AP Syllabus focus: ‘A chromatogram can be used to infer the relative polarities of mixture components based on how strongly each component interacts with the stationary phase versus the mobile phase.’
Chromatography separates mixture components because each substance partitions differently between a moving solvent and a solid (or coated) surface. By reading relative travel distances on a chromatogram, you can infer relative polarity.
What a chromatogram shows
A chromatogram is the visual record of separation: distinct spots (or bands) at different positions along the direction of solvent travel.
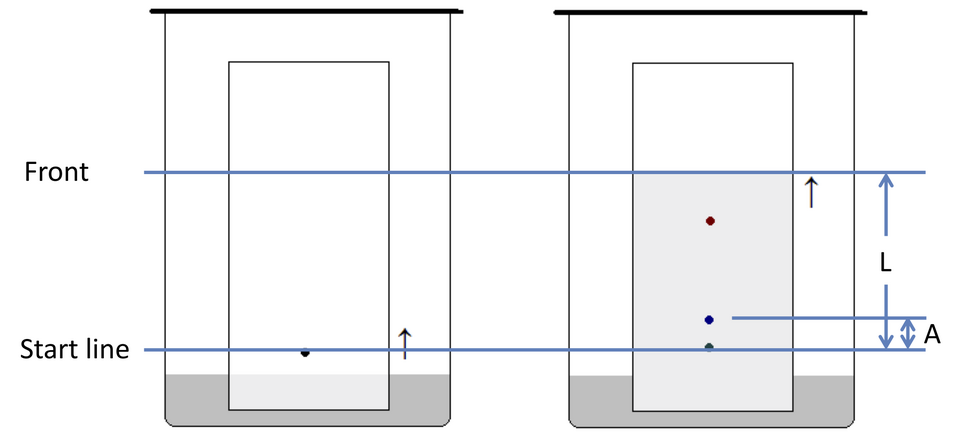
Schematic TLC chromatogram showing the baseline (origin), solvent front, and how distances are measured to compute the retention factor. It visually encodes that is the ratio of spot travel to solvent-front travel, so the same plate can be compared across runs with different solvent-front distances. Source
Each spot corresponds to a different component (or sometimes more than one if unresolved).
Key interactions that control movement
Separation is governed by competition between:
Attraction to the stationary phase (surface the solute can “stick” to)
Solubility in the mobile phase (solvent that carries solute forward)
More time interacting with the stationary phase means slower movement; more time dissolved in the mobile phase means faster movement.
Inferring polarity from spot positions (normal-phase assumption)
Most AP-style paper/TLC scenarios implicitly use a polar stationary phase (e.g., paper cellulose, silica gel). Under this common condition:
More polar components interact more strongly with the stationary surface (via dipole interactions and hydrogen bonding)
travel less far
have smaller
Less polar components interact less strongly with the stationary surface and prefer the solvent
travel farther
have larger
This supports the syllabus idea: compare how strongly each component interacts with the stationary phase versus the mobile phase, then rank relative polarities.
Stationary phase — the immobile phase (solid or a liquid coated on a solid) that provides a surface for solute–surface interactions during chromatography.
A chromatogram therefore lets you rank components: the spot nearest the origin is typically the most polar (in normal-phase), and the spot nearest the solvent front is typically the least polar.
Using to compare travel in a standardised way
Because different chromatograms can have different solvent-front distances, the retention factor normalises spot travel.
Retention factor () — the ratio of the distance travelled by a solute spot to the distance travelled by the solvent front, measured from the origin line on the same chromatogram.
Even when two components both move “far,” the one with the larger is interpreted as interacting more with the mobile phase (and, in normal-phase, being less polar).
= distance from origin to centre of solute spot (same units for both distances)
= distance from origin to solvent front
What “stronger interaction” means at the particle level
On a polar stationary surface, stronger interaction typically arises from:
Dipole–dipole attraction between polar solute groups and polar surface groups
Hydrogen bonding between solute functional groups (e.g., O–H, N–H, C=O) and surface hydroxyl groups
Multiple interaction sites on a single molecule increasing overall adhesion
A component that can make more (or stronger) attractions to the stationary phase spends more time “stuck,” so it advances less during the same solvent run.
Conditions and caveats when inferring polarity
The mobile phase can change the apparent polarity ranking
A more polar mobile phase can compete for stationary-phase sites and pull polar solutes along more effectively, often increasing their . Polarity inference is most meaningful when:
The stationary/mobile phases are specified or consistent
Components are compared on the same chromatogram (same run conditions)
Reverse-phase warning (interpretation flips)


Diagram illustrating polarity-based chromatography modes, emphasizing that the identity of the stationary phase (polar vs nonpolar) determines which analytes are retained. It reinforces the core reasoning: stronger attraction to the stationary phase increases retention, while stronger affinity for the mobile phase increases migration/elution. Source
If the stationary phase is nonpolar (reverse-phase), then:
More polar solutes prefer the (often more polar) mobile phase and move farther
Less polar solutes are retained more and move less far
When asked to infer polarity, always tie your claim explicitly to “interaction with stationary phase versus mobile phase,” not just to distance alone.
Common interpretation pitfalls
Spots at the same height suggest similar interaction strengths (similar effective polarity under those conditions), not necessarily identical identity.
Very faint or streaked spots can make spot centres ambiguous, weakening polarity comparisons.
If a spot is at (or near) the solvent front, it may be “too soluble” in the mobile phase to distinguish well.
FAQ
Look for the stationary phase identity.
Silica/alumina/cellulose paper → typically polar (normal-phase)
C18-coated plates (“ODS”, “RP-18”) → nonpolar (reverse-phase)
Common causes include overloading (too much sample), partial solubility in the mobile phase, or strong adsorption to the stationary phase that drags material during elution.
Adjusting solvent polarity changes competition between solute–surface and solute–solvent interactions, often shifting $R_f$ values differently for each component and improving resolution.
Typical visualisation methods include UV illumination (for UV-active compounds) or staining (e.g., iodine chamber, ninhydrin for amino acids), which reveals spots after the run.
If a solute is partially ionised, electrostatic attraction to the stationary phase (or altered solubility in the mobile phase) can dominate, so observed movement reflects charge state as well as molecular polarity.
Practice Questions
A TLC plate with a polar stationary phase shows two spots from a mixture: spot A has and spot B has . Infer which component is more polar and state why.
Identifies A as more polar (1)
Links lower to stronger interaction with the stationary phase / weaker preference for mobile phase (1)
A student runs paper chromatography to compare three unknowns (X, Y, Z) using the same solvent. The chromatogram shows values: X = 0.10, Y = 0.45, Z = 0.80.
(a) Rank X, Y, Z in order of increasing polarity assuming a polar stationary phase. (2 marks)
(b) Explain, in terms of interactions with the stationary and mobile phases, why the most polar component travels the least. (3 marks)
(a) Z least polar, then Y, then X most polar (either “increasing polarity: Z < Y < X” or equivalent) (2)
(b) Any three distinct points (1 each):
More polar solute forms stronger attractions to polar stationary phase (e.g., dipole interactions / hydrogen bonding)
Increased retention means more time adsorbed and less net movement
Less time dissolved in mobile phase / lower affinity for mobile phase gives smaller

