AP Syllabus focus: ‘Measured molar solubility (ion concentrations) in a saturated solution can be used to calculate the Ksp of a slightly soluble ionic compound.’
Slightly soluble ionic solids establish an equilibrium between undissolved solid and dissolved ions. If you can measure the ion concentrations in a saturated solution, you can determine quantitatively from stoichiometry.
Core idea: saturation links concentration to equilibrium
A saturated solution contains the maximum dissolved amount of a salt at a given temperature, with undissolved solid present.
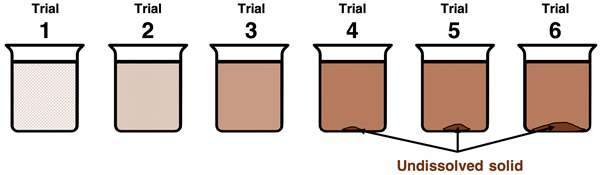
Sequence of beakers illustrating a sparingly soluble salt as more solid is added: the solution concentration increases until the saturation point is reached. Beyond saturation, additional solid accumulates at the bottom while the dissolved ion concentrations remain constant—evidence of equilibrium between solid and ions in solution. Source
This ensures the dissolution reaction is truly at equilibrium, so the equilibrium-constant expression applies.
Key term used in calculations
Molar solubility (): the number of moles of an ionic compound that dissolve per litre of solution to form a saturated solution (units: ).
Molar solubility is often not measured directly; instead, you may measure equilibrium ion concentrations (for example, via conductivity, titration, or spectroscopy) and relate them to using the balanced dissolution equation.
Writing the dissolution reaction correctly
To calculate , first write the balanced dissolution equilibrium for the salt. For a generic salt :
Include the solid as a reactant:
Write aqueous ions with correct stoichiometric coefficients and charges
Ensure the equation is balanced for both atoms and charge
The stoichiometric coefficients determine how ion concentrations depend on (or on each other if is not explicitly used).
From measured ion concentrations to
For slightly soluble salts, AP Chemistry typically treats dilute aqueous solutions as ideal enough that concentrations can be used in place of activities. Solids are omitted from the equilibrium expression.
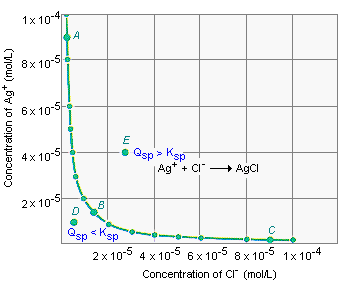
Plot showing combinations of ion concentrations that satisfy the equilibrium condition for a sparingly soluble salt (example shown for a 1:1 salt like AgCl). Points on the curve represent states where the ion product equals ; points off the curve correspond to (unsaturated) or (supersaturated/precipitation until equilibrium is restored). Source
= solubility-product constant (unitless in AP practice)
= equilibrium molar concentration of the cation,
= equilibrium molar concentration of the anion,
= stoichiometric coefficients from
To use measured data, connect the measured equilibrium concentrations to the dissolution stoichiometry:
If and the molar solubility is , then ideally:
If ion concentrations are measured directly, you may not need at all; substitute the measured equilibrium ion concentrations into the expression.
If only one ion concentration is measured:
Use stoichiometric ratios to infer the other ion concentration (e.g., if is known, then ).
Practical checklist for “find from molar solubility”
Confirm the solution is saturated (equilibrium established with excess solid).
Write the balanced dissolution equation.
Translate molar solubility () into equilibrium ion concentrations using coefficients.
Substitute equilibrium ion concentrations into the expression.
Report to a reasonable number of significant figures consistent with the measured data.
FAQ
Because $K_{sp}$ applies at equilibrium for the dissolution reaction. Undissolved solid confirms saturation, meaning ion concentrations are equilibrium values rather than just “some dissolved amount” below saturation.
In more concentrated ionic solutions, electrostatic interactions make activities smaller than concentrations. Using $[,]$ can then give an “apparent” $K_{sp}$. A more rigorous approach uses activity coefficients $\gamma$ so $a_i=\gamma_i[i]$.
Common approaches include:
Titration (e.g., precipitating an ion with a standard solution)
Conductivity (relating total ion concentration to measured conductance)
Ion-selective electrodes (direct potentiometric measurement)
Water of crystallisation changes the molar mass of the solid but not the ion ratio produced upon dissolution. If molar solubility is obtained from mass dissolved, you must use the hydrate’s formula mass to convert mass to moles correctly.
If an ion is basic or acidic (e.g., $CO_3^{2-}$, $S^{2-}$, $F^-$), it can react with water or $H^+$, reducing the free ion concentration used in $K_{sp}$. You may need to distinguish total dissolved species from the equilibrium free ion $[,]$.
Practice Questions
(1–3 marks) Silver chloride has molar solubility at a fixed temperature. Write an expression for and determine in terms of .
Writes dissolution: (1)
Writes (1)
Uses so (1)
(4–6 marks) A saturated solution of is analysed and found to contain . Calculate for at this temperature, stating any stoichiometric relationships you use.
Writes dissolution: (1)
Correct expression: (1)
Stoichiometric link: (or equivalent reasoning) (2)
Substitutes: (1)
Calculates (allow sensible rounding) (1)

