AP Syllabus focus: ‘Coefficients in balanced equations give proportional relationships among substances; use these ratios with the mole concept for chemical calculations.’
Stoichiometry begins with reading a balanced equation as a quantitative statement. The coefficients are not just “balancing numbers”; they encode fixed proportional relationships that connect reactant and product amounts through the mole concept.
What coefficients mean in a balanced equation
A balanced chemical equation uses coefficients to show how many units of each substance participate in a reaction while conserving atoms. Those coefficients establish the only valid proportional relationships between substances in that reaction.
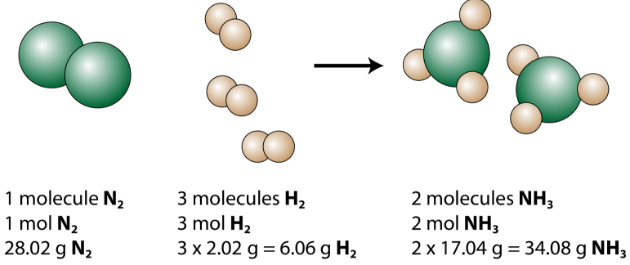
Particle-level representation of a balanced reaction (Haber process), illustrating that coefficients count reacting entities (e.g., 1 + 3 → 2 ). By visualizing discrete molecules, the figure clarifies why mole ratios come from coefficients (counts of whole species) rather than from subscripts inside formulas. Source
Coefficients apply to entire formulas, not individual elements inside a formula.
Coefficients represent relative amounts and can be interpreted as:
numbers of particles (molecules, formula units)
numbers of moles
volumes of gases (only when gases are compared at the same and )
Coefficients do not directly represent masses; mass relationships require molar mass conversions.
Mole ratio (stoichiometric ratio)
A mole ratio is taken from the coefficients of a balanced equation and used as a conversion factor between moles of different substances.
Mole ratio: the ratio of coefficients of two substances in a balanced chemical equation, used to convert between their amounts in moles.
When an equation is correctly balanced, every pair of substances in that equation has a fixed mole ratio that does not change as long as the reaction follows that stoichiometry.
Extracting and using mole ratios correctly
Identifying the correct ratio
To build a valid ratio, match the substances exactly as written in the balanced equation (including their formulas and charges, if ionic species are shown).
Choose the given substance and the target substance.
Read their coefficients as “moles of substance” for stoichiometric purposes.
Write the ratio so that unwanted units cancel (dimensional analysis mindset).
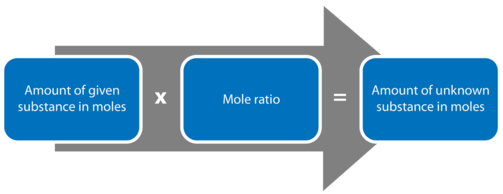
Flowchart showing the core stoichiometry pathway from “amount of given substance (mol)” to “amount of unknown substance (mol)” using a mole ratio derived from coefficients. This diagram reinforces that the coefficient ratio is the essential conversion factor in a mole-to-mole step, independent of mass or particles until additional conversions are applied. Source
Core relationship from coefficients
The coefficients provide the proportional relationship between moles consumed/produced for any extent of reaction.
= moles of substance (mol)
= moles of substance (mol)
= coefficient of in the balanced equation (unitless)
= coefficient of in the balanced equation (unitless)
This relationship is the justification for using coefficients as conversion factors between substances.
Converting from one substance to another (mole-to-mole)
Most stoichiometric links begin as a mole-to-mole conversion, even if the original information is given in grams or particles (those require additional conversions outside the coefficient step).
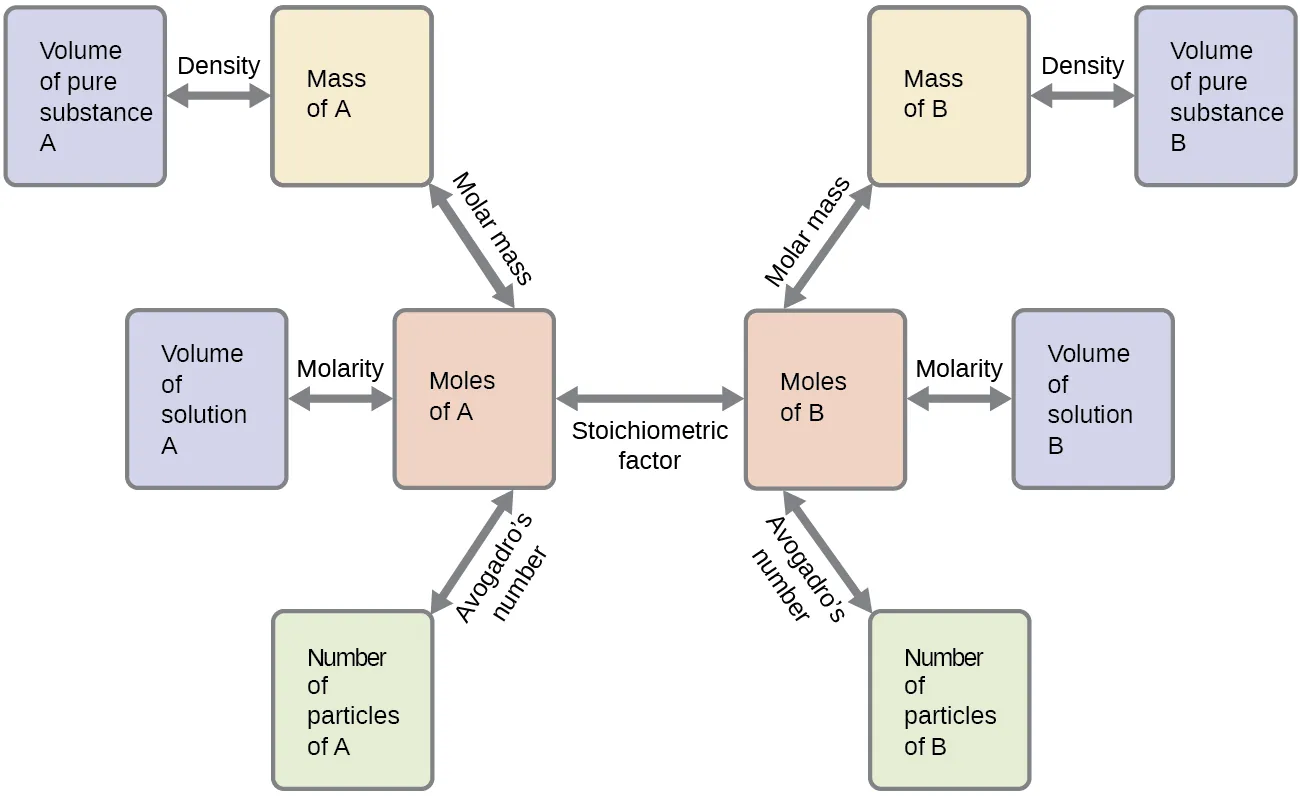
Comprehensive stoichiometry conversion flowchart showing how measurements like mass, volume, and particle count connect through moles, with the “stoichiometric factor” linking to . The diagram emphasizes that coefficients supply only the mole-to-mole bridge; all other links require separate physical/chemical conversion factors (e.g., molar mass, Avogadro’s number). Source
Balance the equation first; unbalanced equations give wrong ratios.
Convert the given amount to moles of the given substance (if it is not already in moles).
Multiply by the appropriate coefficient ratio to get moles of the target substance.
