AP Syllabus focus: ‘A disturbance to a system at equilibrium causes Q to differ from K, taking the system out of equilibrium until it responds to restore agreement between Q and K.’
Chemical equilibrium is best diagnosed quantitatively: a system is at equilibrium only when its current composition matches the equilibrium condition. Disturbances change the composition, so the reaction quotient changes immediately while the equilibrium constant does not.
Core idea: disturbance creates a mismatch
At equilibrium, the system’s measured composition satisfies . A disturbance (also called a stress) changes the amounts of one or more species, so the ratio of products to reactants is no longer the equilibrium ratio.
Reaction quotient (Q). The ratio of product amounts to reactant amounts at a particular moment, each raised to stoichiometric coefficients, using current concentrations or partial pressures.
Immediately after a disturbance, the system has a new Q value but (at the same temperature) the same K value, so and the system is out of equilibrium.
What stays the same vs what changes
Equilibrium constant K (unchanged by composition changes)
K is fixed for a given reaction at a given temperature. Changing how much reactant or product is present does not redefine the equilibrium condition; it only changes whether the system currently meets that condition.
Equilibrium constant (K). The value of Q that corresponds to equilibrium for a specific reaction at a particular temperature.
Reaction quotient Q (changes instantly when you disturb the system)
Because Q depends on the current composition, it responds immediately to:
Adding a reactant or product
Removing a reactant or product
Dilution (for aqueous systems, changing volume changes concentrations)
Changing volume/pressure (for gases, changing volume changes partial pressures)
Using Q and K to recognise “not at equilibrium”
To decide whether a disturbed system is still at equilibrium, you:
Write the balanced reaction.
Write the corresponding Q expression (same form as K).
Substitute the post-disturbance concentrations or partial pressures to get Q.
Compare with the given K (at that temperature).
= current molar concentration,
= stoichiometric coefficient (power on each species)
= “product of”: multiply all required terms together
A key interpretation is purely diagnostic: if , the system cannot be at equilibrium, so there must be a net chemical change occurring (even if the change is not instantly visible).
How disturbances typically change Q (qualitative patterns)
Because Q is a ratio, disturbances push it in predictable ways:
Add product → numerator increases → Q increases
Remove product → numerator decreases → Q decreases
Add reactant → denominator increases → Q decreases
Remove reactant → denominator decreases → Q increases
For gases, a sudden change in volume changes partial pressures, which can change Q even if moles have not changed. For solutions, dilution lowers all solute concentrations, which can also shift Q because exponents and which species appear in Q determine whether the ratio changes.
What “out of equilibrium” means in practice
When a disturbance makes Q differ from K, the current mixture has the “wrong” product-to-reactant ratio for equilibrium at that temperature.
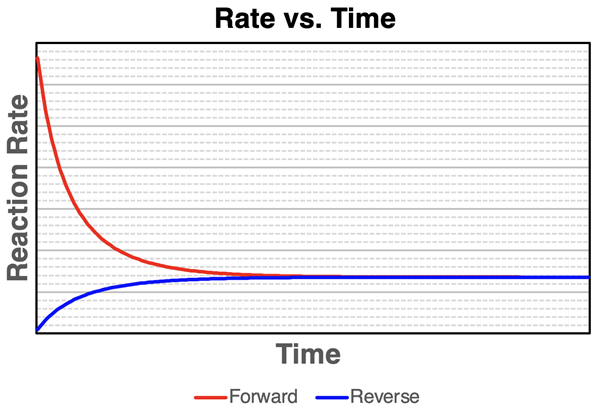
Rate–time curves for a reversible reaction show the forward rate decreasing and the reverse rate increasing until they become equal. The intersection corresponds to dynamic equilibrium: reactions continue microscopically, but there is no net macroscopic change. After a disturbance, the rates are no longer equal, and the system evolves until the rates match again. Source
As a result:
The forward and reverse reaction rates are no longer equal.
A net reaction occurs, changing composition over time.
Q will change as composition changes, trending back toward the condition .
FAQ
Yes. If all species that appear in Q are scaled by exactly the same factor and the overall powers cancel, the ratio may remain the same.
This depends on the stoichiometric exponents in Q, not on whether the system “looks” changed.
$Q$ is a calculation from the current composition, so it updates as soon as the composition is changed.
Returning toward equilibrium requires reaction progress, which takes time because it depends on collision frequency and activation energy.
At constant volume, adding an inert gas does not change the partial pressures of reacting gases, so $Q_p$ does not change.
At constant pressure, volume increases and partial pressures of reacting gases decrease, so $Q_p$ can change.
For AP Chemistry, you compute $Q_c$ with concentrations and $Q_p$ with partial pressures; the comparison is only valid when Q and K are expressed in the same form.
Be consistent with which form is being used for the reaction conditions.
Often there is no immediate visible sign, but you may detect changing properties over time, such as pressure drift, absorbance/colour intensity drift, or pH drift.
The key idea is that any time the measurable composition changes, $Q$ is changing and may not equal K.
Practice Questions
(3 marks) For , the system is at equilibrium at constant temperature. A small amount of is injected into the vessel. State what happens to immediately, and how compares to immediately after the injection.
increases immediately (1)
unchanged at constant temperature (1)
Immediately after injection, so the system is no longer at equilibrium (1)
(6 marks) Consider at constant temperature, initially at equilibrium in a closed container. The volume is suddenly decreased. Explain, in terms of and , why the system is taken out of equilibrium immediately after the change. Use an expression for in your answer.
Writes a correct form of : (1)
States that decreasing volume increases partial pressures of all gases (1)
Recognises that because the exponents differ, the ratio changes so changes (1)
States is unchanged because temperature is constant (1)
Concludes that immediately after compression, so the system is out of equilibrium (1)
Links “out of equilibrium” to unequal forward/reverse rates (net reaction must occur) (1)

