AP Syllabus focus: ‘Chromatography (paper, thin-layer, or column) separates species by differences in interaction strengths among solution components (mobile phase) and with the stationary phase.’
Chromatography is a core separation technique in chemistry that relies on competition between two sets of attractions. Understanding how particles interact with a moving fluid and a fixed surface explains why mixtures separate into distinct bands.
Core idea: separation by competing attractions
In chromatography, each component of a mixture repeatedly “chooses” between spending time in the mobile phase (moving) or adhering to the stationary phase (not moving). Components that interact more strongly with the stationary phase move more slowly and separate from faster-moving components.
Chromatography: A separation method where mixture components distribute differently between a moving phase and a stationary phase, producing different migration rates.
A key point for AP Chemistry is that separation is driven by differences in interaction strengths, not by differences in mass alone.
Phases and what they do
Stationary phase (fixed)
The stationary phase is the surface that provides adsorption sites and intermolecular interactions (often hydrogen bonding, dipole–dipole, and London dispersion attractions, depending on the materials involved).
Stationary phase: The non-moving solid (or a liquid coated on a solid) that mixture components can adhere to through intermolecular attractions.
The more strongly a component is attracted to the stationary phase, the longer it is retained and the less distance it travels in a given time.
Mobile phase (moving)
The mobile phase is the fluid that carries dissolved mixture components through or over the stationary phase. It competes with the stationary phase by solvating the components.
Mobile phase: The moving solvent (or solvent mixture) that transports mixture components and competes with the stationary phase for intermolecular interactions.
If the mobile phase interacts strongly with a component (good solvation), that component spends more time moving and less time stuck to the stationary surface.
How interactions control migration
Migration rate reflects a dynamic balance:
Adsorption to stationary phase slows movement (stronger attractions → longer “sticking” time).
Solvation by mobile phase speeds movement (stronger solvation → more time traveling with the solvent front).
Separation occurs because different components experience different net interaction strengths due to:
Differences in polarity and functional groups
Differences in ability to form hydrogen bonds
Differences in surface interactions (e.g., with silica, paper cellulose, or alumina)
Differences in how well the mobile phase solvates each component
Major AP-relevant chromatography types
Paper chromatography
Stationary phase: cellulose paper (polar surface with many –OH groups)
Mobile phase: solvent rising by capillary action
Components that hydrogen bond strongly to cellulose tend to move less.
Thin-layer chromatography (TLC)
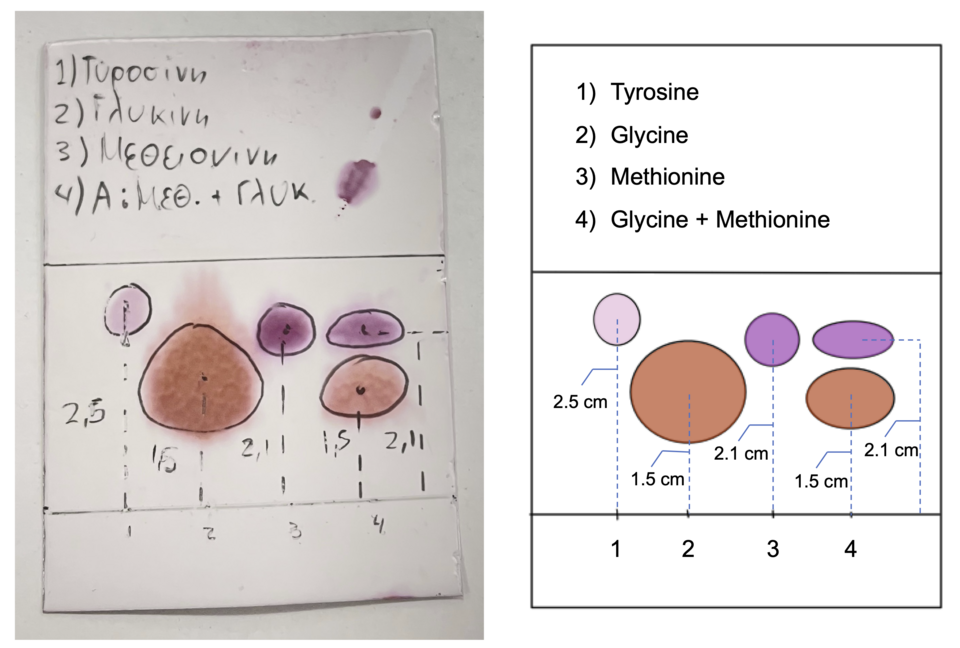
A labeled thin-layer chromatography (TLC) plate showing a baseline (origin), solvent front, and multiple spots for standards versus an unknown mixture. The different spot heights visualize different migration rates caused by different strengths of interaction with the silica/alumina stationary phase versus the mobile solvent. This is the experimental “band/spot separation” your notes describe in TLC. Source
Stationary phase: thin layer of silica gel or alumina (commonly polar)
Mobile phase: solvent in a developing chamber
Highly adsorbing components lag behind; weakly adsorbing components travel farther.
