AP Syllabus focus: ‘In exothermic reactions, thermal energy is transferred to the surroundings; in endothermic reactions, thermal energy is transferred from the surroundings.’
Chemical reactions can warm up or cool down what they are in contact with. Correctly classifying a reaction as exothermic or endothermic depends on tracking the direction of thermal energy transfer between the reacting chemicals and everything else.
The system and the surroundings
To discuss energy transfer, you must define what you are focusing on (the reaction) versus what is outside it.
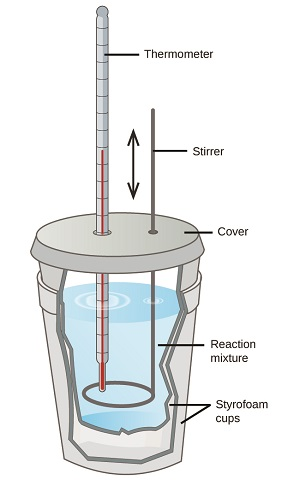
A labeled “coffee-cup calorimeter” schematic showing an insulated cup setup with a lid, thermometer, and stirrer surrounding the reaction mixture. This supports the system–surroundings framework by making clear that the temperature you record is the temperature of the calorimeter contents (the surroundings that are in thermal contact with the reaction), not the reacting bonds directly. Source
System: The part of the universe chosen for study; in this context, the reacting chemicals (and sometimes the solution they are dissolved in).
The key idea is that temperature changes you observe are usually changes in the surroundings (like the solution, container, or air), caused by heat flowing to or from the reaction.
Surroundings: Everything outside the system that can exchange energy with it (for example, the water in a cup, the thermometer, and the air).
Exothermic reactions: thermal energy flows out to the surroundings
A reaction is exothermic when the reaction (system) releases thermal energy to what surrounds it.
Exothermic reaction: A reaction in which thermal energy is transferred from the system to the surroundings.
What you typically observe in the lab (because the surroundings are easier to measure than the system directly):
The reaction container or solution feels warmer.
The measured temperature of the surroundings increases.
Heat is leaving the reacting chemicals and entering the nearby materials.
Important interpretation points:
The system loses thermal energy, but the surroundings gain thermal energy.
If the surroundings are mainly water (common in lab setups), even a modest heat release can produce a measurable temperature rise because the water is in direct thermal contact with the reaction.
Endothermic reactions: thermal energy flows in from the surroundings
A reaction is endothermic when the reaction (system) absorbs thermal energy from its surroundings.
Endothermic reaction: A reaction in which thermal energy is transferred from the surroundings to the system.
Common observations:
The container or solution feels cooler.
The measured temperature of the surroundings decreases.
Heat is entering the reacting chemicals from the nearby materials.
Important interpretation points:
The system gains thermal energy, but the surroundings lose thermal energy.
If the reaction mixture is surrounded by a large thermal reservoir (like a big beaker of water or the air), the temperature change you measure may be smaller than expected because the surroundings can supply or absorb energy without changing temperature much.
Energy conservation and sign conventions for heat flow
Heat transfer between system and surroundings is constrained by conservation of energy: energy lost by one is gained by the other. A consistent sign convention helps you avoid mixing up “who gets warmer” with “who gains energy.”
= Heat absorbed by the system (J or kJ); negative for exothermic reactions and positive for endothermic reactions.
= Heat absorbed by the surroundings (J or kJ); positive when the surroundings warm and negative when they cool.
Practical meaning:
Exothermic: surroundings warm, so the surroundings gain heat; the system must lose the same amount of heat.
Endothermic: surroundings cool, so the surroundings lose heat; the system must gain the same amount of heat.
Avoiding common mistakes when classifying reactions
Misclassification usually comes from mixing up temperature change with “energy of the reaction” without identifying system vs. surroundings.
A temperature increase you measure is evidence that the surroundings gained thermal energy, so the reaction is exothermic.
A temperature decrease you measure is evidence that the surroundings lost thermal energy, so the reaction is endothermic.
Always state the direction clearly: “thermal energy transferred to the surroundings” (exothermic) or “thermal energy transferred from the surroundings” (endothermic).
“Heat produced” and “heat released” refer to energy leaving the system, not appearing from nowhere; it must enter the surroundings.
FAQ
Yes, if you redefine the system boundary.
For example, including the solvent as part of the system can change what you call “surroundings,” altering which temperature change you focus on.
The heat may be spread through a large mass of surroundings.
A small heat release into a large volume (high heat capacity) can produce a negligible temperature change.
“Heat released” describes energy transfer; “temperature increase” is a consequence that depends on the surroundings.
A given heat transfer can cause different temperature changes in different surroundings.
The surroundings can already store thermal energy due to particle motion.
Heat flow is a redistribution of existing energy, not the creation of energy by the surroundings.
Not necessarily.
Some endothermic reactions proceed by drawing heat from immediate surroundings until they cool; whether they continue depends on conditions such as mixing and heat exchange with the environment.
Practice Questions
(2 marks) A student mixes two aqueous solutions and observes the temperature of the mixture rise by . Classify the reaction as exothermic or endothermic, and state the direction of thermal energy transfer.
Exothermic (1)
Thermal energy transferred to the surroundings / from system to surroundings (1)
(5 marks) Define “system” and “surroundings” for a reaction carried out in a polystyrene cup. The temperature of the solution decreases during the reaction. Use correct terminology and sign conventions to explain what this implies about and .
System defined as reacting chemicals (or reaction mixture) chosen for study (1)
Surroundings defined as everything outside system (e.g., cup, air, thermometer, solution if not included in system) (1)
Temperature decrease indicates surroundings lose thermal energy; reaction is endothermic (1)
is positive (heat absorbed by system) (1)
is negative and equal in magnitude to (1)

