AP Syllabus focus: ‘The solubility of a substance can be calculated from its Ksp, and Ksp values can be used to predict the relative solubility of different salts.’
Solubility equilibria connect what dissolves in a saturated solution to an equilibrium constant. By translating Ksp into ion concentrations using stoichiometry, you can calculate solubility and compare how readily different ionic solids dissolve.
Core ideas: Ksp and solubility
A slightly soluble ionic solid establishes a dissolution equilibrium between the solid and its aqueous ions.
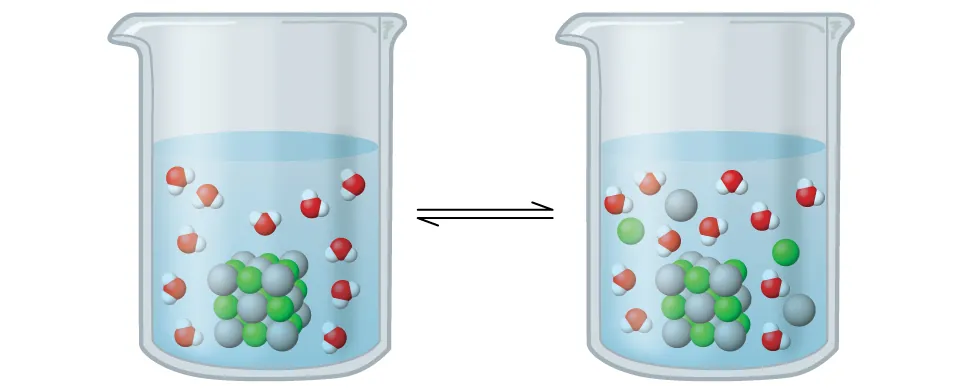
OpenStax illustrates a sparingly soluble salt (AgCl) in water as a heterogeneous equilibrium: undissolved solid persists while a small, steady concentration of and ions exists in solution. The diagram helps connect the particulate picture of dissolution/precipitation to the idea that is defined using only aqueous ion concentrations. Source
The equilibrium constant for this process is the solubility-product constant.
Molar solubility — the number of moles of an ionic solid that dissolve per litre of solution to form a saturated solution (units: ).
When converting between Ksp and solubility, the key skill is writing ion concentrations in terms of molar solubility using the balanced dissolution reaction.
Writing the Ksp expression
Write the dissolution reaction with correct stoichiometric coefficients.
Write Ksp as the product of equilibrium ion concentrations raised to those coefficients.
Do not include the solid in the expression (its “concentration” is constant).
= solubility-product constant (unit depends on powers used)
= equilibrium molar concentration of cation,
= equilibrium molar concentration of anion,
= stoichiometric coefficients from
= molar solubility in pure water,
A correct expression is essential because the stoichiometric coefficients become exponents, strongly affecting the relationship between Ksp and s.
Calculating solubility from Ksp (no worked arithmetic)
To calculate solubility, you conceptually follow these steps:
Balance the dissolution equation (this sets the ion ratios).
Let the molar solubility be (mol of solid dissolved per litre).
Convert to equilibrium ion concentrations using stoichiometry:
For : ,
For : ,
For : ,
Substitute these expressions into the Ksp expression to form an equation in .
Solve for , then interpret as the molar solubility.
Keep the interpretation clear: Ksp is not solubility; it is an equilibrium constant built from ion concentrations, which depend on how many ions each formula unit produces.
Comparing solubilities using Ksp
Ksp values can be used to predict relative solubility, but only with appropriate care.
Safe comparisons (same ion stoichiometry)
If two salts produce ions in the same ratio (same exponents in Ksp), then:
Larger Ksp generally means higher molar solubility. Examples of “same form” comparisons include:
vs (both 1:1)
vs (both 1:2)
Comparisons requiring conversion to molar solubility
If salts have different stoichiometries, comparing Ksp directly can be misleading because exponents differ.
You should compare molar solubilities (or calculated ion concentrations) instead of raw Ksp values.
A salt that produces more ions per formula unit can have a different than expected from Ksp alone.
Common interpretation checks
A very small Ksp indicates a solid that dissolves only slightly (low equilibrium ion concentrations).
A larger Ksp indicates more dissolved ions at equilibrium, but the resulting molar solubility still depends on stoichiometry.
FAQ
In AP Chemistry, $K_{sp}$ is commonly computed from concentrations without normalising to a standard state, so “units” can appear from algebra. Comparisons still work when handled consistently.
$K_{sp}$ is temperature-dependent. If temperature changes, you must use the $K_{sp}$ value at that temperature; otherwise the calculated solubility will be incorrect.
Yes. Different stoichiometries change the exponents in the $K_{sp}$ expression, so the same $s$ can produce different products of ion concentrations.
Compare the equilibrium ion concentrations or total ionic concentration (e.g., $[cation]+[anion]$). Molar solubility $s$ counts dissolved formula units, not total ions.
Real solutions can deviate from ideality (activity effects), and impurities or incomplete equilibration can shift measured ion concentrations away from the idealised $K_{sp}$ prediction.
Practice Questions
Q1 (2 marks) Silver chloride has at 25°C. Write the expression for AgCl(s) and state the relationship between and in a saturated solution.
(1)
In saturation from AgCl, (1)
Q2 (5 marks) For each salt below, write (i) the dissolution equation, (ii) the expression, and (iii) the expression for in terms of molar solubility in pure water: (a) (b) .
(a) (1); (1); (1)
(b) (1); and (1)

