AP Syllabus focus: ‘Physical and chemical processes can be represented symbolically with balanced equations.’
Chemical changes and many physical processes can be communicated efficiently using chemical equations. In AP Chemistry, you must translate descriptions into symbols, write correct formulas, and balance equations so they accurately represent particle-level changes.
What a chemical equation represents
A chemical equation is a symbolic model showing which species are present and how they change.
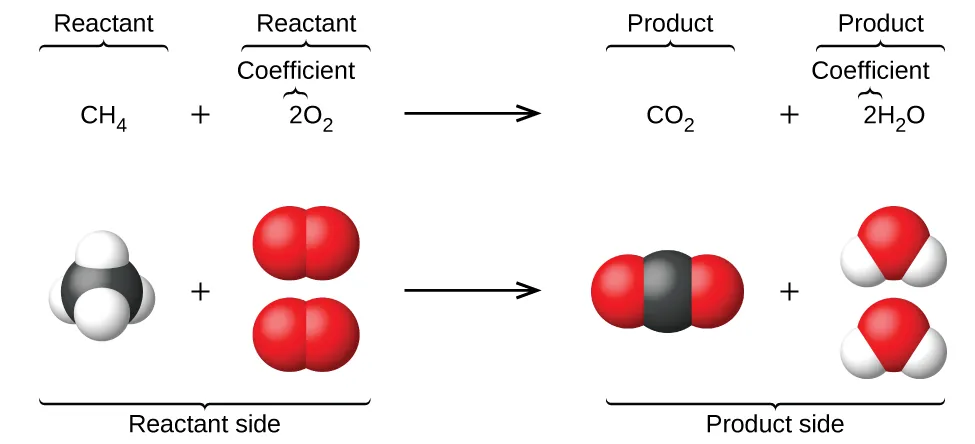
A balanced molecular equation is shown alongside space-filling models of the reactant and product molecules for methane combustion. The diagram visually connects coefficients to relative numbers of particles while preserving each substance’s identity (the formulas themselves do not change). This helps students interpret an equation as a particle-level model consistent with conservation of atoms. Source
It communicates:
Reactants (left) and products (right)
Relative numbers of particles through coefficients
Often, physical form using state symbols: (s), (l), (g), (aq)
Sometimes, conditions above the arrow (e.g., heat, catalyst), without changing balancing
Balanced chemical equation: A symbolic representation of a process in which coefficients are chosen so each element has the same number of atoms on both sides of the reaction arrow.
Balanced equations can represent:
Chemical reactions (atoms rearrange to form new substances)
Physical processes when composition stays the same (e.g., phase changes), using the same substance formula with different states
Interpreting symbols correctly
Key conventions to read and write accurately:
Subscripts are part of the formula and describe composition (do not change when balancing).
Coefficients multiply the entire formula unit and indicate relative amounts (these are adjusted to balance).
The reaction arrow () indicates a process; it does not imply rate or extent by itself.
State symbols clarify what is actually present (important for solutions and gases).
Writing a balanced equation (conceptual procedure)
To represent a described change with a balanced equation:
Identify the species present (names correct formulas).
Write a skeleton equation with correct reactant/product formulas.
Add appropriate state symbols if the context specifies phase or aqueous solution.
Balance by adjusting coefficients only:
Balance elements that appear in fewer compounds first.
Treat unchanged polyatomic ions as a unit when appropriate (only if they truly remain intact in the written formulas).
Leave elemental diatomics as written (e.g., when they appear as elements).
Reduce coefficients to the lowest whole-number ratio.
Do a final check that the balanced equation still matches the described process (correct species and phases).
A compact way to express balancing is to use placeholders for coefficients.
= Stoichiometric coefficients (whole-number ratios of particles or moles)
In this form, changing and changes the relative amounts, while the formulas and must remain chemically correct.
Representing physical processes with equations
Some non-reaction changes can still be written as balanced equations because the identity of the substance is unchanged:
Phase changes: same formula, different state symbols (e.g., liquid to gas)
Mixing without reaction: may be described without a chemical equation, but if written, it should not invent new products
The key requirement is that the symbols match the real chemical identity: if no new substances form, the equation must reflect that.
Common pitfalls to avoid
Changing subscripts to balance (this changes the substance).
Forgetting diatomic elements in their elemental form.
Omitting state symbols when the question hinges on phase (e.g., gas formation).
Writing products that violate basic composition (e.g., impossible formulas from incorrect charge reasoning).
FAQ
Coefficients count relative numbers of particles or moles. Fractions can be cleared by multiplying all coefficients to give an equivalent whole-number ratio, which is the standard chemical convention.
Include them when the physical form is given or affects interpretation (e.g., gas evolution, aqueous solutions). They can be omitted if not specified and not required to distinguish species in the context.
Write an equation when a prompt describes a defined change with identifiable species. If the description is only “mixing” with no reaction, an equation may be unnecessary or could be misleading.
Count each element on both sides, using coefficients as multipliers. A quick scan often starts with elements that appear in only one reactant and one product.
Subscripts define the compound’s identity and composition. Changing them creates a different substance, so the symbols would no longer represent the stated reactants/products.
Practice Questions
(1–3 marks) Write the balanced chemical equation, including state symbols, for the formation of liquid water from hydrogen gas and oxygen gas.
Correct formulas and states: , , (1)
Balanced coefficients: (1)
(4–6 marks) Ethanol combusts completely in oxygen to form carbon dioxide and water vapour. Write the balanced chemical equation with state symbols, and state two conventions that must be followed when balancing.
Correct reactant and product formulas: , , , (2)
Correct state symbols consistent with statement: , , , (1)
Correct balancing: (2)
Two valid conventions (any two): “change coefficients not subscripts”, “use lowest whole-number ratio”, “keep diatomic elements as ”, “ensure formulas are chemically correct” (1)

