AP Syllabus focus: ‘Radioactive decay is an important real‑world example of first‑order kinetics.’
Radioactive decay provides a clean, data-rich example of first-order behavior because its rate depends only on how many unstable nuclei are present. This makes decay useful for modelling exponential change and interpreting half-life information.
Radioactive decay as a first-order process
What is decaying, and what is “rate”?
In radioactive decay, an unstable nucleus transforms into a more stable nucleus (often producing particles and/or electromagnetic radiation). The key kinetic observation is that each nucleus “chooses” to decay independently and randomly.
Radioactive decay: A spontaneous nuclear process in which an unstable nucleus changes into a different nucleus, emitting radiation; the probability of decay per nucleus is constant at a given isotope.
Because the probability of decay per nucleus is constant, a larger sample (more unstable nuclei) produces more decays per second. As the sample shrinks, the decay rate slows proportionally.
The first-order relationship
A first-order process is one in which the rate is directly proportional to the amount present. For radioactive decay, the “reactant” is the number of undecayed nuclei, often written as N. The proportionality constant is the decay constant, k, and it behaves like a first-order rate constant.
Decay constant (k): The proportionality constant in radioactive decay that links the decay rate to the number of undecayed nuclei; its units are inverse time (such as per second).
This proportionality leads to exponential decrease over time, which is the hallmark of first-order kinetics.
= number of undecayed nuclei (count)
= decay constant (time)
= initial number of nuclei (count)
= number of nuclei at time (count)
= elapsed time (time)
= half-life (time)
Half-life: why it stays constant
The half-life is the time required for half of the unstable nuclei in a sample to decay.
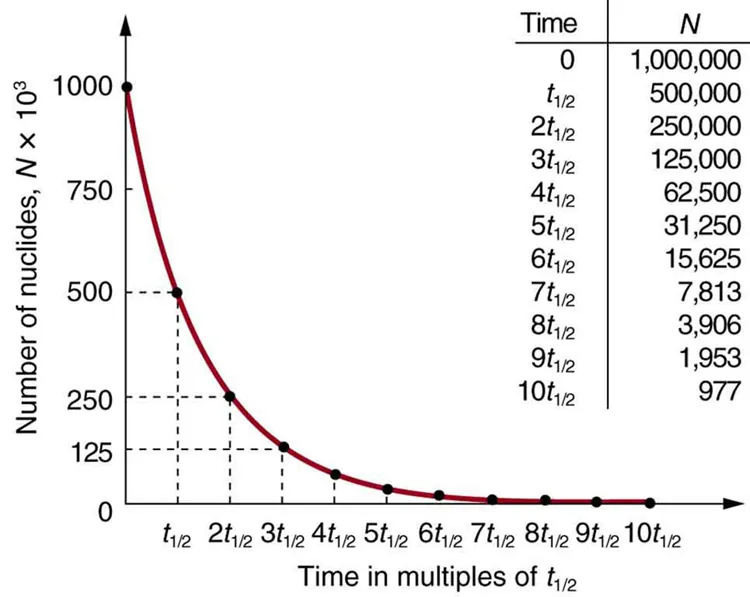
This plot shows the number of radioactive nuclei decreasing exponentially with time, with the time axis marked in successive half-lives. It makes the “halving” pattern visually explicit: after each interval, the remaining amount drops by a factor of 2, even though the absolute amount lost each interval becomes smaller. Source
In a first-order process, the fraction lost per equal time interval is constant, even though the absolute number lost decreases as the sample gets smaller. That is why half-life does not depend on how much isotope you start with.
Half-life: The time required for the number of undecayed nuclei in a sample to decrease to one-half of its current value.
Connecting decay to measurable laboratory quantities
In practice, you rarely count nuclei directly. Instead, you measure activity, the number of decay events per unit time. Activity is proportional to N, so it also decreases exponentially and follows first-order behavior.
If activity is plotted on a natural-log scale versus time, a straight line indicates first-order decay.
