AP Syllabus focus: ‘Solutions can be solids, liquids, or gases; use composition information to determine the number of solute particles present in a given sample of solution.’
Solutions are not limited to liquids. In AP Chemistry, you must recognise solution states and correctly count solute particles from composition data by tracking chemical identity, dissociation, and moles-to-particles conversions.
States of solutions (solid, liquid, gas)
What makes something a solution?
A solution is a homogeneous mixture at the particulate level: one component is dispersed uniformly within another, regardless of overall phase.
Solvent: the component present in the greatest amount that defines the solution’s phase.
Solute: any component dissolved in the solvent (present in a lesser amount).
Liquid solutions
These are the most familiar: a liquid solvent containing dissolved solute(s).
Solute can be a gas (e.g., dissolved CO₂), a liquid (miscible liquids), or a solid (ionic or molecular compound).
Particle counting depends on whether the solute exists as molecules or ions in solution.
Solid solutions
A solid solution is a single solid phase with uniform composition.
Common in alloys (atoms mixed in a metallic lattice).
Particle counting treats the solute as the dissolved species’ entities (usually atoms), based on amount and composition information provided.
Gas solutions
A gas solution is a homogeneous mixture of gases.
The solvent is the gas present in greatest amount; other gases are solutes.
Particle counting typically treats each gas as molecules (or atoms for monatomic gases), using moles and Avogadro’s number.
Counting solute particles: what counts as a “particle”?
The “particle” depends on chemical form
In AP Chemistry, “particles” are the discrete entities present after mixing:
Molecular solutes (nonelectrolytes): particles are molecules (e.g., glucose stays as C₆H₁₂O₆ units).
Ionic solutes (electrolytes): particles are ions produced upon dissolving (e.g., NaCl → Na⁺ and Cl⁻).
Some solutes may partially ionise; when not explicitly stated, AP-style problems often specify behaviour (or imply strong electrolytes for soluble salts/strong acids).
Formula unit: the lowest whole-number ratio of ions in an ionic compound, used to represent one “unit” of an ionic solid (not a molecule).
Convert amount of solute to number of particles
Particle counting generally follows: determine moles of solute, adjust for particles produced per formula unit (if any), then convert to number of particles.

This conversion “road map” shows the standard factor-label pathway from mass (g) to amount (mol) and then to particle count using Avogadro’s number. It highlights where molar mass () and () enter the calculation, which is exactly the logic behind and . Source
= number of particles (entities)
= amount of substance (mol)
= Avogadro’s constant,
This relationship applies to molecules in liquid/gas solutions and to ions generated in ionic solutions.
= amount of substance (mol)
= mass of solute (g)
= molar mass (g mol)
