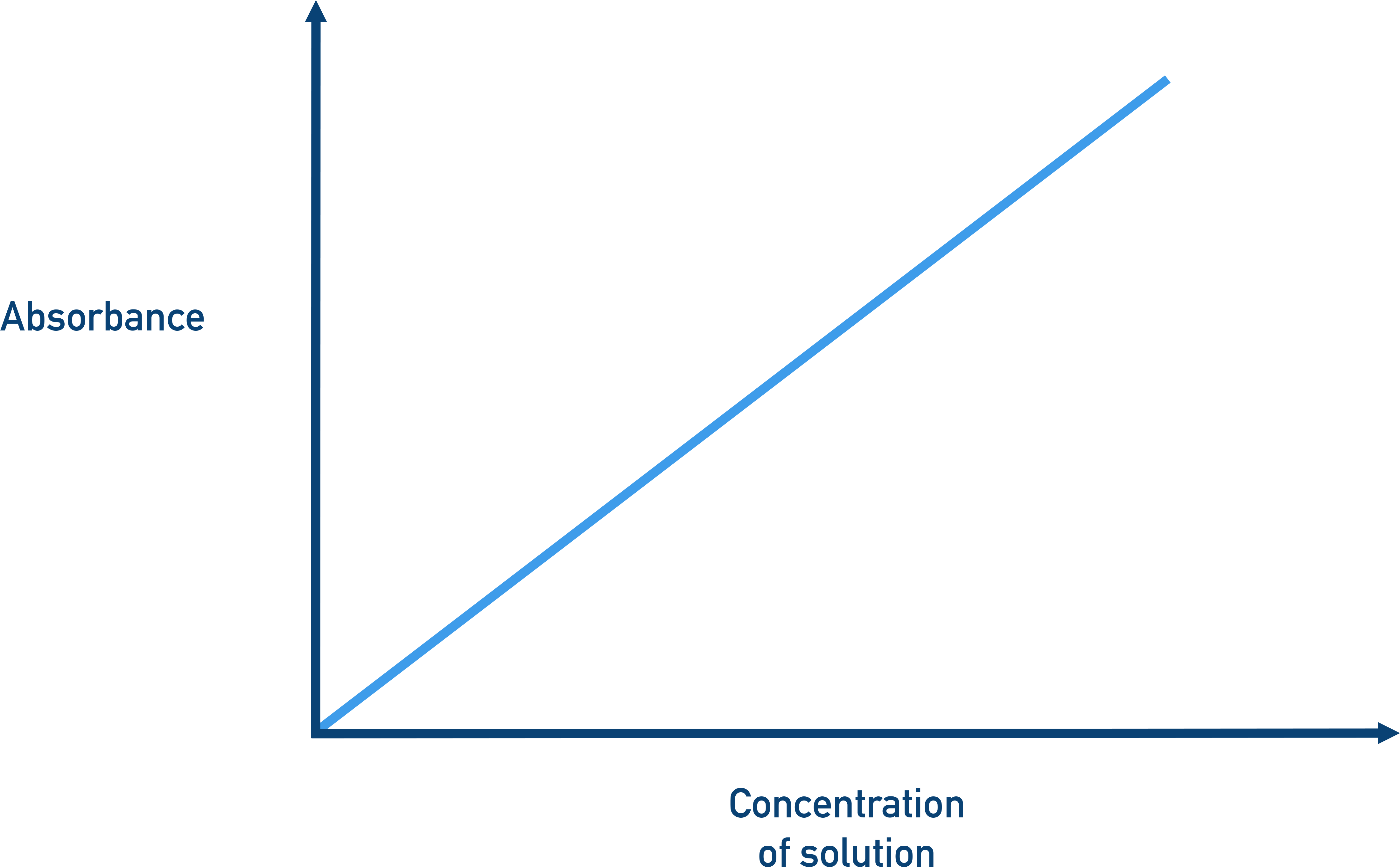
Graph of absorbance versus concentration illustrating the linear trend predicted by Beer–Lambert law under constant path length and wavelength. The straight line emphasizes that absorbance behaves as a proportional response variable when and are fixed. This is the basis for calibration curves used to determine an unknown concentration from a measured absorbance. Source
Particle-level explanation of linearity
A helpful way to justify proportionality without heavy math is to think in “thin layers” of solution:
Divide the cuvette into many extremely thin slices along the beam direction.
Each slice contains a number of absorbers proportional to concentration.
Each slice removes a small fraction of the remaining light.
Increasing concentration increases the absorber count in every slice, so each slice removes more light in the same consistent way across the path.
Because absorbance is defined on a logarithmic scale, these repeated fractional losses across slices translate into a linear dependence of on for solutions that behave ideally in the measurement.
Conditions implicit in “absorbance is proportional to concentration”
The statement applies best when the sample and measurement setup ensure that only absorption (not other effects) is changing:
The absorbing species must remain chemically the same as concentration changes (no significant association, dissociation, or reaction shifting the absorbing form).
The solution should be sufficiently dilute that optical and molecular interactions do not distort the linear relationship.
The sample should be uniform so the “concentration in the light path” is well-defined.
FAQ
A logarithmic scale turns multiplicative light losses along the path into an additive quantity.
This often produces a straight-line relationship with concentration over a useful range.
It depends on how strongly a species interacts with light at a particular wavelength.
Electronic structure and transition probability largely control its magnitude.
At high concentrations, particles can interact or the refractive index can change noticeably.
These effects can alter how light propagates, reducing linearity.
Stray light increases detected intensity even when the sample absorbs strongly.
This can make absorbance readings appear too low, especially at higher true absorbance.
The detector measures combined attenuation from all absorbers present.
In many cases, total absorbance can be treated as the sum of individual contributions if they do not interact chemically.
Practice Questions
(2 marks) In a colorimeter experiment, the cuvette path length and the wavelength are kept constant. Explain why the measured absorbance increases when the concentration of the absorbing ion increases.
More absorbing ions per unit volume means more absorbers in the beam path / higher probability of photon absorption. (1)
Therefore transmitted intensity decreases and absorbance increases (consistent with Beer–Lambert proportionality). (1)
(5 marks) A student claims that doubling the concentration of an absorbing dye will always double the absorbance reading. Using the idea that absorbance is proportional to concentration when path length and wavelength are held constant, assess this claim and state the assumptions required for it to be valid.
States that with constant path length and constant wavelength, so doubling doubles (Beer–Lambert behaviour). (1)
Mentions must be constant at that wavelength (same absorbing species/form). (1)
Notes solution must be sufficiently dilute / within the linear range. (1)
Notes measurement must reflect absorption rather than other light losses (e.g. no significant scattering/turbidity). (1)
Notes concentration in the light path must be uniform (well-mixed, consistent cuvette filling). (1)

