AP Syllabus focus: ‘A solid’s structure is influenced by interparticle interactions and how well particles can pack together, affecting orderliness and rigidity at the macroscopic scale.’
Solids are not just “frozen liquids.” Their macroscopic properties depend on how particles arrange in space and how strongly they attract at the distances imposed by that arrangement.
Packing and interactions: the central link
In a solid, particles (atoms, ions, or molecules) are held in fixed positions relative to one another while still undergoing small vibrations. Two ideas work together to determine structure:
Interparticle interactions set the preferred spacing and orientation between neighbors.
Packing describes how efficiently particles can arrange to maximize attractions (and minimize repulsions) throughout the solid.
What “packing” means in AP Chemistry terms
Packing efficiency: How closely particles in a solid can fit together (high efficiency = little empty space), determined by particle shape/size and how well those particles can arrange in a repeating pattern.
Packing is not only about “space”; it also controls how many close neighbors each particle has, which changes the total stabilizing attraction per particle.
How interparticle interactions influence solid structure
Interparticle forces are distance-dependent: attractions dominate at moderate separations, while very short separations produce strong repulsions (electron cloud overlap).
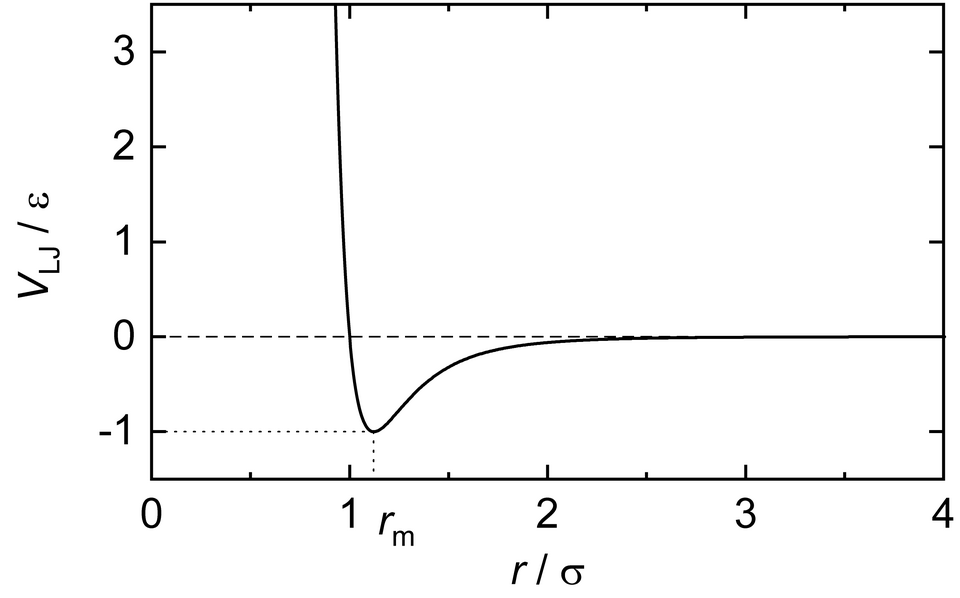
Lennard–Jones potential energy curve showing how interaction energy varies with particle separation. The steep rise at short distance represents strong repulsion from electron-cloud overlap, while the negative well indicates net attraction at intermediate distances. The minimum corresponds to the equilibrium separation where attractive and repulsive effects balance, giving a stable spacing in a solid. Source
A stable solid structure corresponds to an arrangement where:
Neighboring particles are close enough for substantial attraction
Particles are not forced so close that repulsions destabilise the arrangement
For non-spherical or polar particles, orientations that maximise attraction are favoured
Interaction strength vs structural orderliness
Stronger attractions generally promote:
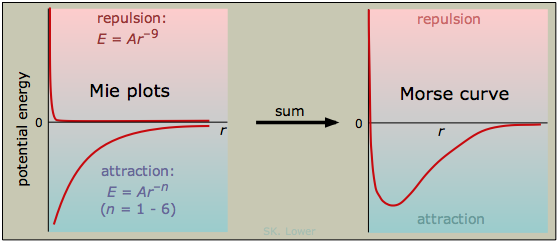
Intermolecular potential-energy curves showing a short-range repulsive term and a longer-range attractive term, and how their sum produces a stable minimum. The minimum represents the preferred separation (lowest potential energy) that particles “lock into” in a solid. A deeper, narrower well corresponds to stronger restoring forces for small displacements, connecting stronger attractions to greater rigidity. Source
Greater orderliness (more consistent particle positions/orientations) because deviations cost more energy
Greater rigidity because displacing particles from their equilibrium positions requires overcoming larger restoring forces
Weaker attractions allow particles to tolerate a wider range of positions and orientations, often reducing orderliness and making the solid easier to deform.
