AP Syllabus focus: ‘Represent differences among solid, liquid, and gas phases using particulate-level models that connect particle arrangement, spacing, and motion to macroscopic behavior.’
Understanding phases is easiest when you picture matter as particles in motion.
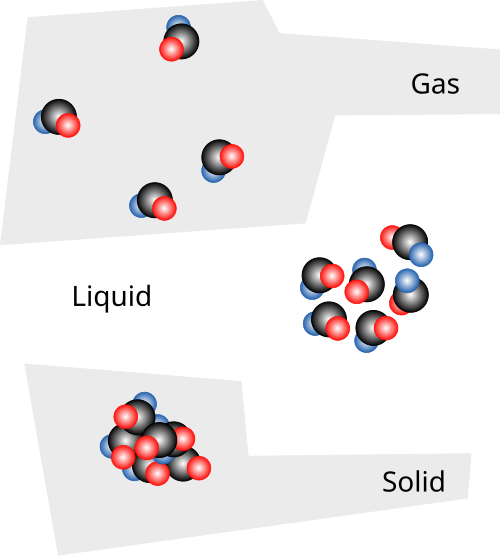
Particle-level sketches compare a solid, liquid, and gas by showing how particle spacing and structural order change across phases. This kind of diagram supports qualitative explanations of macroscopic properties (e.g., density and compressibility) by making the “empty space” and packing differences visible. Source
Particulate models link what you can’t see (arrangement, spacing, motion) to what you can measure (shape, volume, density, compressibility, and flow).
Particulate models: what they show and why they matter
Particulate-level model: a representation of matter as discrete particles whose spacing, arrangement, and motion explain observable (macroscopic) properties.
A good particulate model focuses on three linked features:
Arrangement: whether particles have an ordered pattern or not
Spacing: average distance between particles (close-packed vs far apart)
Motion: how particles move (vibrations, sliding past, random translation)
These microscopic features are used to explain macroscopic behaviors such as:
Shape and volume (definite vs variable)
Compressibility (ability to decrease volume under pressure)
Fluidity (ability to flow)
Density (mass per volume, explained qualitatively by packing)
Solids: tightly packed, fixed positions, vibration
Solid: a phase in which particles are in close contact and maintain fixed average positions; particles primarily vibrate rather than translate through the sample.
In a particulate diagram of a solid, you should expect:
Particles drawn very close together with minimal empty space
A regular, repeating pattern in many solids (especially crystalline solids), reflecting long-range order
Motion indicated as small vibrations about fixed positions (often shown with short “wiggle” marks)
Macroscopic properties explained by this model:
Definite shape and definite volume: fixed particle positions and strong particle–particle contacts prevent bulk flow.
Very low compressibility: because there is little empty space to remove; pushing particles closer requires overcoming strong repulsions at short distances.
High rigidity: small particle displacements are resisted because changing positions disrupts many close contacts at once.
When temperature increases (still remaining solid), particulate models show increased vibration amplitude, which helps explain thermal expansion and why solids soften as melting is approached.
Liquids: close contact, disordered arrangement, sliding motion
Liquid: a phase in which particles remain in close contact but continuously move and reorganise; particles can slide past one another while maintaining cohesion.
A particulate diagram of a liquid typically shows:
Particles close together, but with a less regular arrangement than a solid
Small, transient gaps as particles shift positions
Motion indicated by short arrows showing continuous movement and collisions
Macroscopic properties explained by this model:
Definite volume: close contact and attractions keep particles together, resisting separation.
Indefinite shape: because particles can rearrange, the sample flows to match the container.
Low compressibility (but slightly more than solids): there is still limited free space; modest compression is possible by reducing temporary gaps.
Fluidity and viscosity trends (qualitative): easier sliding corresponds to lower resistance to flow; restricted sliding corresponds to thicker, more viscous liquids.
Heating a liquid is represented by faster particle motion and more frequent rearrangements. This supports observations like faster diffusion/mixing and a greater tendency for particles to separate into a gas phase at higher temperatures.
Gases: far apart, random motion, frequent collisions
Gas: a phase in which particles are far apart relative to their size and move in rapid, random, straight-line motion between collisions, so the sample fills its container.