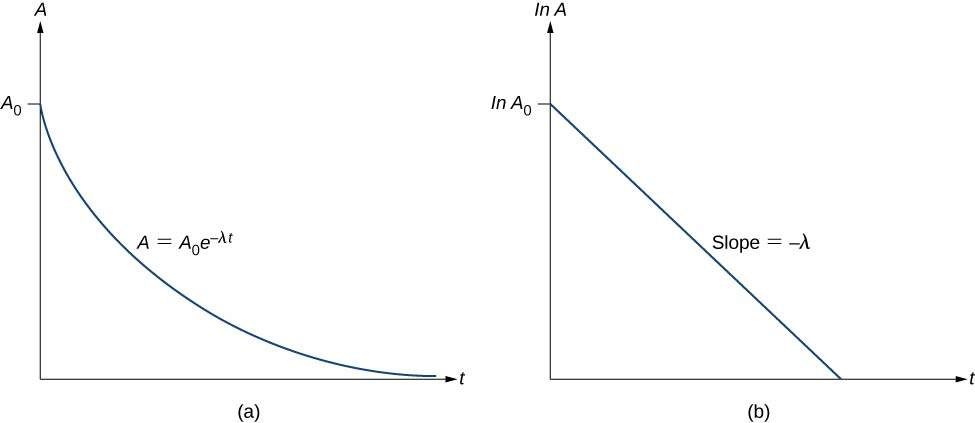
Panel (a) shows activity decreasing exponentially over time according to . Panel (b) shows that plotting versus produces a straight line with slope , which is the graphical signature of first-order kinetics for radioactive decay. Source
The slope of that line is negative and has magnitude k.
The intercept corresponds to the initial activity (or initial amount), depending on what is plotted.
What stays the same (and what does not)
Radioactive decay is largely unaffected by typical chemical or physical changes (temperature, pressure, phase, bonding), because it is a nuclear process. What determines the kinetics is the identity of the isotope and its decay pathway, captured by k and the half-life.
Constant for a given isotope: k, half-life, and the functional form of the decay curve.
Changes with time for a given sample: N and activity, both decreasing in a first-order (exponential) manner.
FAQ
They play analogous kinetic roles, but differ in origin. For decay, $k$ reflects nuclear instability and is largely independent of chemical environment; chemical rate constants depend strongly on temperature and molecular interactions.
Half-life is governed by nuclear energy barriers and quantum probabilities, not molecular collisions. Ordinary temperature changes affect electron energies far more than nuclear structure, so $k$ and $t_{1/2}$ remain effectively unchanged.
Activity is the true number of decays per second. Detectors often measure a lower count rate because of:
limited efficiency
geometry (not all radiation reaches the detector)
absorption by materials
A calibration factor is needed to relate count rate to activity.
If an isotope decays by more than one pathway, the total decay is still first-order in the parent nuclei. The overall $k$ equals the sum of pathway-specific decay constants, and branching ratios describe how products are distributed.
First-order kinetics ensures predictable time evolution, but dating relies on accurate parent/daughter amounts. Adding/removing parent or daughter isotopes (or loss by diffusion) shifts the ratio, giving an apparent age that is too old or too young.
Practice Questions
(2 marks) Explain why radioactive decay is described as a first-order process.
States that the decay rate is proportional to the number of undecayed nuclei present (1).
Links this proportionality to exponential decrease/constant half-life or a constant probability per nucleus (1).
(5 marks) A radioactive isotope has decay constant . Describe how you could use activity–time data to confirm first-order kinetics and determine . Include the key plot and what features of the plot you would analyse.
Identifies activity as proportional to number of undecayed nuclei, so it follows the same kinetics (1).
States to plot against time (or equivalent linearisation) (1).
Predicts a straight line for first-order decay (1).
States the gradient is (or magnitude gives ) (1).
Mentions intercept corresponds to initial activity (or allows checking consistency with initial value) (1).

