AP Syllabus focus: ‘Chemical changes form new substances; signs can include heat/light, gas formation, precipitate formation, and/or color change.’
Chemical reactions can often be detected by careful observation. In AP Chemistry, you should recognise common signs that new substances may have formed and judge how convincing each sign is.
Core idea: evidence vs proof
A chemical change produces products with different composition and properties than the reactants. Observations provide evidence of chemical change, but any single sign can sometimes have a non-chemical explanation, so context matters.
Chemical change: a process in which reactants are converted into new substances with a different chemical composition.
Evidence of chemical change: the major observable signs
The AP Chemistry emphasis is on four common signs that new substances may be forming: heat/light, gas formation, precipitate formation, and/or color change.
Heat and/or light
Energy changes frequently accompany bond breaking/forming, so you may observe:
Temperature increase of the surroundings (reaction feels warmer): evidence for an exothermic chemical change.
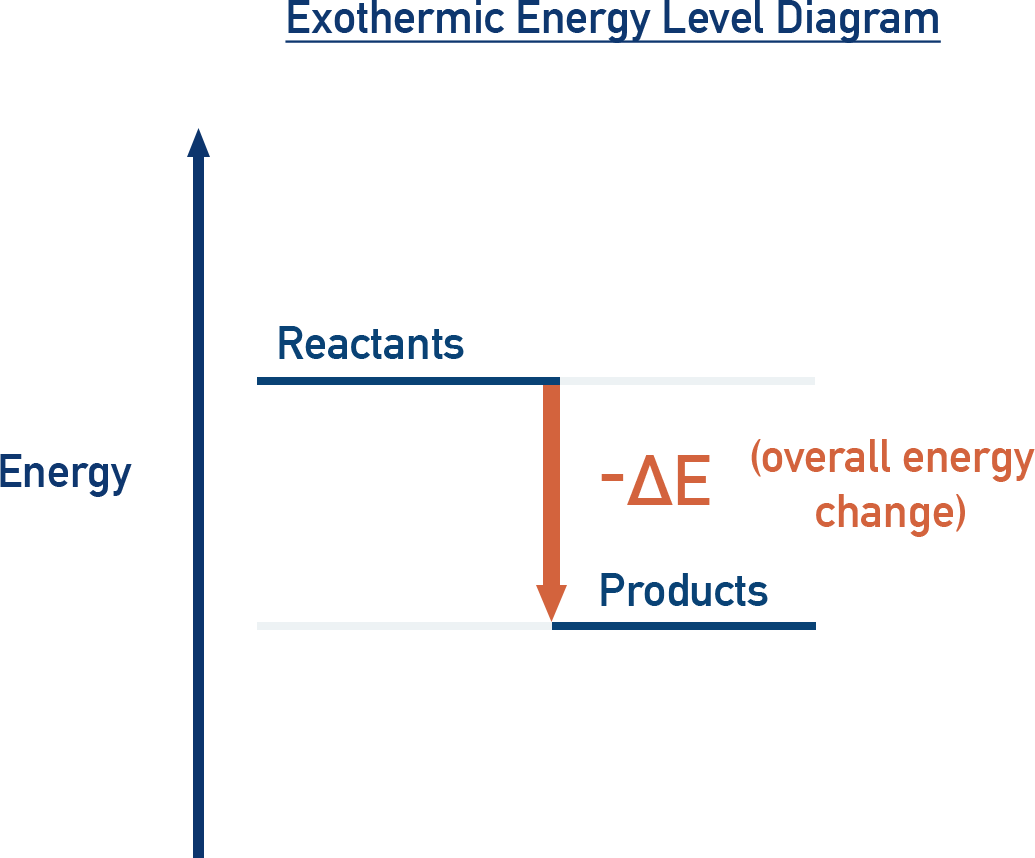
An exothermic energy-level diagram shows products at lower potential energy than reactants, so energy is released to the surroundings. The labeled emphasizes that the overall energy change is negative, matching the observation of a temperature increase (and sometimes light emission) during product formation. Source
Temperature decrease of the surroundings (reaction feels cooler): evidence for an endothermic chemical change.
Light emission (glow, spark, sustained luminescence): strong evidence that energy is being released during product formation.
Interpretation tips:
Treat temperature change as evidence, not automatic proof: some non-reaction processes (like strong mixing or dissolving) can also warm or cool a solution.
