AP Syllabus focus: ‘After a stress, concentrations or partial pressures redistribute so that Q returns to equal K, establishing a new equilibrium state.’
When an equilibrium system is disturbed, it temporarily becomes “out of balance.” The mixture then responds by changing its composition until the equilibrium condition is satisfied again, producing a new equilibrium with constant macroscopic properties.
What “re-establishing a new equilibrium” means
A system at equilibrium has a fixed relationship between the amounts of reactants and products (at a given temperature). A stress (such as changing a species amount, pressure/volume for gases, or other conditions) disrupts that relationship, so the system is no longer at equilibrium.
At that moment, the composition corresponds to a value of Q that does not match K, and the reaction proceeds with a net direction until the equality is restored.
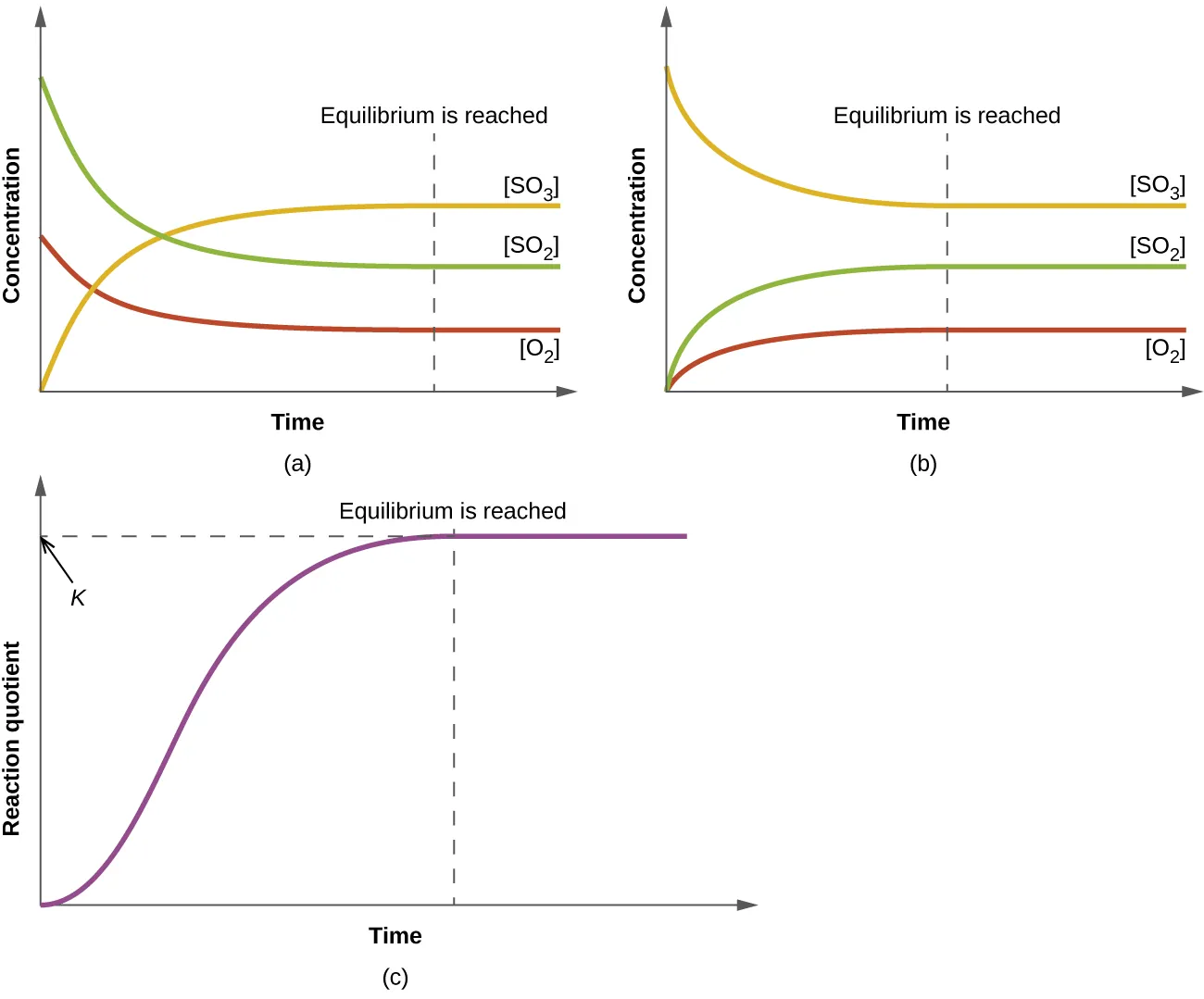
Reaction progress causes to change continuously until it levels off at the equilibrium value, where . The “leveling off” emphasizes that equilibrium is reached when composition stops changing macroscopically, not when the reaction stops microscopically. Source
Reaction quotient (Q): A ratio of product-to-reactant amounts (using concentrations or partial pressures) calculated at any time, with the same form as the equilibrium constant expression.