How packing affects orderliness and rigidity
Packing determines how effectively a solid can build many favourable contacts simultaneously.
Efficient packing increases stabilising contacts
When particles pack well:
Each particle can have more near neighbours at attractive distances
The solid gains more total attraction per particle
The structure is harder to disrupt, increasing rigidity at the macroscopic scale
Even if the interaction type is the same, a more efficiently packed arrangement can yield a more rigid material because it creates a more interconnected “contact network.”
Poor packing creates free volume and weak points
If particles do not pack well (due to mismatched sizes, irregular shapes, or constraints on orientation):
Empty spaces (“free volume”) increase
Fewer neighbours are at optimal attractive distances
Local regions can shift more easily under stress
The solid may be less rigid and less orderly
Shape and orientation matter (especially for molecular particles)
For molecular solids, packing is strongly influenced by whether molecules can align to maximise attractions:
Molecules that can align closely and repeatedly tend to form more orderly, rigid solids
Bulky or highly branched shapes often hinder close approach, decreasing packing efficiency and rigidity
If attractive interactions require specific orientations, the solid’s order depends on whether many particles can adopt that orientation pattern without creating steric crowding
Structural imperfections and real-solid behaviour
Perfect packing is an idealisation.
Real solids contain defects that interrupt order and change rigidity:
Missing particles or misaligned regions reduce the number of favourable contacts
Distortions introduce local stress, creating sites where deformation can begin
Mixtures of different-sized particles can disrupt regular packing, lowering orderliness even if attractions remain similar
These ideas help connect particle-level structure to observations such as a solid being stiff versus easily scratched or bent.
What to emphasise in explanations
When asked to explain differences in orderliness or rigidity between solids using particulate reasoning, focus on:
How close particles can approach (packing efficiency and geometry)
How many attractive neighbour contacts each particle can maintain
Whether particle orientations are constrained to achieve attraction
How defects or mismatched particle sizes interrupt repeating arrangements
FAQ
Tight packing increases contact, but rigidity also depends on how steeply energy rises when particles are displaced.
If attractions are weak and the repulsive “springiness” is small, the solid may deform more even when closely packed.
Look for a repeating pattern over long distances.
Short-range alignment without long-range repetition suggests lower orderliness even if particles are close together.
Size mismatch can prevent a repeating arrangement and create gaps.
Small particles may occupy spaces between larger ones, but if the size ratio is unfavourable the result is disrupted packing and reduced rigidity.
If attractions are strongest only for certain orientations, molecules must align in specific ways.
When many molecules cannot simultaneously adopt the best orientation without crowding, packing becomes less efficient and order decreases.
Defects reduce the number of stabilising contacts locally.
Under stress, deformation often begins at these regions because less energy is required to shift particles where packing/order is already disrupted.
Practice Questions
A solid X is made of particles that pack very efficiently, while solid Y is made of similarly sized particles that pack poorly due to irregular shape. State and explain which solid is likely to be more rigid.
Identifies X as more rigid. (1)
Explains that efficient packing gives more/larger number of close-contact neighbour interactions (greater total attraction per particle and/or less free volume), so particles resist displacement. (1)
Two molecular solids, A and B, have comparable intermolecular attraction strengths between individual pairs of molecules. Solid A forms a highly ordered arrangement; solid B forms a less ordered arrangement. Using packing and interparticle interaction ideas, explain how A can be more rigid than B despite similar pairwise interaction strengths.
States that rigidity depends on the total network of interactions, not only a single pairwise interaction. (1)
Explains that better packing in A increases the number of near neighbours per molecule and/or keeps more neighbours at optimal attractive distance. (1)
Links higher neighbour contact count/optimised distances to greater total stabilising attraction per molecule in A. (1)
Explains that poorer packing in B increases free volume and allows easier local shifting/reorientation under stress. (1)
Mentions that reduced order/defects/misalignment in B interrupts repeating interactions, creating weak points that reduce rigidity. (1)