Column chromatography
Stationary phase: packed solid (often silica/alumina)
Mobile phase: solvent flowing through the column
Components separate into bands; those with stronger stationary-phase attraction elute later.

A schematic of normal-phase column chromatography showing colored bands separating as solvent flows through a polar stationary phase. The figure emphasizes the competition between analyte–stationary-phase attraction (retention) and analyte–mobile-phase attraction (carrying forward), explaining why bands move at different speeds. It also visually connects retention strength to elution order (later elution for more strongly retained components). Source
Elution: The process of a component being carried out of a column by the mobile phase; stronger stationary-phase attraction generally means later elution.
Practical levers that change separation (interaction-focused)
Without relying on calculations, AP Chemistry expects qualitative reasoning about what happens when conditions change:
Changing the mobile phase composition changes competition for interactions.
A mobile phase that better solvates a component can reduce that component’s adsorption and increase its migration rate.
Changing the stationary phase material changes which attractions dominate.
A more polar stationary surface tends to retain polar species more strongly (via dipole interactions and hydrogen bonding).
If two components have very similar interaction strengths with both phases, they may not separate well (overlapping bands/spots).
FAQ
Silica surfaces present polar $\text{Si–O}$ bonds and many $\text{Si–OH}$ groups, enabling strong dipole interactions and hydrogen bonding with suitable solutes.
Specific interactions can differ even at similar overall polarity, such as differing hydrogen-bond donor/acceptor ability, shape, or how well each fits adsorption sites.
It can compete for adsorption sites on the stationary phase and improve solvation of polar solutes, often increasing migration and reducing retention.
Common causes include overloading the sample, strong irreversible adsorption, or partial solubility issues that create a range of interaction strengths during migration.
Normal-phase uses a polar stationary phase with a relatively nonpolar mobile phase; reversed-phase uses a nonpolar stationary phase with a more polar mobile phase, flipping which interactions dominate.
Practice Questions
In thin-layer chromatography using a polar silica stationary phase, component A travels a shorter distance than component B in the same solvent. Explain what this implies about the relative interactions of A and B with the stationary phase.
A interacts more strongly with (is more strongly adsorbed to) the stationary phase than B. (1)
Stronger stationary-phase attraction reduces migration rate, so A travels less/further is B. (1)
A student separates a mixture using column chromatography with a silica stationary phase. (a) Describe how differences in interaction strengths with the mobile and stationary phases produce separation. (3 marks) (b) The student switches to a mobile phase that is better at solvating one component. Predict how that component’s elution time changes and justify. (2 marks)
(a)
Components repeatedly adsorb to the stationary phase and dissolve in the mobile phase. (1)
Different components have different strengths of attraction to the stationary phase, causing different retention times. (1)
Stronger attraction to stationary phase means slower movement/later elution; weaker attraction means faster movement/earlier elution. (1)
(b)
That component elutes sooner (reduced retention). (1)
Better solvation by the mobile phase decreases adsorption to the stationary phase/increases time spent in the mobile phase. (1)