A particulate diagram of a gas should show:
Particles widely spaced, with large areas of empty space
No fixed arrangement (completely disordered)
Motion indicated by long arrows in random directions, emphasising continuous translation
Macroscopic properties explained by this model:
No definite shape and no definite volume: particles move independently and spread out to occupy all available space.
High compressibility: because most of the volume is empty space that can be reduced by pushing particles closer.
Low density compared with solids and liquids: fewer particles in a given volume because of large spacing.
Pressure (qualitative): collisions of gas particles with container walls produce pressure; more frequent or more forceful collisions correspond to higher pressure.
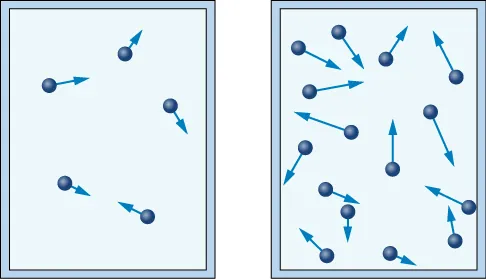
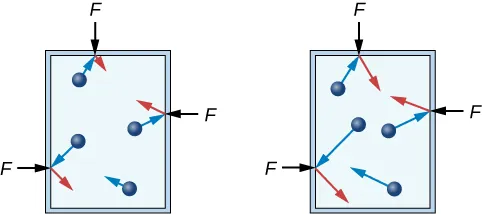
A particle-in-a-container model shows gas particles moving in straight-line paths and colliding with the container walls. The accompanying panels illustrate how changing particle number or particle speed increases collision frequency/impulse, supporting the particulate explanation of why gas pressure rises under those conditions. Source
Using particulate models to justify phase differences
When asked to “represent differences” among phases, your model should explicitly connect:
Close contact (solid/liquid) vs large spacing (gas) to compressibility and density
Fixed positions (solid) vs mobile rearrangement (liquid/gas) to shape and flow
Type of motion (vibration vs sliding vs free translation) to rigidity vs fluidity and how properties change with temperature
In AP-style explanations, the strongest responses state both:
what the particles are doing (arrangement/spacing/motion), and
what that implies for the observed property (shape/volume/compressibility/flow).
FAQ
Use conventions: short wiggles for vibration (solid), short arrows in many directions for sliding/rearrangement (liquid), and long arrows for free translation (gas).
A good diagram also implies relative speed by arrow length and randomness by varied directions.
Drawing gas particles too close together (looks like a liquid).
Using a perfectly regular lattice for a liquid (looks like a crystalline solid).
Changing particle size between phases (same substance should have same particles).
Forgetting to show large empty space in gases.
It is simply the region not occupied by the particles themselves.
In gases this space dominates the volume, which is why gases compress easily; in liquids/solids the unoccupied space is small and transient.
Yes. “Particles” can mean atoms, molecules, or ions, depending on the substance.
For this subtopic, the key is not particle identity but how those particles are spaced, arranged, and moving in each phase.
At the surface, some particles have enough energy and the right direction of motion to escape the liquid into the gas region, while others re-enter the liquid.
This dynamic exchange can be shown by a few particles leaving the liquid surface in the particulate model.
Practice Questions
(2 marks) Describe, using a particulate-level model, one key difference between a liquid and a gas that explains why gases are much more compressible.
States that gas particles are much farther apart / have much more empty space between them (1)
Links this to compressibility: volume can decrease a lot because empty space is reduced before particles are forced close together (1)
(5 marks) A student compares three sealed containers holding the same substance as a solid, liquid, and gas (one phase per container). Using particulate models, explain the differences in (i) shape, (ii) volume, and (iii) ability to flow.
Solid: particles closely packed with fixed average positions; only vibrate (1)
Liquid: particles close together but disordered and able to move/slide past one another (1)
Gas: particles far apart with rapid random motion (1)
Links particle behaviour to shape/flow: solid keeps shape (no rearrangement) whereas liquid and gas take container shape (rearrangement/translation) (1)
Links particle spacing to volume: solid and liquid have (approximately) fixed volume due to close contact; gas fills container so volume equals container volume because particles spread out (1)