Only after the mole ratio step, convert moles of target to any other requested unit if needed.
A compact way to represent the conversion is:
= moles of target substance (mol)
= moles of given substance (mol)
= coefficient of in the balanced equation (unitless)
= coefficient of in the balanced equation (unitless)
Do not treat this as a new “formula to memorise”; it is a direct consequence of proportionality in the balanced equation.
Coefficients vs subscripts: a common source of errors
A subscript is part of a chemical formula and indicates the composition of one particle (or one formula unit). A coefficient counts how many particles (or moles) are involved.
Subscripts change only if the identity of the substance changes (which would be a different substance).
Coefficients can be changed to balance the equation without changing substance identities.
Never create ratios from subscripts (for example, the “2” in is not a reaction ratio by itself).
Scaling an equation and why ratios stay the same
If every coefficient in a balanced equation is multiplied by the same factor, the equation still represents the same reaction stoichiometry.
The absolute coefficients change, but every ratio of coefficients stays constant.
Therefore, mole ratios are unaffected by scaling (e.g., doubling all coefficients does not change any conversion factor).
This is why balanced equations are usually written with the smallest whole-number coefficients: they are simplest, but not uniquely “more correct” than scaled versions.
Practical interpretation: what the ratio tells you
A mole ratio indicates the relative amount of each substance that reacts or forms when the reaction proceeds according to the balanced equation.
A reactant-to-reactant ratio indicates the proportions required for complete consumption (stoichiometric mixing).
A reactant-to-product ratio indicates the proportional yield of products per mole of reactant consumed.
A product-to-product ratio indicates the proportional formation of products relative to each other.
Correct mole-ratio use depends on selecting the ratio that matches the specific pair of substances relevant to the question’s target quantity.
FAQ
Coefficients describe the reacting proportions in particles (and therefore moles). Molar masses vary by substance and are only used to convert between mass and moles.
A mass ratio can be derived, but only after applying the mole ratio and then converting moles to grams.
Yes. Fractional coefficients still represent valid proportional relationships.
In many AP contexts, equations are presented with whole-number coefficients for convenience, but any set of correctly balanced coefficients produces the same mole ratios.
No. State symbols provide physical context but do not change stoichiometric coefficients.
Mole ratios depend only on the balanced numbers in front of each chemical formula.
A coefficient ratio can be read as a particle-count ratio (molecules/formula units) at the microscopic level.
For example, a 2:1 coefficient ratio means 2 particles of one substance per 1 particle of the other, and the same proportionality holds for moles.
Write the conversion factor so the unit you want cancels properly.
Put the given substance in the denominator.
Put the target substance in the numerator.
Use the matching coefficients with those substances.
Practice Questions
Q1 (1–3 marks) For the balanced reaction , state the mole ratio of to and the mole ratio of to .
(1 mark)
(1 mark)
Ratios correctly taken from coefficients and matched to the order asked (1 mark)
Q2 (4–6 marks) Consider the balanced equation . (a) Write the mole ratio of to . (b) Write the conversion factor (as a fraction) that converts moles of to moles of . (c) If moles of react completely, express the moles of formed in terms of .
(a) (2 marks: correct coefficients (1), correct order (1))
(b) Conversion factor or simplified with units shown as a mole ratio (2 marks: correct substances (1), correct coefficients orientation for cancellation (1))
(c) Moles of (2 marks: identifies 1:1 ratio from coefficients (1), correct expression in terms of (1))