A new equilibrium is reached when the mixture’s composition has redistributed so that the equilibrium condition is again met.
New equilibrium state: The post-disturbance condition in which reactants and products are present and macroscopic properties are constant because Q has returned to equal K.
The mathematical condition for the new equilibrium
For a general reaction, the system “re-equilibrates” by changing species amounts until Q equals K again.
= Concentration at that instant (typically mol/L)
= Partial pressure at that instant (typically atm)
Because Q uses current amounts, it changes immediately when a stress changes any concentration or partial pressure. Re-establishing equilibrium is the process of the system evolving until Q once again equals the constant K (for that temperature).
How the system redistributes after a stress
Immediate effect: Q changes first
Right after a disturbance:
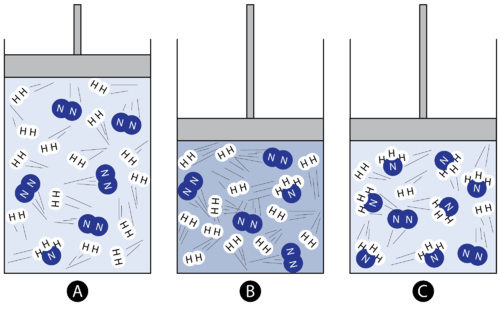
A pressure/volume stress changes gas partial pressures instantly, so the system is no longer at equilibrium. The diagram illustrates the system shifting composition in the direction that reduces the stress (favoring the side with fewer moles of gas) until a new equilibrium is reached. Source
The amounts of certain species may change abruptly (for example, adding or removing a reactant/product, or changing gas volume).
Therefore Q changes immediately because it is computed from the instantaneous composition.
The value of K does not “chase” Q; instead, the system composition changes until Q matches K again (at the same temperature).
Net reaction occurs: rates are no longer balanced
Once Q differs from K, the forward and reverse processes are no longer balanced in their effects on the mixture:
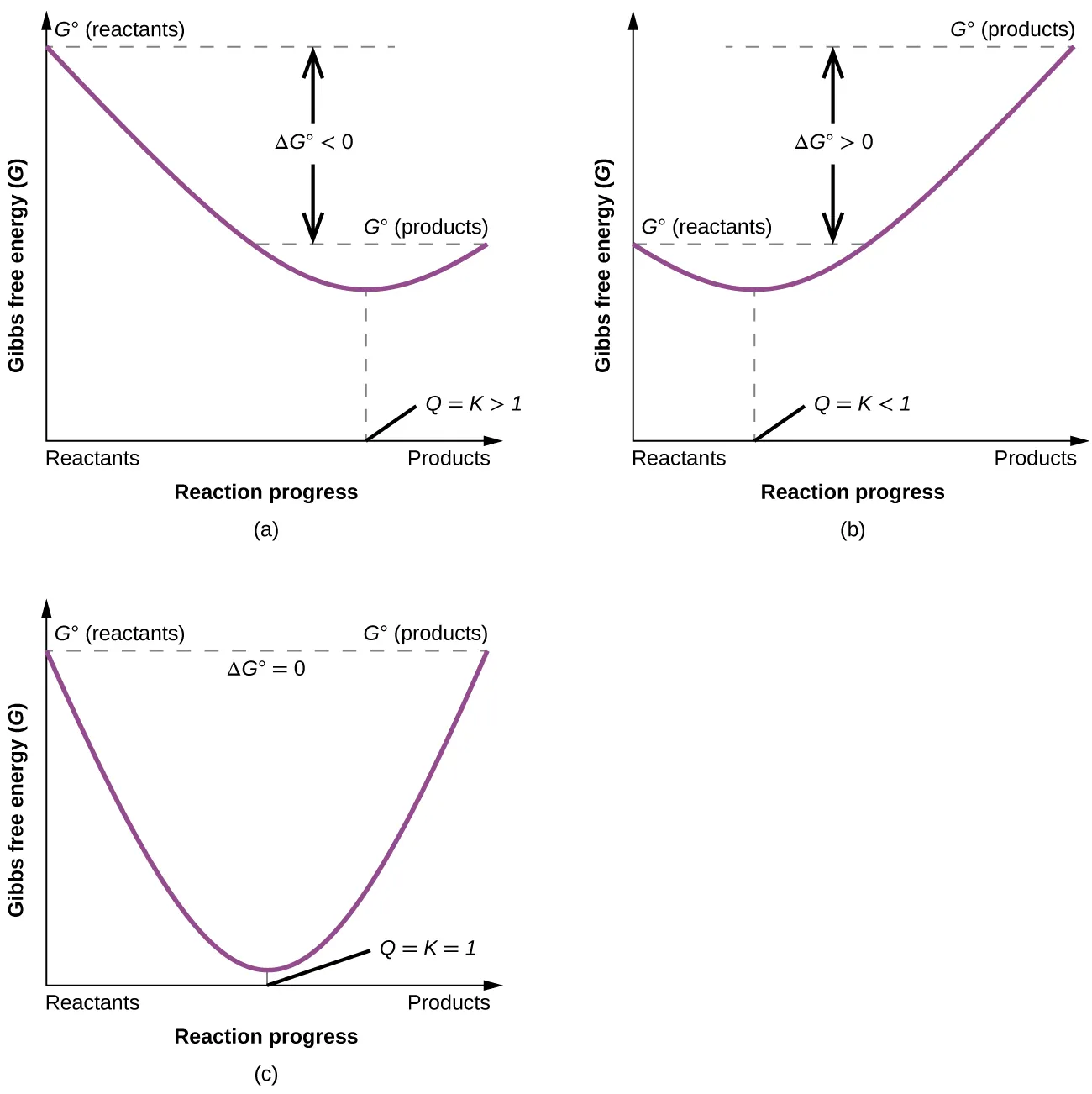
These curves show Gibbs free energy versus reaction progress, with the minimum occurring at equilibrium where . When , the system moves spontaneously “downhill” in until it reaches that minimum, corresponding to the new equilibrium composition. Source
One direction becomes faster in its net effect than the other, so concentrations/partial pressures begin changing in a definite net direction.
As the reaction proceeds, the changing composition continuously updates Q, moving it toward K.
This redistribution is not “trying to use up” an added substance; it is specifically adjusting the product-to-reactant ratio until it matches K.
Approach to the new equilibrium: changing rates and leveling off
During the shift:
The net rate is greatest soon after the stress (largest mismatch between Q and K).
As the composition changes, the mismatch shrinks, and the net rate decreases.
Eventually, the system reaches a point where Q equals K again; at that point, the forward and reverse rates are equal, and macroscopic properties stop changing.
Recognising that a new equilibrium has been established
In a closed system at constant temperature, you can identify the new equilibrium by either of these equivalent ideas:
