AP Syllabus focus: ‘The enthalpy change of a reaction is the heat released (negative ΔH) or absorbed (positive ΔH) at constant pressure.’
Thermochemistry often reports energy changes as enthalpy changes, because many reactions occur in open containers at (approximately) constant atmospheric pressure. Correctly interpreting the sign and meaning of ΔH is essential for connecting measurements to chemical change.
Enthalpy and constant-pressure heat
Chemical reactions can exchange energy with their surroundings in different forms, but AP Chemistry commonly focuses on heat flow at constant pressure (lab bench conditions).
Enthalpy (H): A state function that tracks a system’s energy content in a way that makes constant-pressure heat flow easy to interpret; for processes at constant pressure, changes in enthalpy correspond to heat transferred.
At constant pressure, the heat that flows into or out of the reacting system is directly tied to the reaction’s enthalpy change. This is why reaction energy changes are usually reported as ΔH (instead of ΔE).
= Heat absorbed by the system at constant pressure (J or kJ)
= Enthalpy change of the system for the process (J or kJ)
This relationship is an interpretation tool: it tells you that a measured heat flow under constant-pressure conditions can be labelled as the reaction’s ΔH (with the correct sign).
What ΔH means for a reaction
Reaction enthalpy is the enthalpy of products compared to reactants, evaluated for the same overall process and conditions.
Enthalpy change of reaction (ΔH): The difference between the enthalpy of the products and the enthalpy of the reactants for the reaction as written, under specified conditions.
A common way to express this idea is:
= Enthalpy change for the reaction as written (kJ)
= Enthalpy of products (kJ)
= Enthalpy of reactants (kJ)
Because H is a state function, ΔH depends only on initial and final states (reactants and products under the stated conditions), not on the path taken.
Sign conventions: linking ΔH to heat absorbed or released
Interpreting the sign of ΔH is central to the syllabus statement.
ΔH < 0 (negative): the reaction is exothermic at constant pressure
The system releases heat to the surroundings.
The surroundings gain thermal energy; the reaction mixture often feels warmer (though temperature change depends on amounts and heat capacities).
ΔH > 0 (positive): the reaction is endothermic at constant pressure
The system absorbs heat from the surroundings.
The surroundings lose thermal energy; the reaction mixture often feels cooler.
A quick language check:
“Heat released” refers to heat leaving the system (so ΔH is negative).
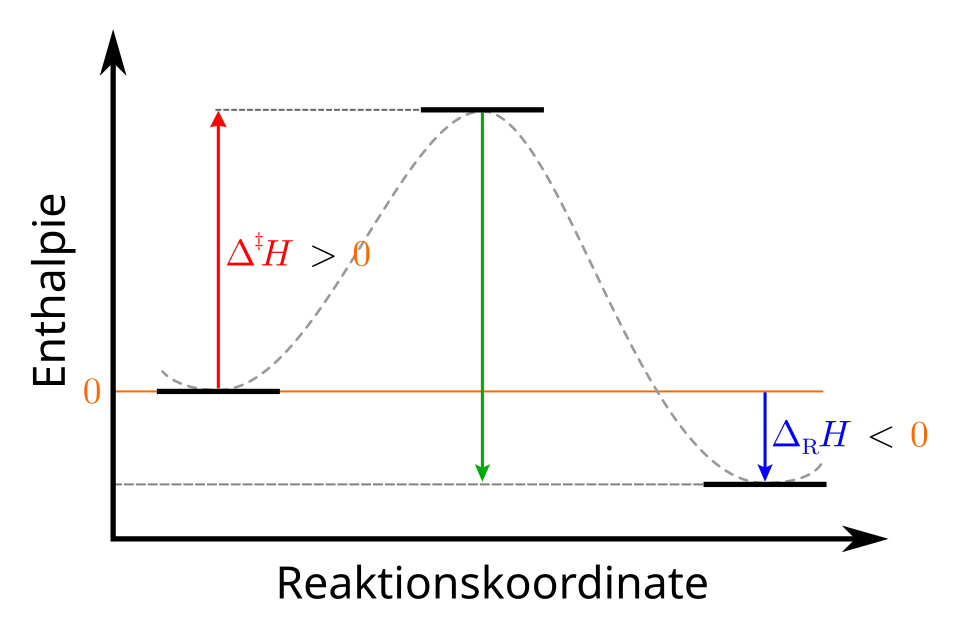
Enthalpy (reaction coordinate) diagram for an exothermic reaction. The products lie at a lower enthalpy than the reactants, and the vertical gap corresponds to the reaction enthalpy change, , which is interpreted as heat released by the system at constant pressure. Source
“Heat absorbed” refers to heat entering the system (so ΔH is positive).
