AP Syllabus focus: ‘The Maxwell–Boltzmann distribution shows particle energy spread; it helps estimate the fraction of collisions energetic enough to react and how this fraction increases with temperature.’
Chemical particles at a given temperature do not all move with the same energy. The Maxwell–Boltzmann distribution describes this spread and explains why heating a system can dramatically increase reaction rates.
Maxwell–Boltzmann Distribution: What the Curve Means
Energy is distributed, not uniform
Maxwell–Boltzmann distribution: A probability distribution showing how particle energies are spread across a range at a given temperature, with many particles at moderate energies and fewer at very low or very high energies.
On a typical plot of “number (or fraction) of particles” vs. kinetic energy:
The curve starts near zero at very low energy (few particles are nearly “still”).
It rises to a peak at the most probable energy.
It falls off with a long right-hand tail (a small fraction have very high energy).
The area under the curve represents the total number of particles (or total fraction = 1), so changing temperature reshapes the curve but does not change the total area.
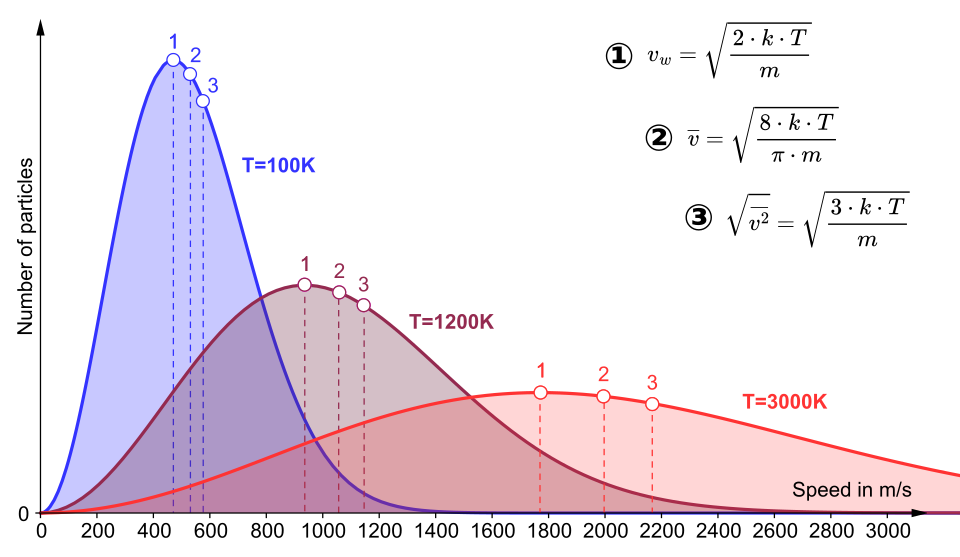
Maxwell–Boltzmann speed distributions plotted for several different temperatures, showing the peak shifting to higher speeds and becoming broader as increases. The labeled markers highlight characteristic speeds (e.g., most probable vs. average), making it easier to connect “typical” particle motion to the overall distribution shape. Source
Temperature and average kinetic energy
A higher temperature means a higher average kinetic energy, and the distribution shifts accordingly.
= average translational kinetic energy per mole of particles, J mol
= gas constant, J mol K
= absolute temperature, K
This relationship supports the core idea: increasing raises typical particle energies, which changes collision outcomes.
Connecting the Distribution to Reaction Rate
The activation energy threshold
Activation energy (): The minimum energy reactant particles must have during a collision for the collision to be energetic enough to lead to reaction.
On a Maxwell–Boltzmann plot, is visualised as a vertical line at some energy value.
Only particles with energies to the right of this line are candidates for energetically successful collisions.
“Fraction of collisions energetic enough to react”
The specification focus is the fraction of particles (and thus collisions) with energy , which is represented by:
The area under the curve to the right of .
If that area is small, most collisions are too low-energy to proceed, even if particles collide frequently.
Why Increasing Temperature Increases Rate
How the curve changes when temperature increases
When temperature increases:
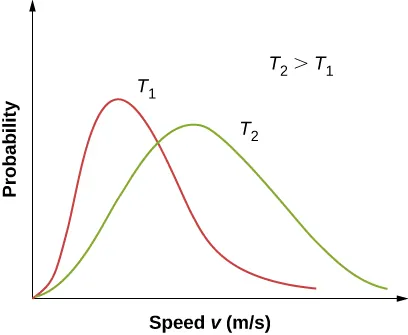
OpenStax figure comparing Maxwell–Boltzmann speed distributions at two temperatures, with the higher-temperature curve broadened and shifted to higher speeds. This visual supports the idea that heating increases the fraction of particles in the high-energy (right-tail) region, even though the total area under each curve remains the same. Source
The peak becomes lower (the distribution spreads out).
The curve shifts to higher energy (more particles at higher energies).
The right-hand tail becomes “fatter”, increasing the area beyond .
Crucially, even a moderate temperature increase can cause a large relative increase in the fraction beyond because the tail region is very sensitive to temperature.
What this implies for collisions
Raising temperature generally increases reaction rate because:
More particles have enough energy to overcome (more energetically effective collisions).
Particles move faster on average, increasing collision frequency (though the distribution-based increase in the fraction is often the dominant conceptual point).
Interpreting “effective” without overextending
The Maxwell–Boltzmann distribution addresses the energy requirement for a successful collision. It does not, by itself, guarantee reaction from every high-energy collision; it supports estimating how the fraction of sufficiently energetic collisions changes with temperature, which aligns with observed rate increases.
FAQ
Energies cannot be negative, so the distribution is bounded at low energy but unbounded at high energy.
A few particles can have very high energies, producing the long right-hand tail.
The most probable energy is at the peak (mode): the single energy value with the highest frequency.
The average energy is larger than the most probable energy because the high-energy tail pulls the mean to the right.
The area beyond $E_a$ often starts small.
Heating mainly boosts the high-energy tail, so the relative increase in particles with $E \ge E_a$ can be large even if $T$ changes modestly.
It is most directly introduced for gases, where translational motion is clear.
The core idea—particles having a spread of energies at a given $T$—is also used qualitatively for liquids/solutions when discussing energetic thresholds.
Energy may be shown per particle (J) or per mole (kJ mol$^{-1}$); the curve’s interpretation is the same if $E_a$ uses consistent units.
Temperature must be in kelvin when relating energy scales to $T$.
Practice Questions
A reaction has activation energy . On a Maxwell–Boltzmann distribution diagram, explain what the area to the right of represents and how it changes when temperature increases.
Identifies area right of as the fraction (or number) of particles/collisions with energy (1).
States that increasing temperature increases this area/fraction (1).
Describe how the Maxwell–Boltzmann distribution changes when temperature increases and use this to explain why reaction rate increases. Your answer must refer to the peak, the spread of energies, and the fraction of particles above .
Peak becomes lower (1).
Distribution becomes broader/more spread out (1).
Distribution shifts to higher energy (1).
Fraction/area above increases (1).
Links increased fraction above to more energetically successful collisions and a higher rate (1).