Constant macroscopic properties over time: concentrations/partial pressures become constant (even though particles continue reacting).
The equilibrium condition: calculating Q from measured amounts gives Q = K.
Operationally, “re-establishing a new equilibrium” means the system’s measurable composition settles to new constant values (not necessarily the original ones), consistent with the same K at that temperature.
Key features students often miss
Equilibrium does not mean equal amounts. The new equilibrium can contain mostly reactants or mostly products; it only requires Q = K.
Both reactants and products are present at the new equilibrium (for a reversible reaction in a closed system).
The redistribution changes multiple species at once because of stoichiometry: as the reaction proceeds in one net direction, all species adjust according to the balanced equation.
FAQ
Yes. The new equilibrium generally has different equilibrium amounts from the original state.
Observable properties depend on the new equilibrium composition, not on the requirement to “undo” the disturbance.
A larger disturbance typically creates a larger initial mismatch between $Q$ and $K$, which can increase the initial net rate.
However, the time to settle also depends on activation energy and the reaction mechanism.
Yes. Small measurement uncertainties in concentration, pressure, or temperature can cause calculated $Q$ to fluctuate around $K$.
In practice, equilibrium is inferred when values become steady within experimental tolerance.
Some systems can form metastable states (e.g., delayed phase changes), where the observable composition changes slowly.
Once nucleation or a pathway becomes available, the system proceeds toward the equilibrium condition again.
If matter continuously enters or leaves, the system may not reach a true equilibrium state because composition is being driven externally.
Instead, it may approach a steady state that is not characterised by $Q=K$.
Practice Questions
(2 marks) A system is at equilibrium with . A small amount of product is suddenly added at constant temperature. State what happens to immediately, and what must be true when the new equilibrium is reached.
increases immediately (or immediately after adding product). (1)
At the new equilibrium, returns to equal (so again). (1)
(5 marks) A reversible gas-phase reaction is at equilibrium in a sealed container at constant temperature. The container volume is suddenly decreased. Explain, in terms of and , how a new equilibrium is established and what happens to measurable amounts over time.
Recognises that the disturbance changes partial pressures/concentrations immediately, so changes immediately. (1)
States that remains constant at the same temperature. (1)
Explains that because , there is a net reaction in one direction, changing composition. (1)
Explains that as composition changes, moves back towards . (1)
States that a new equilibrium is reached when again and measurable partial pressures/concentrations become constant. (1)

