AP Syllabus focus: ‘Express solution composition using molarity, M = nsolute / Lsolution, and calculate solute amount, solution volume, or molarity using dimensional analysis and appropriate units.’
Molarity is the primary concentration unit used in AP Chemistry problem solving. Mastering unit conversions and dimensional analysis lets you move confidently between measured mass, moles of solute, and the volume of solution.
Core idea: molarity links moles to litres
Molarity connects the amount of dissolved solute to the final solution volume. It is a ratio, so correct units and careful rearrangement matter more than memorising procedures.
Molarity (M): Moles of solute per litre of solution.
Molarity always uses litres of solution (not litres of solvent).
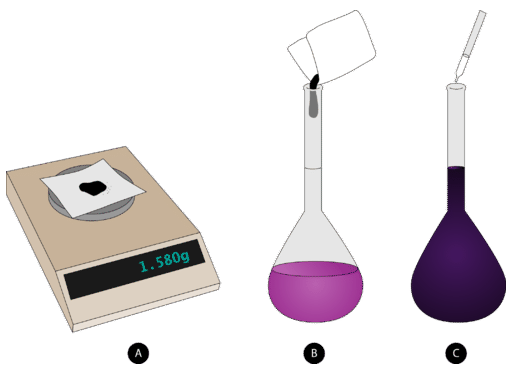
This diagram illustrates the standard technique for preparing a solution of a specified molarity using a volumetric flask: accurately weigh the solute, dissolve it in a partial volume of solvent, then dilute to the calibration mark to reach the final solution volume. It reinforces that the volume in is the total solution volume after dissolution, not the amount of solvent initially added. Source
In practice, solution volumes are often measured in mL and must be converted.
= molarity,
= moles of solute, mol
= volume of solution, L
Use the form that isolates the unknown, then check that units cancel to give the desired unit.
Dimensional analysis and unit discipline
Essential unit conversions
Correct molarity calculations rely on converting to consistent base units before substituting:

This OpenStax example shows a molarity calculation carried out with explicit unit conversions, converting mass to moles (via molar mass) and then dividing by solution volume in litres. The layout mirrors dimensional analysis: each factor is chosen so units cancel cleanly to yield . Source
Volume:
Amount: use moles (mol), not particles or grams, in the molarity equation
Mass-to-moles conversion requires molar mass in
A quick sanity check: if you increase solution volume while keeping moles fixed, molarity must decrease (dilution lowers concentration).
Interpreting what “solute” means in
In molarity, is the number of moles of the chemical formula you dissolved (or that is present).
For molecular solutes (e.g., glucose), moles refer to molecules.
For ionic solutes (e.g., NaCl), moles refer to formula units of NaCl present in solution.
Be careful not to mix up “moles of solute” with “moles of ions produced”; unless a question explicitly asks for particles/ions, molarity problems typically track the solute formula.
Finding an unknown: what information you need
Calculating molarity,
To find molarity, you must know:
moles of solute (directly, or from mass and molar mass)
final solution volume in litres
Common pitfalls:
using the volume of water added rather than the final solution volume
leaving volume in mL
using grams directly in without converting to moles
Calculating moles of solute,
To find moles of solute, you must know:
molarity
solution volume in litres
This is frequently used when determining how much solute is present in a measured aliquot of solution (a portion with a known volume).
Calculating solution volume,
To find the solution volume corresponding to a given amount and molarity, you must know:
moles of solute
molarity
This often appears when planning solution preparation or checking whether sufficient volume exists for a target concentration.
Dilution as a molarity calculation
A dilution changes concentration by changing volume, while the moles of solute stay constant (assuming no reaction and no solute lost). This can be handled by:
computing initial moles with , then computing the new molarity with , or
using the relationship (valid only when the solute amount is conserved)
Key idea: dilution problems are still dimensional analysis problems—track what stays constant (moles) and what changes (volume, thus molarity).
Significant figures and reporting
Because molarity is a ratio of measured quantities, the final reported molarity (or calculated volume/amount) should reflect appropriate significant figures based on given data. Keep units explicit at every step; unit-cancellation is part of the reasoning AP Chemistry expects.
FAQ
Molarity is defined using the final, measurable volume after mixing.
Using solvent volume would ignore volume changes on dissolution and mixing, so it would not match how solutions are prepared in volumetric glassware.
Molarity depends on solution volume, and volume changes with temperature due to expansion/contraction.
In precise work, solutions are made up to the calibration mark at the specified temperature (often $20^\circ\text{C}$ or $25^\circ\text{C}$), then used near that temperature.
Use a volumetric flask when the final concentration must be accurate, because it is calibrated for a specific volume.
Graduated cylinders have larger tolerances and are better for approximate volumes.
Base the significant figures on the limiting measured quantity (often the mass or volume provided with the fewest significant figures).
Keep extra digits during intermediate steps, then round once at the end.
Mixing up $V_1$ (volume of stock taken) with $V_2$ (final total volume after dilution).
A quick check is that if $M_1 > M_2$, then $V_1$ must be smaller than $V_2$.
Practice Questions
Q1 (3 marks) A student dissolves of to make of solution. Calculate the molarity of the solution. (Molar mass of .)
Converts mass to moles: (1)
Converts volume to litres: (1)
Uses with correct unit and appropriate s.f. (1)
Q2 (5 marks) A laboratory has a stock solution of with concentration . A student needs of . (a) Determine the volume of stock solution required.
(b) State one check, based on units or magnitude, that confirms your value is reasonable.
Uses conservation of moles for dilution: or equivalent mole-tracking method stated (1)
Substitutes with (1)
Calculates in litres (1)
Converts to mL with correct s.f. (1)
Reasonableness check: e.g., required stock volume is less than because stock is more concentrated; or unit-cancellation leading to volume units (1)

