AP Syllabus focus: ‘Tables of standard enthalpies of formation provide values that can be used to calculate standard enthalpies of reactions.’
Standard enthalpy of formation tables are a compact way to encode chemical energy information. They report reference enthalpy values for substances under defined conditions, letting you interpret relative stability and later determine reaction enthalpy changes.
What a standard enthalpy of formation table contains
The quantity being tabulated:
Standard enthalpy of formation, : The enthalpy change when 1 mol of a substance is formed from its elements in their standard states under standard conditions.
A table lists a value for many substances (elements, compounds, and sometimes ions), each tied to a specific chemical formula and physical state (such as (s), (l), (g), (aq)).
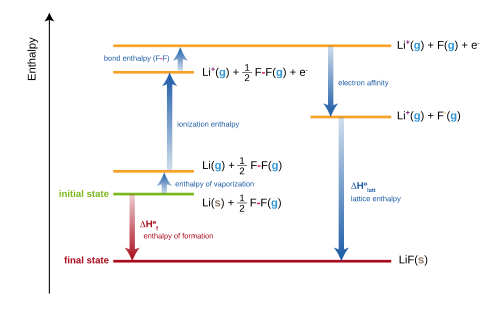
Example rows from a standard enthalpy of formation data table, showing how each entry pairs a species with a specific phase and a numerical \Delta H_f^\circ��. Source
Interpreting a value requires reading the entire entry: substance + state + number.
At its core, each value refers to a particular formation reaction that produces exactly 1 mol of the substance from its constituent elements in their standard states.
= standard enthalpy of formation, in
Standard conditions and standard states
The “” indicates a standard-state value (commonly 1 bar pressure; AP problems often use 1 atm as an approximation). Unless otherwise specified, values are typically reported near 298 K.
Standard state: The most stable form of a substance at 1 bar and a specified temperature (commonly 298 K), with solutes at a defined reference concentration.
A key reading skill is recognising that standard state is not “any pure form.” It is the thermodynamically most stable form under the stated conditions.
How to read and interpret entries
Physical state matters
Formation enthalpies depend on phase because intermolecular forces change energy. Therefore:
is different from .
Carbon may appear as graphite (s) rather than diamond (s), because graphite is the standard state at 298 K and 1 bar.
Always match the phase in the table to the substance in your chemical context; otherwise you are using the wrong reference energy.
Sign conventions: what negative and positive values imply
A formation enthalpy value tells you whether forming the substance from its elements is exothermic or endothermic:
Negative : forming the substance releases heat to the surroundings at constant pressure; the product is typically lower in enthalpy than its elements.
Positive : forming the substance requires heat input; the compound is higher in enthalpy than its elements.
These signs help you compare relative energetic “heights” of substances, but they do not, by themselves, predict reaction direction or spontaneity.
Reference point: elements in their standard states are zero
A formation table uses a consistent baseline:
for any element in its standard state is defined as 0 .
Examples include , , and (at 298 K, 1 bar). This convention makes the table internally consistent and allows values to function as reusable building blocks.
Be careful: an element not in its standard state does not have to be zero (for instance, a different allotrope or phase would not share the same reference value).
What the table “tells you” conceptually (and how it is used)
Formation values are reusable energy building blocks
Because each entry corresponds to a formation process from elements, the set of values can be combined to determine overall enthalpy changes for reactions under the same standard conditions.
This is why the AP specification emphasises that these tables “provide values that can be used to calculate standard enthalpies of reactions.”
Stoichiometry and “per mole of substance formed”
A common interpretation error is forgetting that is reported per 1 mol of the product substance as written in the table entry, not per mole of atoms or per mole of reaction as you might casually describe it. When later combining values, the numerical contribution of an entry must track the amount (in moles) of that species involved.
Units and magnitude
Values are almost always in . Large magnitudes often reflect substantial changes in bonding and structure upon formation, but you should avoid over-interpreting magnitude without considering phase and formula.
FAQ
Some tables extend formation enthalpies to ions in aqueous solution to support ionic thermochemistry.
Because isolated ions cannot be formed directly from elements in a unique way, conventions are adopted (for example, assigning a reference value to $\mathrm{H^+(aq)}$) so other ionic values can be reported consistently.
They may be obtained indirectly using measured enthalpy changes for carefully chosen reactions and combining them algebraically.
Typical sources include combustion calorimetry, solution calorimetry, and other reaction enthalpy measurements, linked through energy conservation.
Yes. Enthalpy depends on temperature because heat capacities are not zero, so warming changes the enthalpy content of substances.
298 K is a convenient reference near room temperature; using a fixed reference allows consistent comparison and combination of values.
Differences can come from:
Updated experimental measurements
Different standard-state assumptions (1 bar vs 1 atm)
Rounding conventions and significant figures
Choice of reference data sets and corrections
Small discrepancies are common and usually within stated uncertainties.
A value may be missing because the substance is unstable, poorly characterised, or has insufficient high-quality thermochemical data.
Some species (especially short-lived intermediates) are difficult to isolate under standard conditions, making reliable standard-state enthalpy assignments challenging.
Practice Questions
Question 1 (3 marks) A table lists . State what this value means and write the balanced formation equation for in standard states.
States that it is the enthalpy change to form 1 mol of from its elements in their standard states under standard conditions (1 mark)
Correct reactants in standard states: and (1 mark)
Balanced equation producing 1 mol: (1 mark)
Question 2 (6 marks) Explain why a standard enthalpy of formation table assigns to and but not necessarily to or . In your answer, comment on the meaning of “standard state” and the significance of the sign of for a compound.
States that of an element in its standard state is defined as zero by convention (1 mark)
Identifies as oxygen’s standard state at 298 K, 1 bar (1 mark)
Identifies as sodium’s standard state at 298 K, 1 bar (1 mark)
Explains that and are not standard states under those conditions (e.g., less stable form/incorrect phase) so not assigned zero (1 mark)
Defines/clarifies standard state as the most stable form at the stated pressure and temperature (1 mark)
Explains sign meaning for compounds: negative indicates exothermic formation (lower enthalpy than elements), positive indicates endothermic formation (higher enthalpy than elements) (1 mark)

