AP Syllabus focus: ‘For an elementary step, the rate law can be inferred from the collision stoichiometry (molecularity) of the reacting particles.’
Reaction rates connect particle-level events to measurable concentration changes. For elementary steps, the balanced step itself reveals the rate law form, because it represents a single collision or decomposition event.
Elementary Steps and Molecularity
What “elementary” means
Elementary step: A single, microscopic reaction event that occurs in one step (one collision or one molecule breaking apart), with no hidden intermediate steps.
Because an elementary step is a single event, its reactant coefficients describe how many particles must participate simultaneously.
Molecularity: the collision stoichiometry
Molecularity: The number of reactant particles that participate in an elementary step (e.g., 1, 2, or 3 particles).
Molecularity applies only to one elementary step (not to an overall balanced equation that may represent many steps).
Unimolecular step: one reactant particle involved (often a rearrangement or decomposition).
Bimolecular step: two reactant particles collide.
Termolecular step: three reactant particles participate in a single event (uncommon in practice, but the definition still holds).
Rate Laws for Elementary Steps
Core idea (AP exam-critical)
For an elementary step, the rate law exponents match the stoichiometric coefficients of the reactants in that step. This is the direct meaning of “the rate law can be inferred from the collision stoichiometry (molecularity).”
A general way to express an elementary-step rate law is:
= reaction rate (typically )
= rate constant (units depend on )
= reactant molar concentrations ()
= reaction orders for and in this elementary step (equal to reactant coefficients in the step)
This matching rule is valid because the probability of the elementary event depends on how often the required particles meet (or how often a single particle activates, for unimolecular steps).
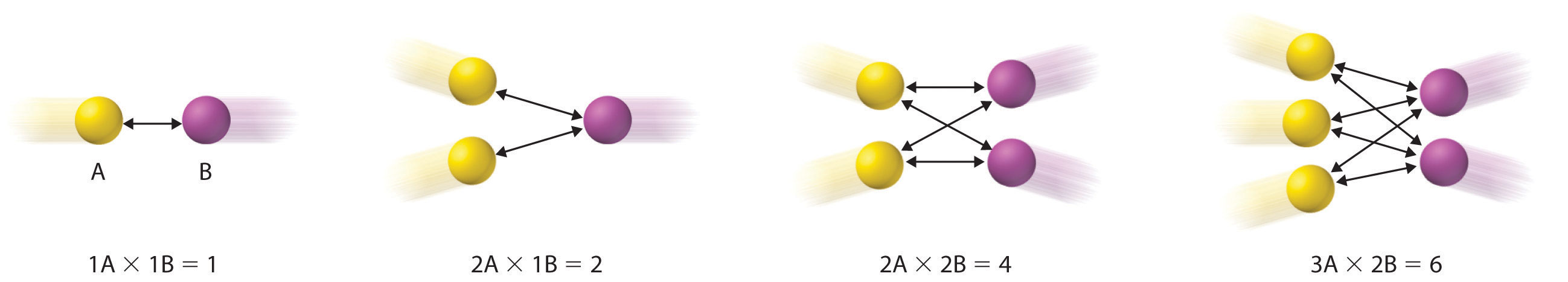
Diagram illustrating that the number of effective – collisions scales with the product of the numbers (and thus concentrations) of and . This provides a particle-collision justification for elementary-step rate laws such as , where each exponent reflects how many of that reactant must participate in the event. Source
Recognising patterns quickly
Unimolecular: products
Rate law form: Rate (first order in )
Bimolecular: products
Rate law form: Rate
Bimolecular: products
Rate law form: Rate
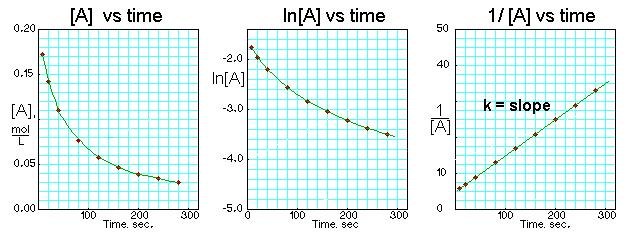
Three time-based plots for a second-order process: vs. and vs. are curved, while vs. is linear with slope . The figure reinforces what “second order in ” looks like in common kinetics plots and how the squared concentration dependence leads to a linear transformation. Source
Termolecular: products
Rate law form: Rate
Termolecular: products
Rate law form: Rate
What You Can and Cannot Infer
What you can infer (only for an elementary step)
The orders with respect to each reactant in that step.
The overall order for that step as the sum of exponents (e.g., bimolecular steps are second order overall).
What you cannot infer (from an overall equation alone)
You generally cannot write a correct experimental rate law by copying coefficients from an overall balanced reaction, because the overall equation may summarise multiple elementary events. Only when you are explicitly told a reaction is an elementary step is coefficient-copying justified.
Common student pitfalls
Treating “molecularity” as the same thing as “overall reaction order” for a full reaction (it is not; molecularity is step-specific).
Assigning molecularity to products (molecularity counts reactant particles only in the elementary step as written).
Forgetting that a coefficient of 2 in an elementary step implies a squared concentration dependence in the step’s rate law.
FAQ
Overall equations may combine several microscopic events. Molecularity describes the number of particles in a single event, so it is only meaningful when no hidden steps are bundled together.
Count $M$ as a reactant particle for molecularity and include it in the elementary-step rate law. Although $M$ is regenerated, it participates in the collision that enables stabilisation.
Not in the elementary-step interpretation used in AP Chemistry. Fractional orders can arise in overall rate laws from multi-step behaviour, but that goes beyond inferring directly from molecularity.
If an experimentally determined rate law does not match the coefficients of the proposed one-step equation (e.g., coefficient 2 but first order observed), the process cannot be that single elementary step as written.
For an elementary step, yes: $2A \rightarrow$ products implies a bimolecular event involving two $A$ particles. For an overall equation, a “2” may simply be stoichiometric bookkeeping and does not prove a two-particle collision.
Practice Questions
(2 marks) The step is stated to be elementary. Write the rate law expression.
(1)
Correct orders match coefficients; included (1)
(5 marks) For each elementary step below, (i) state the molecularity and (ii) write the rate law.
(a)
(b)
(c) (where is a third-body collider)
(a) unimolecular (1); (1)
(b) bimolecular (1); (1)
(c) termolecular (1); (1)

