AP Syllabus focus: ‘Most collisions do not react; successful collisions need enough energy to overcome activation energy and the correct orientation for bond rearrangement.’
Reaction rates depend on which particle collisions actually produce products. Collision theory explains why many collisions fail: particles may collide with too little energy or in an unproductive orientation, even when concentration is high.
Effective collisions require energy and orientation
A collision between reactant particles is only effective if it can initiate the necessary bond breaking and bond making. Two conditions must be satisfied at the instant of collision:
Energy condition: the colliding particles must have enough kinetic energy to surmount an energy barrier.
Orientation condition: the particles must collide with the correct relative geometry so the right atoms/regions interact to allow bond rearrangement.
When either condition fails, the particles may bounce apart, transfer energy without reacting, or form no productively connected arrangement.
Energy condition: overcoming the activation energy barrier
Activation energy (): The minimum energy barrier that reactant particles must overcome during a collision for the reaction to proceed to products.
In collision terms, is the threshold separating mostly nonreactive collisions from potentially reactive ones. It is not “stored” in molecules as a fixed amount; instead, it refers to an energetic requirement for reaching a short-lived, high-energy arrangement of atoms during the collision.
The energy requirement can be stated as a simple inequality:
= kinetic energy available in the collision (especially along the line of impact), J
= activation energy barrier for that reaction pathway, J
Even if particles collide frequently, only a fraction of collisions have at or above . Raising temperature increases the fraction of particles with sufficient kinetic energy, so a larger fraction of collisions can satisfy the energy condition.
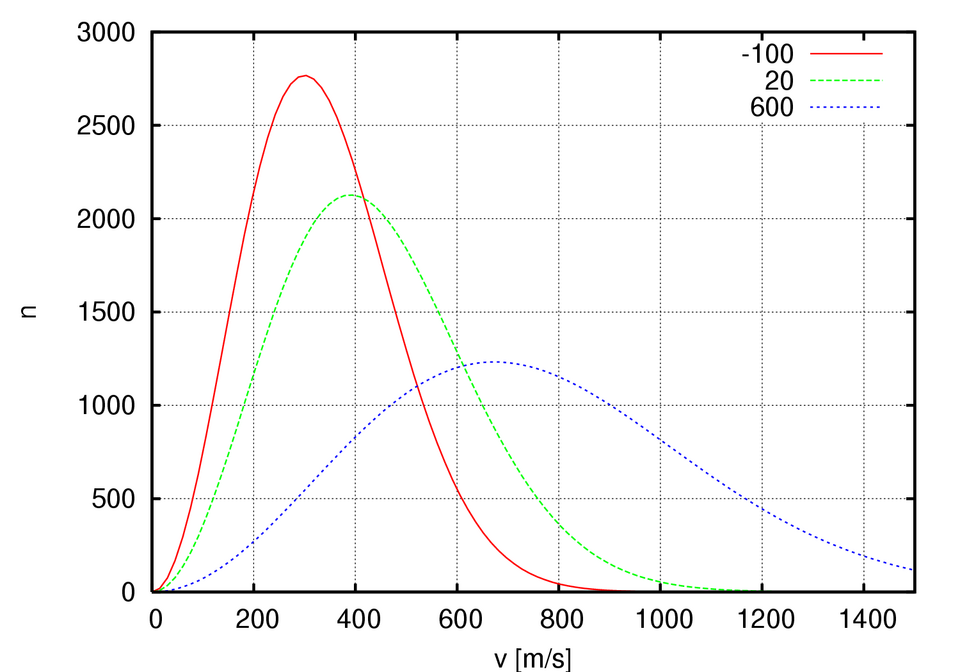
Maxwell–Boltzmann energy distribution curves at multiple temperatures, showing how the peak flattens and shifts as temperature increases. The key collision-theory takeaway is that the high-energy tail grows at higher , increasing the fraction of particles capable of meeting an activation-energy threshold. Source
Orientation condition: correct geometry for bond rearrangement
Orientation requirement (steric requirement): The need for reactant particles to collide with a specific relative alignment so that electron density can be redistributed and the correct bonds can break/form.
Energy alone does not guarantee reaction.
For many reactions, only certain parts of molecules are reactive (specific atoms, functional groups, or regions of partial charge). A collision must bring these reactive regions into contact in a way that supports:
Proper approach direction (e.g., “end-on” vs “side-on” for certain bonds)
Productive orbital/electron-density overlap (allowing new bonding interactions)
Simultaneous weakening of old bonds while new bonds begin to form
As molecular complexity increases, there are more ways to collide incorrectly, so the orientation requirement can strongly limit the reaction rate even when collisions are energetic.
Putting both requirements together
Most collisions do not react because failing either requirement is enough to prevent product formation:
High-energy but wrong orientation: particles collide hard, but the reactive sites do not align, so no bond rearrangement occurs.
Correct orientation but low energy: particles align well, but lack the energy needed to cross the barrier, so they separate unchanged.
Both satisfied: an effective collision occurs, allowing the system to pass the barrier and proceed toward products.
A useful conceptual way to think about rate is that it scales with:
how often particles collide, and
what fraction of those collisions are both energetic enough and properly oriented.
Orientation effects are sometimes summarised by a steric factor (a probability of correct alignment), but for AP Chemistry the key idea is qualitative: reactions can be slow because effective collisions are rare, not because collisions themselves are rare.
FAQ
No. $E_a$ is the barrier to reach the high-energy arrangement during a collision, whereas the overall energy change (enthalpy change) compares products and reactants.
A reaction can be exothermic and still have a large $E_a$.
Orientation matters most when bonding changes must occur at specific, localised sites.
Examples include cases where only one end of a polar molecule is reactive, or where a bond must be approached from a particular direction to allow electron density to shift correctly.
Solvent molecules can hinder or help alignment:
They can form solvation shells that block reactive sites.
They can stabilise certain approaches via intermolecular attractions.
Increased viscosity can reduce the ability of molecules to reorient during encounters.
Sometimes. If the reacting functional groups meet properly, the rest of the molecule may be a “spectator”.
However, bulky groups can physically block access to reactive sites, effectively making correct alignment less probable.
They may compare rates for structurally similar reactants:
If adding bulk near a reactive site greatly slows the reaction without changing $E_a$ much, that suggests orientation/steric effects.
If rate changes track mainly with temperature sensitivity, that suggests the energy threshold is more limiting.
Practice Questions
Question 1 (2 marks) State two distinct reasons, based on collision theory, why most collisions between reactant particles do not result in reaction.
1 mark: Colliding particles often have insufficient energy to overcome the activation energy, .
1 mark: Collisions often occur with incorrect orientation so the required bond rearrangement cannot occur.
Question 2 (5 marks) A student increases the temperature of a reacting system and observes a faster reaction rate. (a) Using activation energy ideas, explain why the rate increases. (3 marks) (b) Explain why the reaction may still be slow even at the higher temperature, referring to orientation requirements. (2 marks)
1 mark: A higher temperature means particles have greater average kinetic energy.
1 mark: A larger fraction of collisions have .
1 mark: Therefore a greater proportion of collisions are effective, increasing the rate. (b)
1 mark: Collisions must have the correct relative orientation for bond rearrangement.
1 mark: Many collisions remain incorrectly oriented, so not all energetic collisions lead to reaction.