A reliable observation describes both direction and circumstances (e.g., “temperature rose immediately after combining solutions”).
Gas formation
Formation of a new gaseous product is commonly seen as:
Bubbling/effervescence within a liquid
Fizzing at a solid–liquid interface
Expansion/pressure increase in a closed container
Interpretation tips:
Gas formation is most convincing when bubbling begins upon mixing reactants and persists beyond brief trapped-air release.
Distinguish “new gas produced” from “gas escaping” (for example, air released from porous solids or a warm liquid releasing dissolved gas).
Precipitate formation
When ions or molecules in solution form an insoluble product, you may observe:
Cloudiness appearing in a previously clear mixture
Solid particles forming and settling

A precipitation reaction can produce a visibly separate solid phase in an otherwise liquid mixture. Here, insoluble lead(II) iodide forms as a bright yellow solid that settles, illustrating how new solid particles appearing in solution are strong evidence of new substances forming. Source
A solid that can be collected by filtration (in a lab context)
Precipitate: an insoluble solid that forms from a solution during a chemical reaction.
Interpretation tips:
Precipitate evidence is stronger when the solid appears rapidly after combining two aqueous solutions.
Do not confuse a precipitate with temporary cloudiness from mixing, or with crystallisation that occurs simply due to cooling or evaporation.
Color change
A visible color change can indicate the formation of new species with different light absorption. Useful observations include:
A new persistent color after mixing
Color that appears with simultaneous evidence (such as precipitate or gas)
A change that cannot be explained by simple dilution (lighter shade) or by layering before full mixing
Interpretation tips:
Record the initial and final colors precisely (e.g., “colourless to pale yellow” is more informative than “changed colour”).
Some color changes can occur without forming new substances (for example, changes in concentration or physical dispersion), so look for supporting signs.
Using evidence well (what AP expects)
When asked whether a chemical change occurred, strong answers:
Identify one or more of the listed signs (heat/light, gas, precipitate, color change).
Connect the observation to the idea that new substances form in chemical changes.
Acknowledge that multiple consistent signs are more convincing than one sign alone.
FAQ
Some reactions form products that look similar to reactants (no precipitate, no strong colour).
Evidence may require measurements rather than appearance, such as:
mass in a closed system
changes in electrical conductivity
instrument-based detection (e.g., spectroscopy)
Look for whether bubbling:
starts only after reactants contact
continues steadily rather than stopping quickly
correlates with other changes (temperature, odour, new solid)
Collecting the gas and testing its properties provides stronger confirmation.
Apparent solids can come from:
emulsions (tiny droplets suspended)
undissolved reactant due to poor mixing
dust/contamination
temperature-driven crystallisation during cooling
Checking whether the solid re-dissolves upon warming or dilution can help distinguish causes.
Colour can change due to:
dilution (same species, lower concentration)
scattering from suspended particles
lighting/background effects
mixing two coloured solutions to make an intermediate shade
Instrumental absorbance measurements can separate true chemical change from optical effects.
If a reaction is very slow, signs (gas, colour, precipitate) may appear gradually and be overlooked. If very fast, brief signs can be missed or seem ambiguous.
Improving observation often involves controlling:
concentration
temperature
mixing order and timing
Practice Questions
Question 1 (3 marks) A student mixes two colourless aqueous solutions. The mixture immediately turns cloudy and a solid settles at the bottom. State two observations that provide evidence of a chemical change and name the type of evidence each represents.
Cloudiness/solid appears: precipitate formation (1)
Solid settles/forms in solution (or can be filtered/collected): precipitate formation (1)
Explicit statement linking to chemical change/new substance formed (e.g., “an insoluble solid product formed”): (1)
Question 2 (6 marks) A student combines two aqueous solutions and observes the following: the beaker becomes warm, bubbles appear for about 10 seconds, and the solution changes from blue to green. Evaluate how strongly these observations support that a chemical change occurred. Your answer should reference the specific signs and comment on the reliability of each sign.
Warm beaker identified as heat release evidence (1)
Note that heat change alone is not definitive (may occur for non-chemical reasons) (1)
Bubbles identified as gas formation evidence (1)
Note that brief bubbling could be ambiguous (e.g., trapped air) so duration/context matters (1)
Blue to green identified as colour change evidence (1)
Overall evaluation that multiple consistent signs together provide stronger support of chemical change than any single sign (1)

