AP Syllabus focus: ‘Estimate energy released by summing average bond energies for bonds formed in the product molecules.’
Understanding “energy out” from bond formation lets you connect molecular structure to thermal energy changes. These notes show how to identify product bonds, use average bond enthalpies correctly, and interpret the sign of the energy released.
Core idea: bond formation releases energy
When new chemical bonds form in the products, the system’s potential energy decreases and energy is transferred out of the system (typically as heat).
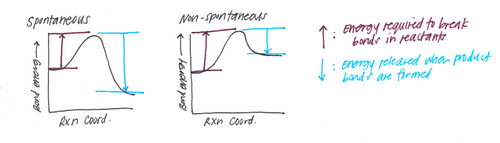
Potential-energy diagrams comparing bond breaking versus bond formation. The key takeaway is that forming a bond corresponds to moving to a lower potential-energy state, so energy is released to the surroundings, whereas breaking a bond requires energy input to separate atoms. Source
In bond enthalpy calculations, this released energy is estimated from average bond energies (tabulated values).
What “average bond energy” means
Bond energy tables list values measured as averages across many molecules in the gas phase, so your result is an estimate rather than an exact value.
Average bond enthalpy (bond energy) — the average energy required to break one mole of a specific type of bond in gaseous molecules, producing gaseous atoms (units: kJ/mol).
Because tables are defined for breaking bonds (an energy input), you must handle the sign carefully when you use them to represent forming bonds (an energy release).
Estimating “energy out”: sum of bonds formed
“Energy out” refers to the total magnitude of energy released when the product bonds form. The practical method is to count each type of bond in the products, multiply by its average bond energy, and add them.
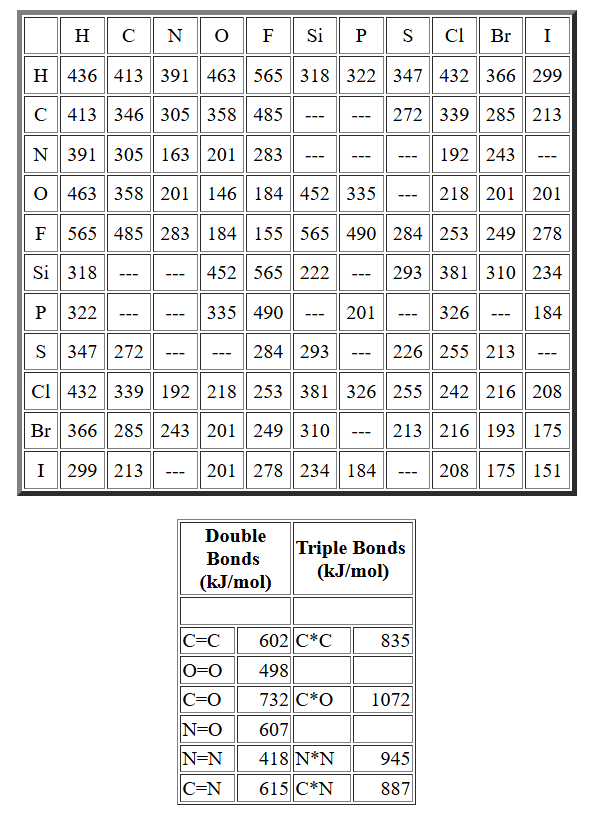
Reference table of average bond enthalpies (bond energies) in for many common single, double, and triple bonds. This is the kind of data source used to look up each before multiplying by the number of product bonds formed and summing to estimate . Source
Step-by-step procedure (products only)
Write the balanced chemical equation (so bond counts reflect the correct stoichiometric amounts).
Draw or mentally picture the Lewis structures (or reliable structural formulas) for the product molecules.
Identify the bond types formed in the products (single, double, triple; specific atom pairs such as C–H, C=O).
Count how many of each bond type appear across all products, using coefficients.
Multiply each bond count by the corresponding average bond enthalpy from the table.
Add the contributions to get the total energy out (magnitude).
Sign convention: magnitude vs. thermochemical sign
Bond energy tables give positive values for breaking bonds. If you treat “energy out” as a positive magnitude, you then apply a negative sign to represent the system’s energy change due to bond formation (energy leaving the system).
= total energy released as a positive magnitude, kJ (for the reaction as written)
= number of bonds of type formed in the products, dimensionless
= average bond enthalpy for bond type , kJ/mol
= energy change associated with forming product bonds (negative if energy is released), kJ
In words: the more (or stronger) bonds formed in the products, the larger the magnitude of energy released.
How to count bonds formed accurately
Use coefficients carefully
A common error is counting bonds in one molecule but forgetting to multiply by the stoichiometric coefficient. Always scale bond counts to match “as written” amounts of products.
Count bond types, not electron pairs
Bond energy tables distinguish bond orders and atom pairs.
Treat C–O and C=O as different bond types.
Treat N–H and O–H as different bond types.
Do not assume resonance structures create fractional bonds for this method; use the best single-structure approximation that matches the table bond types.
Polyatomic products and ionic solids
Average bond enthalpy estimates work best for covalent molecules in the gas phase. If products include ionic lattices or extended network solids, “bonds formed” may not correspond well to tabulated covalent bond energies, increasing error.
What your “energy out” estimate does and does not tell you
What it tells you
A quantitative estimate of how much energy is released when product bonds form, based on tabulated averages.
A way to compare products: forming multiple strong bonds (for example, double bonds) typically increases energy released.
What it does not guarantee
Exact experimental heat values (because bond enthalpies are averages and depend on molecular environment).
Correctness if product structures are misdrawn or if the reaction is not properly balanced.
FAQ
Bond enthalpies are averages over many gaseous molecules, but real bonds vary with molecular environment (polarity, resonance, neighbouring atoms).
Phase and intermolecular effects can also matter if the real reaction conditions are not gaseous.
Yes—when estimating energy out, you count all bonds present in the products, regardless of whether similar bonds existed in reactants.
The method treats bond breaking and bond forming as separate bookkeeping steps.
Use the best single-bond-type approximation consistent with the bond enthalpy table.
If the bonding is delocalised, any single-structure count introduces additional uncertainty.
You still count covalent bonds within the polyatomic ion using an appropriate Lewis structure.
Be cautious: delocalisation means the “bond type” may not match a single tabulated value well.
Not reliably. Ionic solids are better described by lattice energies, not discrete covalent bonds.
Using covalent bond enthalpies for such products typically gives poor estimates.
Practice Questions
(2 marks) For a reaction that forms 2 moles of H–Cl bonds, explain how the “energy out” from bond formation is estimated using an average bond enthalpy table.
States energy out is found by multiplying number of bonds formed by the average bond enthalpy value (1).
States this gives the magnitude of energy released and that bond formation corresponds to a negative energy change for the system (1).
(5 marks) A reaction produces and . Describe, in a structured method, how to estimate the total energy released from forming product bonds using average bond enthalpies. You do not need numerical values.
Uses balanced products and coefficients to scale bond counts (1).
Identifies bonds in : two C=O per molecule; applies coefficient 2 (1).
Identifies bonds in : two O–H per molecule; applies coefficient 3 (1).
Multiplies each bond count by the corresponding average bond enthalpy and sums to obtain (1).
States sign handling: energy released is taken as a positive magnitude for and corresponds to a negative contribution for bond formation (1).

