AP Syllabus focus: ‘Because atoms are conserved in chemical processes, you can calculate product amounts from reactant amounts, or reactant amounts from product amounts.’
Stoichiometry links measurable amounts of reactants and products by treating a chemical reaction as a fixed “recipe.”
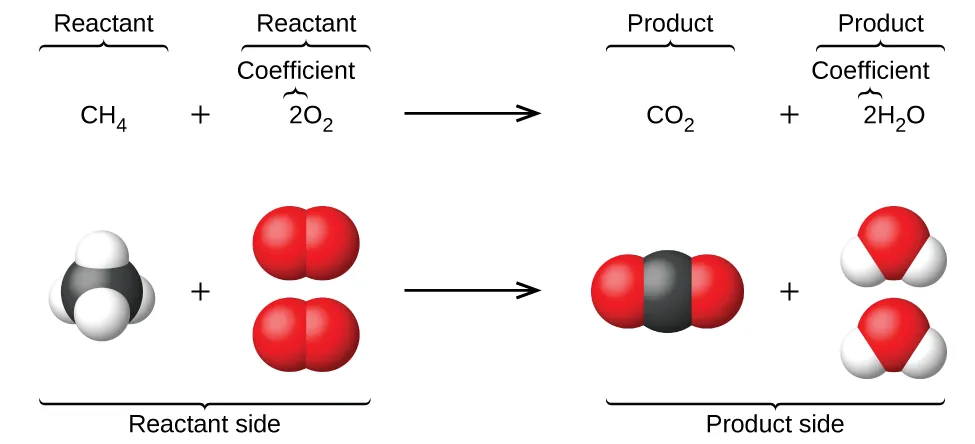
A balanced combustion equation is paired with space-filling molecular models to show how coefficients encode the reacting “recipe” at the particle level. The diagram visually demonstrates conservation by matching the atom inventory before and after reaction while distinguishing reactants from products and highlighting coefficients. Source
The key idea is conservation of atoms, enforced by a balanced chemical equation, which enables quantitative predictions.
Core principle: conservation connects amounts
In any chemical reaction, atoms are rearranged, not created or destroyed. When an equation is balanced, it encodes the fixed proportional relationship among reacting particles, so any starting amount can be related to any resulting amount.
Stoichiometry: The quantitative study of the relationships between amounts of reactants and products in a chemical reaction, based on a balanced equation.
A balanced equation is the “bridge” between substances:
It shows which substances are present (formulas)
It shows their relative particle counts (coefficients)
It ensures the same number of each type of atom appears on both sides
What “amount” means in AP Chemistry
Chemical amounts can be described in several ways (mass, particles, moles). For reaction calculations, moles are the most direct because coefficients in a balanced equation compare numbers of particles, and the mole is the counting unit for particles.
Converting to and from moles (the common pathway)
You typically convert a given quantity to moles, use the balanced equation to relate reactant and product amounts, then convert to the requested unit.

This diagram compares equal-mass samples of different substances to highlight that the number of moles depends on molar mass. It reinforces the relationship (and therefore ) by visually separating the ideas of mass, amount of substance, and molar mass. Source
= amount of substance in moles,
= mass of sample,
= molar mass,
This conversion is often necessary because lab data are commonly collected as masses, but reaction relationships are most naturally tracked in moles.
Using conservation to relate reactants and products
The specification focus means you should be able to move in either direction:
Reactant amount → product amount (predicting how much forms)
Product amount → reactant amount (inferring how much was required or consumed)
Why balancing is non-negotiable
An unbalanced equation violates conservation of atoms, so it cannot be used to relate amounts. Once balanced, the equation implies that:
Each “reaction event” consumes reactants and forms products in a fixed pattern
Scaling up or down does not change the pattern, only the size of the amounts
Interpreting coefficients as conservation-based ratios
Coefficients represent the relative numbers of formula units (or molecules) that react. Because atoms are conserved, these ratios must be followed for matter to be fully accounted for.
Practical implications:
If you know how many moles of one substance participate, you can determine the corresponding moles of another substance tied to it by the balanced equation.
The chemical identity of substances matters: coefficients compare the substances exactly as written (including subscripts in formulas).
Choosing the correct relationship in a reaction
When relating reactants and products, keep these constraints aligned with conservation:
Use the balanced equation for the same reaction (do not mix with a different process)
Use correct formulas (a different formula means a different atom inventory)
Track what’s asked: “produced,” “consumed,” and “left over” are different ideas; conservation applies to atoms, but your calculation targets a specific substance’s amount
Consumed vs produced amounts
The balanced equation relates amounts that react and amounts that form, not necessarily what was initially present. In real mixtures, a reactant can be present in excess; the conservation-based relationship still governs only the portion that actually reacts.
Consistency checks (quick conservation reasoning)
Even before calculating, you can sanity-check relationships using conservation logic:
Mass trends: products may have greater mass than a single reactant if atoms from multiple reactants are incorporated.
Atom accounting: if a product contains a certain element, that element must come from reactants that contain it.
Common pitfalls that break conservation-based reasoning
Using an unbalanced equation, or balancing only some elements
Confusing subscripts and coefficients (changing subscripts changes identity; changing coefficients changes amount)
Skipping the mole step and trying to compare masses directly without accounting for different molar masses
Treating “amount present initially” as identical to “amount that reacted” without chemical justification
FAQ
If a reaction occurs in an open system, gases can escape or be absorbed from air.
Mass is conserved for a closed system containing all reactants and products.
Start by identifying the substance with a known amount and the substance asked for.
Then use the coefficients for those two substances from the balanced equation.
It scales the number of reacting particles by the same factor.
The atom-by-atom “pattern” is unchanged; only the total amount of matter changes.
Coefficients count particles, not grams.
Moles translate mass into particle count using molar mass, keeping the conservation relationship consistent.
Verify that the required atoms exist in the reactants you used.
Also compare molar masses: products incorporating multiple reactants can exceed the mass of a single reactant amount.
Practice Questions
Q1 (1–3 marks) Given the balanced reaction , state the mole amount of required to form of .
Uses coefficient relationship (or ) (1 mark)
Correct amount (1 mark)
Q2 (4–6 marks) For , determine the mass of needed to produce of . Molar mass of is .
States mole ratio from balanced equation (1 mark)
Determines (1 mark)
Uses (or rearranged from ) (1 mark)
Substitutes correctly: (1 mark)
Correct final mass (accept 25.0–25.1 g) with unit (1 mark)

