AP Syllabus focus: ‘Calculate standard reaction enthalpy using ΔH°reaction = ΣΔHf°products − ΣΔHf°reactants, applying correct signs and stoichiometric coefficients.’
Standard reaction enthalpy can be calculated from tabulated formation enthalpies by combining contributions from each species in a balanced equation.
Success depends on careful bookkeeping: physical states, stoichiometric coefficients, and algebraic signs.
What the equation is doing
This method treats a reaction as “building” reactants and products from their elements in standard states. The net energy change is the difference between the energy required to form products and the energy required to form reactants, using standard enthalpies of formation.
Standard enthalpy of formation, ΔHf°: the enthalpy change when 1 mole of a compound forms from its elements in their standard states (most stable form at 1 bar, typically 298 K).
Be attentive to the superscript “°”: you must use formation enthalpies reported for standard conditions and the correct physical state (s, l, g, aq) from the balanced chemical equation.
Calculating ΔH°reaction from ΔHf°
You will compute the standard reaction enthalpy by summing formation enthalpies for products and subtracting the corresponding sum for reactants, with stoichiometric coefficients included.
= standard enthalpy change of reaction, in (per “mole of reaction” as written)
= stoichiometric coefficient (moles of each species in the balanced equation)
= standard enthalpy of formation for a species, in
This relationship only works if the chemical equation is correctly balanced first; coefficients are part of the energy accounting.
Stepwise procedure (no arithmetic shortcuts)
Balance the overall reaction, including correct physical states when provided.
Identify every species whose ΔHf° is needed; remember that elements in their standard states have ΔHf° = 0 and usually do not contribute to the sums.
Compute the products sum:
Multiply each product’s ΔHf° by its coefficient.
Add these values.
Compute the reactants sum the same way.
Subtract: (products sum) − (reactants sum).
Report ΔH°rxn with appropriate units and sign.
Signs, coefficients, and common errors
The most frequent mistakes come from mixing up the “minus reactants” structure and the signs already embedded in tabulated ΔHf° values.
Do not flip signs of ΔHf° values just because a species is a reactant; the subtraction in the formula accounts for reactants.
A negative ΔHf° means forming the compound from elements is exothermic under standard conditions; keep the negative sign when multiplying by coefficients.
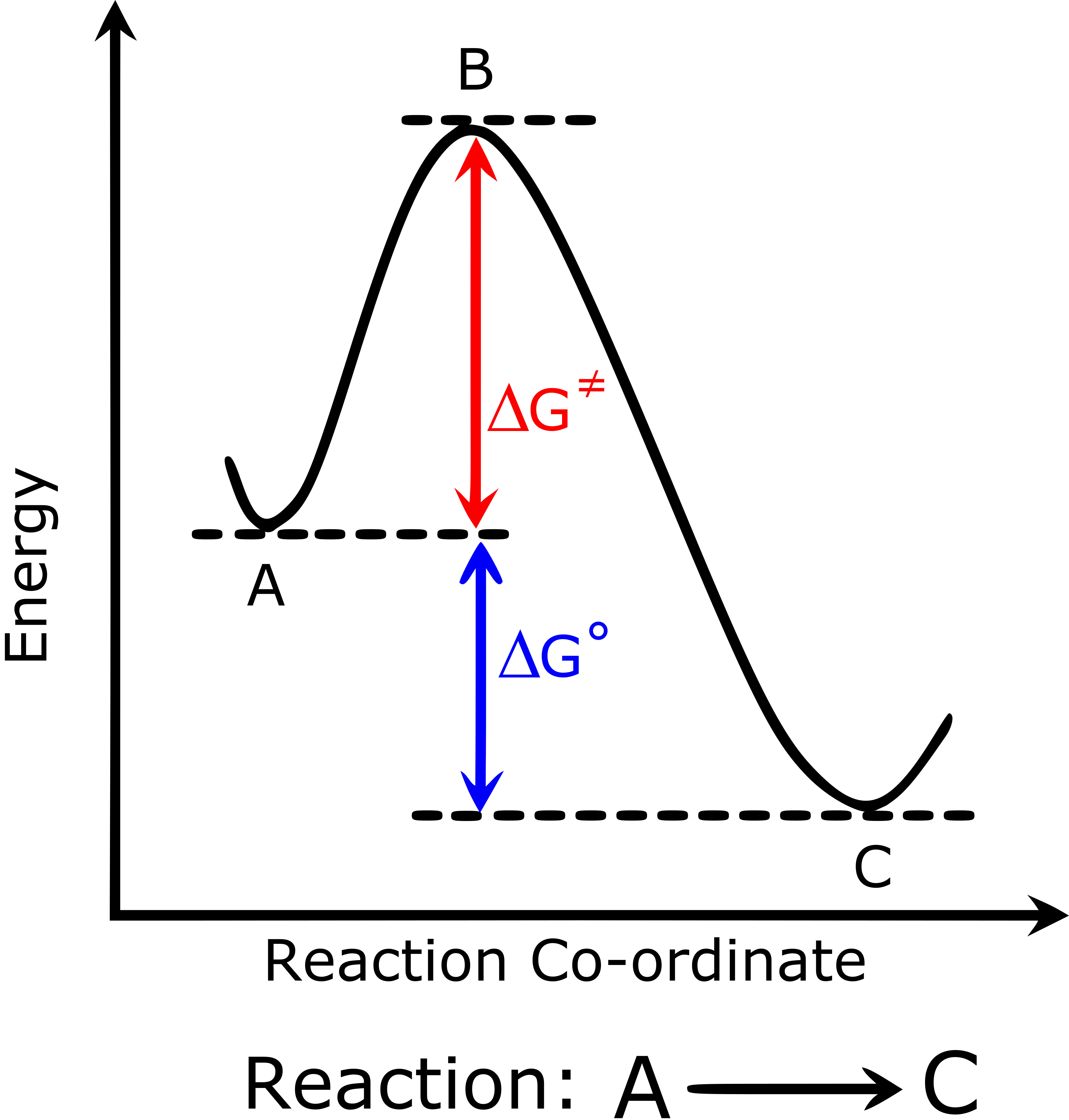
This reaction-coordinate diagram shows reactants progressing to products through a higher-energy transition state, with the vertical axis representing energy/enthalpy and the horizontal axis representing reaction progress. The vertical separation between reactant and product levels corresponds to the reaction enthalpy change , which is what your formation-enthalpy sum ultimately computes. Source
