AP Syllabus focus: ‘Estimate energy required by summing average bond energies for all bonds broken in the reactant molecules.’
Chemical reactions involve rearranging atoms, which requires breaking existing bonds before new ones can form. This page focuses on estimating the energy input for a reaction by adding up the bond energies of bonds broken in reactants.
Core idea: energy in comes from breaking bonds
Breaking a chemical bond always requires an input of energy to overcome the electrostatic attractions holding atoms together.
In thermochemistry problems, that required energy is approximated using tabulated average bond energies.
Average bond energy (bond enthalpy)
Bond energy values provided on data sheets are typically averages measured over many compounds, so they are best for estimates, not exact results.
Average bond energy (average bond enthalpy): The average energy required to break one mole of a specific type of bond in gaseous molecules, producing separated gaseous atoms.
Because bond energies are defined for gas-phase bond breaking, the method conceptually treats reactant molecules as if their bonds are broken into gaseous atoms (even if the real reaction occurs in solution).
What you are estimating in this subsubtopic
You are finding energy required to break all bonds in the reactant molecules that must be broken as reactants convert to products. This corresponds to the “energy in” portion of a bond-enthalpy approach.
The sum-of-bonds-broken method
The key steps are about correct bond counting and correct use of tabulated values.
= total energy required to break bonds in the reactants, in (for the reaction as written)
= number of bonds of type that are broken (unitless count, based on the balanced equation)
= average bond energy for bond type , in (per mole of that bond)
This “sum” is done only over bonds broken in the reactants, not bonds formed in the products.
How to identify and count bonds broken (without calculating)
1) Use the balanced chemical equation as your bond-counting map
The balanced equation sets the relative number of molecules reacting, which directly scales the number of each bond type you will break.
Apply stoichiometric coefficients to every bond count.
If a coefficient doubles a reactant, it doubles every relevant bond of that reactant that is broken.
2) Determine which specific bonds are present in each reactant
Bond energies are listed by bond type (single, double, triple; and which atoms are connected), so you must identify the bonding in each reactant molecule.
Distinguish single vs. double vs. triple bonds (e.g., C–C vs. C=C vs. C≡C).
Treat different environments as the same bond type only if the table does (bond energies are averages).
3) Only include bonds that are actually broken in the reaction’s rearrangement
Conceptually, reactant bonds that do not persist into products are “broken.” For estimating “energy in,” you focus on the set of bonds in reactants that must be disrupted.
In many AP-style problems, it is assumed that the relevant reactant bonds are broken as part of the overall process of rearrangement.
If a reactant appears unchanged as a spectator (uncommon in bond-enthalpy prompts), you would not count its bonds as broken.
4) Be careful with polyatomic structures and repeated bonds
Counting errors are the most common reason “energy in” is wrong.
Count bonds per molecule, then multiply by the coefficient.
For symmetrical molecules, verify you are not undercounting identical bonds.
For molecules with multiple central atoms, check each connection (e.g., multiple C–H bonds in hydrocarbons).
Using bond energy data correctly
Units and interpretation
Bond energies are typically given in (per mole of bonds).
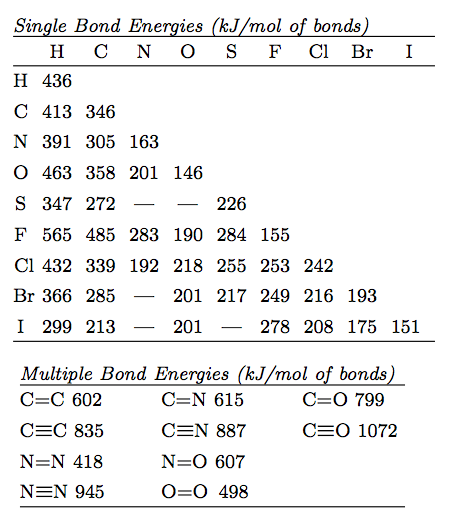
This reference table lists representative average bond energies for common single, double, and triple bonds in . It supports quick lookup of the correct bond type (e.g., C–C vs. C=C vs. C≡C) before multiplying by the counted number of bonds broken in the balanced reaction. Source
Your final is in kJ per reaction as written, after summing all relevant bond contributions.
Why your result is an estimate
Even with perfect bond counting, your “energy in” will be approximate because:
Tabulated bond energies are averages across different compounds.
Real bond energies vary with molecular environment (polarity, resonance, neighbouring atoms).
The method is most reliable for gas-phase species and less exact for condensed-phase reality.
FAQ
They are measured across many different molecules containing the same bond type.
Different chemical environments change bond strength, so tables report a mean value to support approximate thermochemical reasoning.
Use connectivity from a correct Lewis structure (which atoms are bonded and bond order).
Three-dimensional geometry usually does not change the bond count, but it can affect the true bond strength (a reason the method is approximate).
Treat them as having the appropriate bond order in the molecule:
$\text{O}_2$ contains one $\text{O=O}$
$\text{N}_2$ contains one $\text{N}\equiv\text{N}$
Then multiply by the stoichiometric coefficient.
You cannot reliably invent a value.
Common approaches are to:
use a provided “average” value for a similar bond type (if explicitly allowed), or
leave the expression in terms of unknown bond energies (if the question is symbolic).
Bond energies are defined for breaking gaseous bonds into gaseous atoms.
In condensed phases, additional interactions (solvation, lattice effects) influence the measured heat change, so the “bonds broken” estimate omits important energy contributions.
Practice Questions
Q1 (2 marks) The reaction includes breaking mole of bonds. Given , state the energy required to break these bonds.
(1)
Correct unit and clear statement that it is energy required to break the bonds (1)
Q2 (5 marks) For the reaction , estimate the energy input required to break the bonds in the reactants using: and .
Identifies bonds broken: one and one (2)
Uses or equivalent setup (1)
Calculates (1)
Gives with unit (1)