Account for dissociation: particles per formula unit
For ionic solutes that fully dissociate, each formula unit creates multiple solute particles:
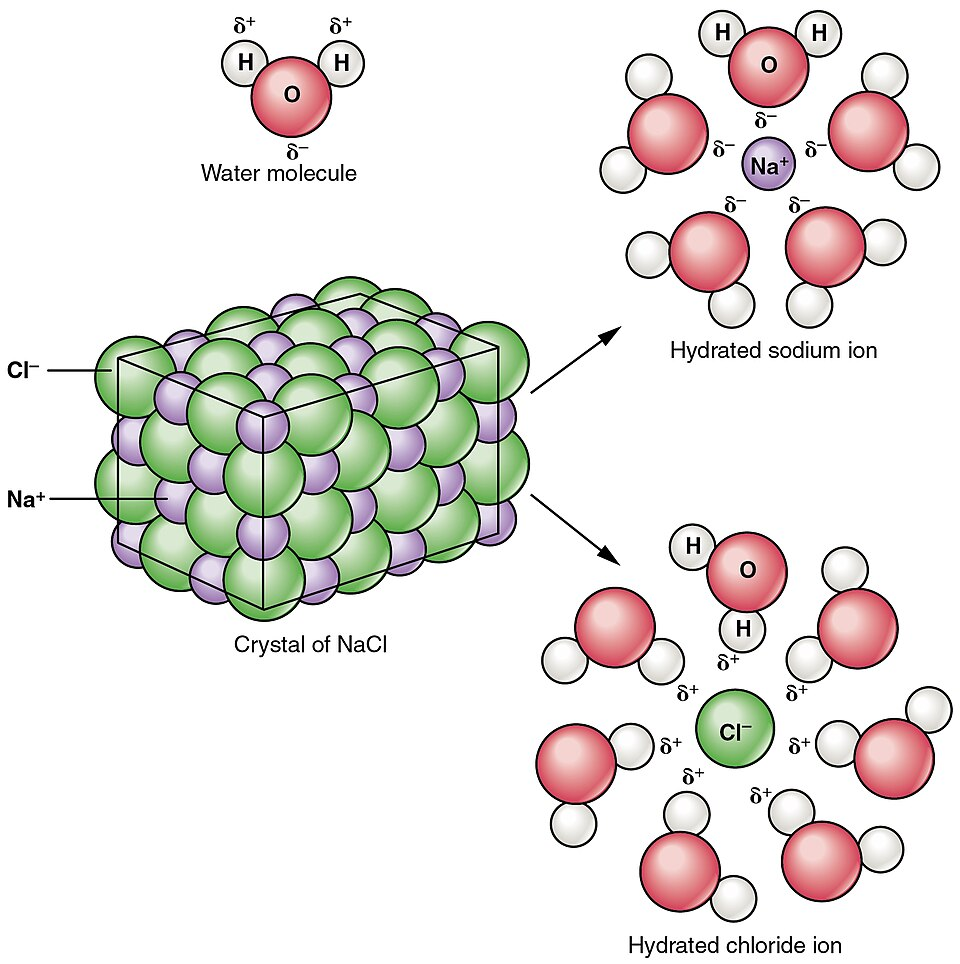
This diagram depicts an NaCl crystal dissolving in water and separating into hydrated and ions. It visually clarifies that the relevant “particles” for an ionic solute are the ions produced upon dissolution, not intact formula units, which is the key step before applying . Source
NaCl → 1 Na⁺ + 1 Cl⁻ (2 particles per formula unit)
CaCl₂ → 1 Ca²⁺ + 2 Cl⁻ (3 particles per formula unit)
Al₂(SO₄)₃ → 2 Al³⁺ + 3 SO₄²⁻ (5 particles per formula unit)
A reliable method is to:
Write the dissociation into ions (with coefficients).
Multiply moles of formula units by the total ion coefficient sum to get moles of ions (total particles), or count specific ion types if asked.
Using composition information to determine solute particles
Common composition cues (conceptual)
Problems may give composition information in several ways; regardless of format, the logic is consistent:
Identify the solute chemical formula and whether it remains molecular or forms ions.
Determine moles of solute units present (from mass, provided amount, or other stated composition data).
Convert to moles of particles actually present in solution (molecules or ions).
Convert moles of particles to number of particles using .
Counting “total solute particles” vs “particles of a given type”
Be precise about what is being counted:
Total solute particles in an ionic solution usually means “total ions present from the solute.”
Particles of a specific solute component means count only that ion or molecular species (e.g., number of Cl⁻ ions).
In gas solutions, “particles” may mean molecules (e.g., N₂) or atoms for noble gases (e.g., Ar).
FAQ
Usually atoms of the solute element are counted as particles.
If an intermetallic compound forms, it is no longer treated as a simple solution.
Water of crystallisation becomes additional water molecules in the mixture.
The ionic compound still dissociates based on its formula unit; the waters do not become ions.
No. Count the particles of the species actually present after reaction.
You must use the reaction stoichiometry to determine final amounts.
It depends on the degree of ionisation, often given or implied.
Without that information, you cannot assume complete conversion into ions.
Not for this topic.
Here, “particles” means molecules, atoms, formula units, or ions as chemically distinct entities.
Practice Questions
(2 marks) A sample contains of dissolved . Assuming complete dissociation, state the number of moles of chloride ions present.
Correct dissociation or ratio: (1)
(1)
(5 marks) A gas solution is made by mixing He and (uniform mixture). Determine: (a) which component is the solvent (1 mark) (b) the total number of solute particles present (4 marks)
(a) He is solvent because it is in greatest amount (1)
(b) Identify solute as (1)
Convert moles solute to particles using (1)
Substitute and (1)
molecules (allow 2 s.f.) (1)