Stoichiometric coefficients scale enthalpy: if the equation uses 2 mol of a product, its ΔHf° contribution is doubled.
Use ΔHf° values that match the state:
H2O(l) and H2O(g) have different ΔHf° values; choosing the wrong one changes ΔH°rxn.
ΔH°rxn is tied to the reaction “as written.” If you later rewrite the equation (for example, to represent 1 mol of a different species), ΔH°rxn must be scaled consistently with the new coefficients.
Units and reporting conventions
ΔHf° and ΔH°rxn are commonly in kJ·mol⁻¹; keep unit consistency across all terms.
ΔH°rxn refers to the enthalpy change for the stoichiometric reaction event; interpret “per mole” as per mole of reaction (the balanced equation’s molar amounts).
Rounding should reflect the precision of the ΔHf° data source; avoid over-reporting digits after subtraction.
FAQ
It sets a consistent reference point (a “zero level”) so formation enthalpies are comparable across substances.
Because compounds are defined as forming from elements, assigning zero to the most stable elemental forms avoids arbitrary offsets in all calculated $ \Delta H^\circ_{\text{rxn}} $ values.
Use the most stable form at 1 bar (often 298 K):
Carbon: graphite (not diamond)
Oxygen: $ \text{O}_2(g) $ (not $ \text{O}(g) $ or $ \text{O}_3(g) $)
Sulfur: typically rhombic sulfur, $ \text{S}_8(s) $
If a reaction uses a non-standard form, its $ \Delta H^\circ_f $ is not zero.
You must use $ \Delta H^\circ_f $ values specifically for the aqueous ions/species shown, e.g. $ \text{Na}^+(aq) $.
Be careful: $ \text{NaCl}(s) $ and $ \text{Na}^+(aq)+\text{Cl}^-(aq) $ are different species with different $ \Delta H^\circ_f $ values.
$ \Delta H^\circ_{\text{rxn}} $ is extensive with respect to the equation: if all coefficients are multiplied by a factor $k$, then $ \Delta H^\circ_{\text{rxn}} $ is multiplied by $k$.
If you reverse the equation, the sign of $ \Delta H^\circ_{\text{rxn}} $ changes.
Match the precision of the least precise $ \Delta H^\circ_f $ value used.
In practice:
Keep a few guard digits during intermediate sums.
Round the final $ \Delta H^\circ_{\text{rxn}} $ to a sensible precision consistent with the data source.
Practice Questions
(2 marks) Use to state how you would include stoichiometric coefficients and signs when calculating .
1 mark: States that each is multiplied by its stoichiometric coefficient .
1 mark: States that you sum products then subtract the sum for reactants (not changing signs simply because a species is a reactant).
(5 marks) For the reaction , describe precisely which values are used in the calculation of , including coefficients, and identify any terms that are zero.
1 mark: Includes in the products sum.
1 mark: Includes in the products sum (correct state).
1 mark: Includes in the reactants sum.
1 mark: Includes in the reactants sum.
1 mark: States because oxygen is an element in its standard state.