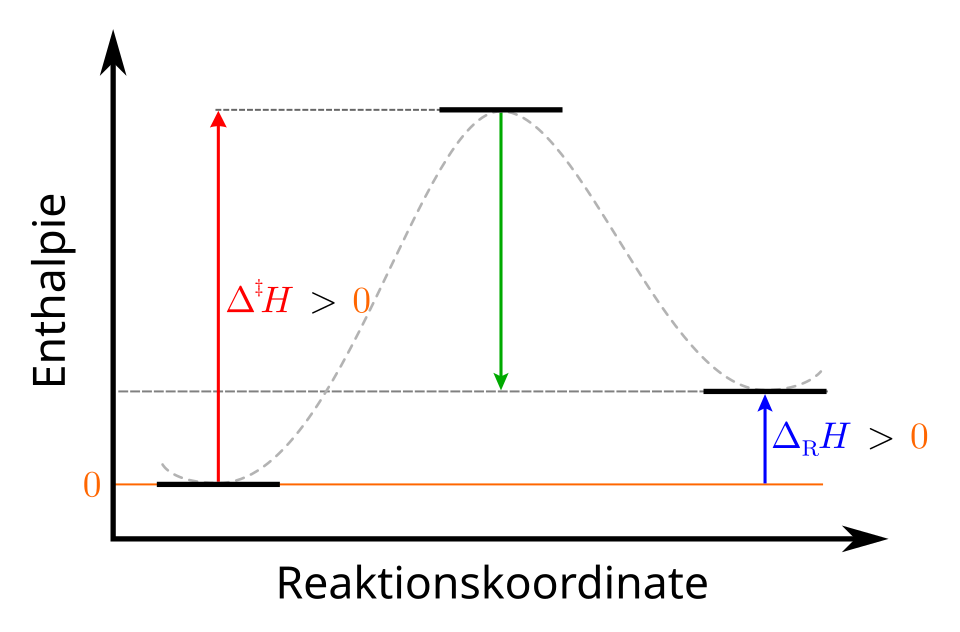
Enthalpy (reaction coordinate) diagram for an endothermic reaction. The products lie at a higher enthalpy than the reactants, so the enthalpy change is positive, , consistent with the system absorbing heat under constant-pressure conditions. Source
Why “constant pressure” matters (and what it implies)
Many reactions occur in cups, beakers, or flasks open to the atmosphere, so pressure is approximately constant. Under these conditions:
Some energy may go into expansion work (the system pushing back the atmosphere as gases form or warm).
Enthalpy is defined to account for this constant-pressure situation, making the practical heat term chemists measure and report as ΔH.
Important implications for interpretation:
The phrase “enthalpy change” in AP Chemistry typically means “heat flow at constant pressure,” not necessarily “total energy change in all forms.”
ΔH is reported for the reaction as written: if the reaction stoichiometry were different, the numerical value of ΔH would correspond to that written amount of reaction (without changing the sign logic).
FAQ
Most school experiments are effectively at constant atmospheric pressure, so the heat measured corresponds to $q_p$, which equals $\Delta H$.
Not always. The temperature change depends on quantities, dilution, and heat capacity of the solution/calorimeter, even if heat is released overall.
It means the stated $\Delta H$ matches the exact balanced equation’s stoichiometric amounts; changing coefficients would scale the enthalpy change accordingly.
No. Expansion/compression work can occur; enthalpy is defined so that, at constant pressure, this is naturally included when relating heat to $\Delta H$.
They are the same at constant pressure: $q_p>0$ implies $\Delta H>0$ (heat absorbed), and $q_p<0$ implies $\Delta H<0$ (heat released).
Practice Questions
(2 marks) A reaction carried out in an open polystyrene cup has . State what this sign tells you about heat transfer between system and surroundings at constant pressure.
States reaction is exothermic / heat released (1)
States heat flows from system to surroundings at constant pressure (1)
(5 marks) Explain what represents for a reaction at constant pressure. Your answer should include (i) a statement linking to , (ii) what the sign of indicates, and (iii) what comparison of enthalpy values defines .
Correct link at constant pressure (1)
Defines as products minus reactants, e.g. (1)
States means exothermic / heat released (1)
States means endothermic / heat absorbed (1)
Correctly identifies direction of heat flow for at least one sign (system to surroundings for negative, or surroundings to system for positive) (1)

